VDM 11Potent, selective anandamide transport inhibitor CAS# 313998-81-1 |
- Anguizole
Catalog No.:BCC1365
CAS No.:442666-98-0
- Asunaprevir (BMS-650032)
Catalog No.:BCC1374
CAS No.:630420-16-5
- Balapiravir
Catalog No.:BCC1396
CAS No.:690270-29-2
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 313998-81-1 | SDF | Download SDF |
| PubChem ID | 9887748 | Appearance | Powder |
| Formula | C27H39NO2 | M.Wt | 409.61 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in ethanol (supplied pre-dissolved in anhydrous ethanol, 5mg/ml) | ||
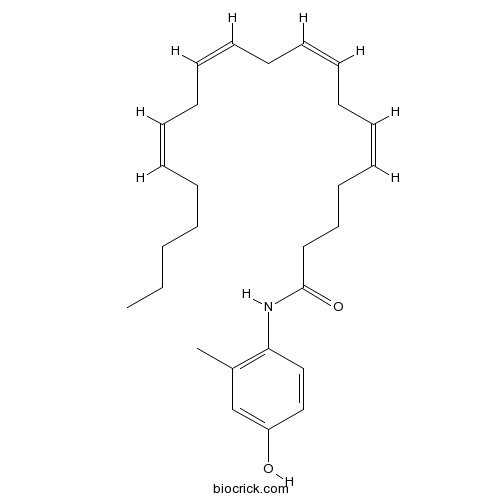
| Chemical Name | (5Z,8Z,11Z,14Z)-N-(4-hydroxy-2-methylphenyl)icosa-5,8,11,14-tetraenamide | ||
| SMILES | CCCCCC=CCC=CCC=CCC=CCCCC(=O)NC1=C(C=C(C=C1)O)C | ||
| Standard InChIKey | WUZWFRWVRHLXHZ-ZKWNWVNESA-N | ||
| Standard InChI | InChI=1S/C27H39NO2/c1-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-27(30)28-26-22-21-25(29)23-24(26)2/h7-8,10-11,13-14,16-17,21-23,29H,3-6,9,12,15,18-20H2,1-2H3,(H,28,30)/b8-7-,11-10-,14-13-,17-16- | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | A potent and selective inhibitor of the anandamide membrane transporter (AMT) with IC50 values of 4-11 μM. Displays negligible agonist activity at the hVR1 receptor and very weak action at CB1 and CB2 receptors. Ki values are > 5-10 μM at CB1 and CB2. Active in vivo. Also available in water-soluble emulsion. |

VDM 11 Dilution Calculator

VDM 11 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.4413 mL | 12.2067 mL | 24.4135 mL | 48.8269 mL | 61.0337 mL |
| 5 mM | 0.4883 mL | 2.4413 mL | 4.8827 mL | 9.7654 mL | 12.2067 mL |
| 10 mM | 0.2441 mL | 1.2207 mL | 2.4413 mL | 4.8827 mL | 6.1034 mL |
| 50 mM | 0.0488 mL | 0.2441 mL | 0.4883 mL | 0.9765 mL | 1.2207 mL |
| 100 mM | 0.0244 mL | 0.1221 mL | 0.2441 mL | 0.4883 mL | 0.6103 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- [cPP1-7,NPY19-23,Ala31,Aib32,Gln34] - hPancreatic Polypeptide
Catalog No.:BCC5750
CAS No.:313988-89-5
- PU 02
Catalog No.:BCC6265
CAS No.:313984-77-9
- o-3M3FBS
Catalog No.:BCC7210
CAS No.:313981-55-4
- FLI-06
Catalog No.:BCC5110
CAS No.:313967-18-9
- [Des-octanoyl]-Ghrelin (human)
Catalog No.:BCC7304
CAS No.:313951-59-6
- 13-Oxo-9,11-octadecadienoic acid
Catalog No.:BCC8437
CAS No.:31385-09-8
- PD 118057
Catalog No.:BCC7499
CAS No.:313674-97-4
- Bombesin
Catalog No.:BCC5708
CAS No.:31362-50-2
- INH1
Catalog No.:BCC6040
CAS No.:313553-47-8
- T0070907
Catalog No.:BCC2261
CAS No.:313516-66-4
- VU 590 dihydrochloride
Catalog No.:BCC7803
CAS No.:313505-85-0
- Reversan
Catalog No.:BCC7764
CAS No.:313397-13-6
- Evans Blue tetrasodium salt
Catalog No.:BCC6815
CAS No.:314-13-6
- (R)-(-)-Apomorphine hydrochloride
Catalog No.:BCC7250
CAS No.:314-19-2
- BPTES
Catalog No.:BCC6506
CAS No.:314045-39-1
- IU1
Catalog No.:BCC2086
CAS No.:314245-33-5
- Isotachioside
Catalog No.:BCN5230
CAS No.:31427-08-4
- Nocodazole
Catalog No.:BCC3826
CAS No.:31430-18-9
- 4-Amino-3-nitrobenzophenone
Catalog No.:BCC8682
CAS No.:31431-19-3
- Mebendazole
Catalog No.:BCC9016
CAS No.:31431-39-7
- 6-Methoxysalicylic Acid
Catalog No.:BCC8288
CAS No.:3147-64-6
- Sunifiram
Catalog No.:BCC4167
CAS No.:314728-85-3
- Crotaline
Catalog No.:BCN4983
CAS No.:315-22-0
- Allopurinol
Catalog No.:BCC3720
CAS No.:315-30-0
Revealing the role of the endocannabinoid system modulators, SR141716A, URB597 and VDM-11, in sleep homeostasis.[Pubmed:27746343]
Neuroscience. 2016 Dec 17;339:433-449.
The endocannabinoid system comprises receptors (CB1 and CB2 cannabinoid receptors), enzymes (Fatty Acid Amide Hydrolase [FAAH], which synthesizes the endocannabinoid anandamide), as well as the anandamide membrane transporter (AMT). Importantly, previous experiments have demonstrated that the endocannabinoid system modulates multiple neurobiological functions, including sleep. For instance, SR141716A (the CB1 cannabinoid receptor antagonist) as well as URB597 (the FAAH inhibitor) increase waking in rats whereas VDM-11 (the blocker of the AMT) enhances sleep in rodents. However, no further evidence is available regarding the neurobiological role of the endocannabinoid system in the homeostatic control of sleep. Therefore, the aim of the current experiment was to test if SR141716A, URB597 or VDM-11 would modulate the sleep rebound after sleep deprivation. Thus, these compounds were systemically injected (5, 10, 20mg/kg; ip; separately each one) into rats after prolonged waking. We found that SR141716A and URB597 blocked in dose-dependent fashion the sleep rebound whereas animals treated with VDM-11 displayed sleep rebound during the recovery period. Complementary, injection after sleep deprivation of either SR141716A or URB597 enhanced dose-dependently the extracellular levels of dopamine (DA), norepinephrine (NE), epinephrine (EP), serotonin (5-HT), as well as adenosine (AD) while VDM-11 caused a decline in contents of these molecules. These findings suggest that SR141716A or URB597 behave as a potent stimulants since they suppressed the sleep recovery period after prolonged waking. It can be concluded that elements of the endocannabinoid system, such as the CB1 cannabinoid receptor, FAAH and AMT, modulate the sleep homeostasis after prolonged waking.
AM404 and VDM 11 non-specifically inhibit C6 glioma cell proliferation at concentrations used to block the cellular accumulation of the endocannabinoid anandamide.[Pubmed:12698235]
Arch Toxicol. 2003 Apr;77(4):201-7.
AM404 [ N-(4-hydroxyphenyl)arachidonylamide] and VDM 11 [(5 Z,8 Z,11 Z,14 Z)- N-(4-hydroxy-2-methylphenyl)-5,8,11,14-eicosatetraenamide] are commonly used to prevent the cellular accumulation of the endocannabinoid anandamide, and thereby to potentiate its actions. However, it has been reported that AM404 can produce an influx of calcium into cells, which might be expected to have deleterious effects on cell proliferation. In the present study, AM404 and VDM 11 were found to reduce C6 glioma cell proliferation with IC(50) values of 4.9 and 2.7 microM, respectively. The inhibition of cell proliferation following a 96-h exposure was not accompanied by dramatic caspase activation, and was not prevented by either a combination of cannabinoid and vanilloid receptor antagonists, or by the antioxidant alpha-tocopherol, suggestive of a non-specific mode of action. Similar results were seen with palmitoylisopropylamide, although this compound only produced significant inhibition of cell proliferation at 30 microM concentrations. AM404 (1 microM), VDM 11 (1 microM) and palmitoylisopropylamide (3-30 microM), i.e. concentrations producing relatively modest effects on cell proliferation per se, reduced the vanilloid receptor-mediated antiproliferative effects of anandamide, as would be expected for compounds preventing the cellular accumulation of anandamide (and thereby access to its binding site on the vanilloid receptor). It is concluded that concentrations of AM404 and VDM 11 that are generally used to reduce the cellular accumulation of anandamide have deleterious effects upon cell proliferation, and that lower concentrations of these compounds may be more appropriate to use in vitro.
The anandamide membrane transporter inhibitor, VDM-11, modulates sleep and c-Fos expression in the rat brain.[Pubmed:18822353]
Neuroscience. 2008 Nov 11;157(1):1-11.
Endogenous cannabinoids or endocannabinoids are lipid molecules that have a variety of biological actions, most notably via activation of the cannabinoid receptors. The family of endocannabinoids includes arachidonoylethanolamide (ANA) which modulates different behaviors, such as sleep. However, it is unknown whether pharmacological elevation of ANA endogenous levels might induce sleep. VDM 11 [(5 Z,8 Z,11 Z,14 Z)-N-(4-hydroxy-2-methylphenyl)-5,8,11,14-eicosatetraenamide] is commonly used as an inhibitor of ANA cellular uptake, and thereby to potentiate its actions. In this study we have examined whether VDM-11 exerts any effect on the sleep-wake cycle and c-Fos expression in brain areas. When assayed alone in rats, VDM-11 (10 or 20 microg/5 microL, i.c.v.) at the beginning of the lights-off period, reduced wakefulness and increased sleep. The CB(1) cannabinoid receptor antagonist, SR141716A, partially reversed the effects of VDM-11 on sleep. Additionally, VDM-11 enhanced c-Fos expression in sleep-related brain areas such as the anterior hypothalamic area, paraventricular thalamic nucleus, and pedunculopontine tegmental nucleus. It is concluded that VDM-11 displays sleep-inducing properties and these effects slightly, albeit significantly, are reversed using SR141716A. Furthermore, c-Fos data suggest a possible underlying neuroanatomical substrate of the sleep-inducing properties of VDM-11. We report evidence suggesting that VDM-11 might be considered for the development of new pharmacological and pharmaceutical approaches to treat sleep disorders such as insomnia.
The administration of endocannabinoid uptake inhibitors OMDM-2 or VDM-11 promotes sleep and decreases extracellular levels of dopamine in rats.[Pubmed:23238438]
Physiol Behav. 2013 Jan 17;109:88-95.
The family of the endocannabinoid system comprises endogenous lipids (such as anandamide [ANA]), receptors (CB(1)/CB(2) cannabinoid receptors), metabolic enzymes (fatty acid amide hydrolase [FAAH]) and a putative membrane transporter (anandamide membrane transporter [AMT]). Although the role of ANA, FAAH or the CB(1) cannabinoid receptor in sleep modulation has been reported, the effects of the inhibition of AMT on sleep remain unclear. In the present study, we show that microdialysis perfusion in rats of AMT inhibitors, (9Z)-N-[1-((R)-4-hydroxbenzyl)-2-hydroxyethyl]-9-octadecenamide (OMDM-2) or N-(4-hydroxy-2-methylphenyl)-5Z,8Z,11Z,14Z-eicosatetraenamide (VDM-11; 10, 20 or 30 muM; each compound) delivered into the paraventricular thalamic nucleus (PVA) increased sleep and decreased waking. In addition, the infusion of compounds reduced the extracellular levels of dopamine collected from nucleus accumbens. Taken together, these findings illustrate a critical role of AMT in sleep modulation.
Experimental parkinsonism alters endocannabinoid degradation: implications for striatal glutamatergic transmission.[Pubmed:12177188]
J Neurosci. 2002 Aug 15;22(16):6900-7.
Cannabinoid receptors and their endogenous ligands have been recently identified in the brain as potent inhibitors of neurotransmitter release. Here we show that, in a rat model of Parkinson's disease induced by unilateral nigral lesion with 6-hydroxydopamine (6-OHDA), the striatal levels of anandamide, but not that of the other endocannabinoid 2-arachidonoylglycerol, were increased. Moreover, we observed a decreased activity of the anandamide membrane transporter (AMT) and of the anandamide hydrolase [fatty acid amide hydrolase (FAAH)], whereas the binding of anandamide to cannabinoid receptors was unaffected. Spontaneous glutamatergic activity recorded from striatal spiny neurons was higher in 6-OHDA-lesioned rats. Inhibition of AMT by N-(4-hydroxyphenyl)-arachidonoylamide (AM-404) or by VDM11, or stimulation of the cannabinoid CB1 receptor by HU-210 reduced glutamatergic spontaneous activity in both naive and 6-OHDA-lesioned animals to a similar extent. Conversely, the FAAH inhibitors phenylmethylsulfonyl fluoride and methyl-arachidonoyl fluorophosphonate were much more effective in 6-OHDA-lesioned animals. The present study shows that inhibition of anandamide hydrolysis might represent a possible target to decrease the abnormal cortical glutamatergic drive in Parkinson's disease.
The uptake by cells of 2-arachidonoylglycerol, an endogenous agonist of cannabinoid receptors.[Pubmed:11277920]
Eur J Biochem. 2001 Apr;268(7):1982-9.
It is not yet clear if the endocannabinoid 2-arachidonoylglycerol (2-AG) is transported into cells through the same membrane transporter mediating the uptake of the other endogenous cannabinoid, anandamide (N-arachidonoylethanolamine, AEA), and whether this process (a) is regulated by cells and (b) limits 2-AG pharmacological actions. We have studied simultaneously the facilitated transport of [14C]AEA and [3H]2-AG into rat C6 glioma cells and found uptake mechanisms with different efficacies but similar affinities for the two compounds (Km 11.0 +/- 2.0 and 15.3 +/- 3.1 microM, Bmax 1.70 +/- 0.30 and 0.24 +/- 0.04 nmol.min-1.mg protein-1, respectively). Despite these similar Km values, 2-AG inhibits [14C]AEA uptake by cells at concentrations (Ki = 30.1 +/- 3.9 microM) significantly higher than those required to either 2-AG or AEA to inhibit [3H]2-AG uptake (Ki = 18.9 +/- 1.8 and 20.5 +/- 3.2 microM, respectively). Furthermore: (a) if C6 cells are incubated simultaneously with identical concentrations of [14C]AEA and [3H]2-AG, only the uptake of the latter compound is significantly decreased as compared to that observed with [3H]2-AG alone; (b) the uptake of [14C]AEA and [3H]2-AG by cells is inhibited with the same potency by AM404 (Ki = 7.5 +/- 0.7 and 10.2 +/- 1.7 microM, respectively) and linvanil (Ki = 9.5 +/- 0.7 and 6.4 +/- 1.2 microM, respectively), two inhibitors of the AEA membrane transporter; (c) nitric oxide (NO) donors enhance the uptake of both [14C]AEA and [3H]2-AG, thus suggesting that 2-AG action can be regulated through NO release; (d) AEA and 2-AG induce a weak release of NO that can be blocked by a CB1 cannabinoid receptor antagonist, and significantly enhanced in the presence of AM404 and linvanil, thus suggesting that transport into C6 cells limits the action of both endocannabinoids.
The activity of anandamide at vanilloid VR1 receptors requires facilitated transport across the cell membrane and is limited by intracellular metabolism.[Pubmed:11278420]
J Biol Chem. 2001 Apr 20;276(16):12856-63.
The endogenous ligand of CB(1) cannabinoid receptors, anandamide, is also a full agonist at vanilloid VR1 receptors for capsaicin and resiniferatoxin, thereby causing an increase in cytosolic Ca(2+) concentration in human VR1-overexpressing (hVR1-HEK) cells. Two selective inhibitors of anandamide facilitated transport into cells, VDM11 and VDM13, and two inhibitors of anandamide enzymatic hydrolysis, phenylmethylsulfonyl fluoride and methylarachidonoyl fluorophosphonate, inhibited and enhanced, respectively, the VR1-mediated effect of anandamide, but not of resiniferatoxin or capsaicin. The nitric oxide donor, sodium nitroprusside, known to stimulate anandamide transport, enhanced anandamide effect on the cytosolic Ca(2+) concentration. Accordingly, hVR1-HEK cells contain an anandamide membrane transporter inhibited by VDM11 and VDM13 and activated by sodium nitroprusside, and an anandamide hydrolase activity sensitive to phenylmethylsulfonyl fluoride and methylarachidonoyl fluorophosphonate, and a fatty acid amide hydrolase transcript. These findings suggest the following. (i) Anandamide activates VR1 receptors by acting at an intracellular site. (ii) Degradation by fatty acid amide hydrolase limits anandamide activity on VR1; and (iii) the anandamide membrane transporter inhibitors can be used to distinguish between CB(1) or VR1 receptor-mediated actions of anandamide. By contrast, the CB(1) receptor antagonist SR141716A inhibited also the VR1-mediated effect of anandamide and capsaicin on cytosolic Ca(2+) concentration, although at concentrations higher than those required for CB(1) antagonism.
Overlap between the ligand recognition properties of the anandamide transporter and the VR1 vanilloid receptor: inhibitors of anandamide uptake with negligible capsaicin-like activity.[Pubmed:11033355]
FEBS Lett. 2000 Oct 13;483(1):52-6.
Some synthetic agonists of the VR1 vanilloid (capsaicin) receptor also inhibit the facilitated transport into cells of the endogenous cannabinoid anandamide (arachidonoylethanolamide, AEA). Here we tested several AEA derivatives containing various derivatized phenyl groups or different alkyl chains as either inhibitors of the AEA membrane transporter (AMT) in intact cells or functional agonists of the VR1 vanilloid receptor in HEK cells transfected with the human VR1. We found that four known AMT inhibitors, AM404, arvanil, olvanil and linvanil, activate VR1 receptors at concentrations 400-10000-fold lower than those necessary to inhibit the AMT. However, we also found three novel AEA derivatives, named VDM11, VDM12 and VDM13, which inhibit the AMT as potently as AM404 but exhibit little or no agonist activity at hVR1. These compounds are weak inhibitors of AEA enzymatic hydrolysis and poor CB(1)/CB(2) receptor ligands. We show for the first time that, despite the overlap between the chemical moieties of AMT inhibitors and VR1 agonists, selective inhibitors of AEA uptake that do not activate VR1 (e.g. VDM11) can be developed.


