BPTESGLS inhibitor CAS# 314045-39-1 |
- Imatinib Mesylate (STI571)
Catalog No.:BCC1115
CAS No.:220127-57-1
- Dasatinib (BMS-354825)
Catalog No.:BCC1281
CAS No.:302962-49-8
- Saracatinib (AZD0530)
Catalog No.:BCC1166
CAS No.:379231-04-6
- DPH
Catalog No.:BCC1538
CAS No.:484049-04-9
- Nilotinib monohydrochloride monohydrate
Catalog No.:BCC1801
CAS No.:923288-90-8
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 314045-39-1 | SDF | Download SDF |
| PubChem ID | 3372016 | Appearance | Powder |
| Formula | C24H24N6O2S3 | M.Wt | 524.68 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 50 mg/mL (95.30 mM; Need ultrasonic) H2O : < 0.1 mg/mL (insoluble) | ||
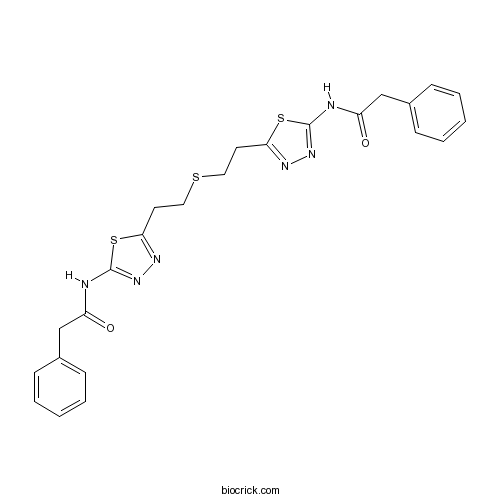
| Chemical Name | 2-phenyl-N-[5-[2-[2-[5-[(2-phenylacetyl)amino]-1,3,4-thiadiazol-2-yl]ethylsulfanyl]ethyl]-1,3,4-thiadiazol-2-yl]acetamide | ||
| SMILES | C1=CC=C(C=C1)CC(=O)NC2=NN=C(S2)CCSCCC3=NN=C(S3)NC(=O)CC4=CC=CC=C4 | ||
| Standard InChIKey | MDJIPXYRSZHCFS-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C24H24N6O2S3/c31-19(15-17-7-3-1-4-8-17)25-23-29-27-21(34-23)11-13-33-14-12-22-28-30-24(35-22)26-20(32)16-18-9-5-2-6-10-18/h1-10H,11-16H2,(H,25,29,31)(H,26,30,32) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Allosteric glutaminase (GLS1) inhibitor (IC50 = 3.3 μM). Selective for GLS1 over GLS2 and γ-glutamyl transpeptidase. Induces cell death of P493 human lymphoma B cells in vitro and reduces tumor volume of P493 cell xenografts in mice. |

BPTES Dilution Calculator

BPTES Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.9059 mL | 9.5296 mL | 19.0592 mL | 38.1185 mL | 47.6481 mL |
| 5 mM | 0.3812 mL | 1.9059 mL | 3.8118 mL | 7.6237 mL | 9.5296 mL |
| 10 mM | 0.1906 mL | 0.953 mL | 1.9059 mL | 3.8118 mL | 4.7648 mL |
| 50 mM | 0.0381 mL | 0.1906 mL | 0.3812 mL | 0.7624 mL | 0.953 mL |
| 100 mM | 0.0191 mL | 0.0953 mL | 0.1906 mL | 0.3812 mL | 0.4765 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
BPTES is a potent and selective kidney-type glutaminase (GLS) inhibitor [1], with a Ki value of approx. 3 μM [2].
Glutaminase hydrolyzes glutamine into ammonia and glutamate. In mammalian tissues, two glutaminase isoforms derived from structurally related but distinct genes, are expressed. GLS is widely distributed in extra-hepatic tissues. Liver-type glutaminase (GLS2) is primarily found in adult liver. GLS is critical in glutaminolysis for many proliferating cells, especially malignant cells with rapid growth [1].
Cell lines with mutations in isocitrate dehydrogenase 1 and 2 (IDH1/2) were used. In all IDH1-mutant AML cells, compared with DMSO, exposure to 20 µmol/L BPTES reduced the cell growth by approximately 50% on day 4. 20 µmol/L BPTES was not significantly different from 40 µmol/L BPTES in the reduction effect. Treatment without drug was not significantly different from treatment with DMSO in the growth of cells. BPTES did not significantly affect the cell growth of wild type AML cells [3]. In tumor cells, BPTES inhibited the conversion of glutamine into glutamate [4].
Glutamate is a substrate of GPT in the transamination of pyruvate to alanine. Compared with controls, BPTES treatment reduced the pyruvate-to-alanine conversion in animals. In replicated experiments, BPTES significantly reduce the alanine-to-pyruvate (Ala/Pyr) flux ratio [4].
References:
[1]. Shukla K, Ferraris DV, Thomas AG, et al. Design, synthesis, and pharmacological evaluation of bis-2-(5-phenylacetamido-1, 2, 4-thiadiazol-2-yl) ethyl sulfide 3 (BPTES) analogs as glutaminase inhibitors. Journal of medicinal chemistry, 2012, 55(23): 10551-10563.
[2]. Robinson MM, Mcbryant SJ, Tsukamoto T, et al. Novel mechanism of inhibition of rat kidney-type glutaminase by bis-2-(5-phenylacetamido-1, 2, 4-thiadiazol-2-yl) ethyl sulfide (BPTES). Biochemical Journal, 2007, 406(3): 407-414.
[3]. Emadi A, Jun SA, Tsukamoto T, et al. Inhibition of glutaminase selectively suppresses the growth of primary acute myeloid leukemia cells with IDH mutations. Experimental hematology, 2014, 42(4): 247-251.
[4]. Dutta P, Le A, Vander Jagt DL, et al. Evaluation of LDH-A and glutaminase inhibition in vivo by hyperpolarized 13C-pyruvate magnetic resonance spectroscopy of tumors. Cancer research, 2013, 73(14): 4190-4195.
- (R)-(-)-Apomorphine hydrochloride
Catalog No.:BCC7250
CAS No.:314-19-2
- Evans Blue tetrasodium salt
Catalog No.:BCC6815
CAS No.:314-13-6
- VDM 11
Catalog No.:BCC7044
CAS No.:313998-81-1
- [cPP1-7,NPY19-23,Ala31,Aib32,Gln34] - hPancreatic Polypeptide
Catalog No.:BCC5750
CAS No.:313988-89-5
- PU 02
Catalog No.:BCC6265
CAS No.:313984-77-9
- o-3M3FBS
Catalog No.:BCC7210
CAS No.:313981-55-4
- FLI-06
Catalog No.:BCC5110
CAS No.:313967-18-9
- [Des-octanoyl]-Ghrelin (human)
Catalog No.:BCC7304
CAS No.:313951-59-6
- 13-Oxo-9,11-octadecadienoic acid
Catalog No.:BCC8437
CAS No.:31385-09-8
- PD 118057
Catalog No.:BCC7499
CAS No.:313674-97-4
- Bombesin
Catalog No.:BCC5708
CAS No.:31362-50-2
- INH1
Catalog No.:BCC6040
CAS No.:313553-47-8
- IU1
Catalog No.:BCC2086
CAS No.:314245-33-5
- Isotachioside
Catalog No.:BCN5230
CAS No.:31427-08-4
- Nocodazole
Catalog No.:BCC3826
CAS No.:31430-18-9
- 4-Amino-3-nitrobenzophenone
Catalog No.:BCC8682
CAS No.:31431-19-3
- Mebendazole
Catalog No.:BCC9016
CAS No.:31431-39-7
- 6-Methoxysalicylic Acid
Catalog No.:BCC8288
CAS No.:3147-64-6
- Sunifiram
Catalog No.:BCC4167
CAS No.:314728-85-3
- Crotaline
Catalog No.:BCN4983
CAS No.:315-22-0
- Allopurinol
Catalog No.:BCC3720
CAS No.:315-30-0
- Testosterone enanthate
Catalog No.:BCC9169
CAS No.:315-37-7
- Acetylheliosupine
Catalog No.:BCN1981
CAS No.:31514-30-4
- PAC-1
Catalog No.:BCC3600
CAS No.:315183-21-2
Glutamine deprivation plus BPTES alters etoposide- and cisplatin-induced apoptosis in triple negative breast cancer cells.[Pubmed:27419628]
Oncotarget. 2016 Aug 23;7(34):54691-54701.
Glutamine provides cancer cells with the energy required to synthesize macromolecules. Methods which block glutamine metabolism in treatment of breast cancer inhibit oncogenic transformation and tumor growth. We investigated whether inhibiting glutamine metabolism produces effects that are synergistic with those produced by drugs which damage DNA in triple-negative breast cancer cells. HCC1937 and BT-549 breast cancer cells were co-treated with either cisplatin or etoposide in combination with BPTES (a specific inhibitor of glutaminase 1) or exposure to a glutamine-free medium, and the cell proliferation and cell apoptosis were measured by flow cytometry, immunoblotting studies, and CCK-8 assays. The results showed that both glutamine deprivation and BPTES pretreatments increased the toxic effects of cisplatin and etoposide on HCC1937 cells, as demonstrated by their reduced proliferation, increased expression of apoptosis-related proteins (cleaved-PARP, cleaved-caspase 9, and cleaved-caspase 3) and decreased Bcl-2/BAX ratio. However, in BT-549 cells, glutamine deprivation and BPTES treatment increased etoposide-induced apoptosis only when used with higher concentrations of etoposide, and the effect on cisplatin-induced apoptosis was minimal. These results suggest that the anti-cancer effects produced by a combined approach of inhibiting glutamine metabolism and administering common chemotherapeutic agents correlate with the tumor cell type and specific drugs being administered.
Effect of lysine to alanine mutations on the phosphate activation and BPTES inhibition of glutaminase.[Pubmed:25510640]
Neurochem Int. 2015 Sep;88:10-4.
The GLS1 gene encodes a mitochondrial glutaminase that is highly expressed in brain, kidney, small intestine and many transformed cells. Recent studies have identified multiple lysine residues in glutaminase that are sites of N-acetylation. Interestingly, these sites are located within either a loop segment that regulates access of glutamine to the active site or the dimer:dimer interface that participates in the phosphate-dependent oligomerization and activation of the enzyme. These two segments also contain the binding sites for bis-2[5-phenylacetamido-1,2,4-thiadiazol-2-yl]ethylsulfide (BPTES), a highly specific and potent uncompetitive inhibitor of this glutaminase. BPTES is also the lead compound for development of novel cancer chemotherapeutic agents. To provide a preliminary assessment of the potential effects of N-acetylation, the corresponding lysine to alanine mutations were constructed in the hGACDelta1 plasmid. The wild type and mutated proteins were purified by Ni(+)-affinity chromatography and their phosphate activation and BPTES inhibition profiles were analyzed. Two of the alanine substitutions in the loop segment (K311A and K328A) and the one in the dimer:dimer interface (K396A) form enzymes that require greater concentrations of phosphate to produce half-maximal activation and exhibit greater sensitivity to BPTES inhibition. By contrast, the K320A mutation results in a glutaminase that exhibits near maximal activity in the absence of phosphate and is not inhibited by BPTES. Thus, lysine N-acetylation may contribute to the acute regulation of glutaminase activity in various tissues and alter the efficacy of BPTES-type inhibitors.
Combination therapy with BPTES nanoparticles and metformin targets the metabolic heterogeneity of pancreatic cancer.[Pubmed:27559084]
Proc Natl Acad Sci U S A. 2016 Sep 6;113(36):E5328-36.
Targeting glutamine metabolism via pharmacological inhibition of glutaminase has been translated into clinical trials as a novel cancer therapy, but available drugs lack optimal safety and efficacy. In this study, we used a proprietary emulsification process to encapsulate bis-2-(5-phenylacetamido-1,2,4-thiadiazol-2-yl)ethyl sulfide (BPTES), a selective but relatively insoluble glutaminase inhibitor, in nanoparticles. BPTES nanoparticles demonstrated improved pharmacokinetics and efficacy compared with unencapsulated BPTES. In addition, BPTES nanoparticles had no effect on the plasma levels of liver enzymes in contrast to CB-839, a glutaminase inhibitor that is currently in clinical trials. In a mouse model using orthotopic transplantation of patient-derived pancreatic tumor tissue, BPTES nanoparticle monotherapy led to modest antitumor effects. Using the HypoxCR reporter in vivo, we found that glutaminase inhibition reduced tumor growth by specifically targeting proliferating cancer cells but did not affect hypoxic, noncycling cells. Metabolomics analyses revealed that surviving tumor cells following glutaminase inhibition were reliant on glycolysis and glycogen synthesis. Based on these findings, metformin was selected for combination therapy with BPTES nanoparticles, which resulted in significantly greater pancreatic tumor reduction than either treatment alone. Thus, targeting of multiple metabolic pathways, including effective inhibition of glutaminase by nanoparticle drug delivery, holds promise as a novel therapy for pancreatic cancer.
Design, synthesis, and pharmacological evaluation of bis-2-(5-phenylacetamido-1,2,4-thiadiazol-2-yl)ethyl sulfide 3 (BPTES) analogs as glutaminase inhibitors.[Pubmed:23151085]
J Med Chem. 2012 Dec 13;55(23):10551-63.
Bis-2-(5-phenylacetamido-1,2,4-thiadiazol-2-yl)ethyl sulfide (BPTES) is a potent and selective allosteric inhibitor of kidney-type glutaminase (GLS) that has served as a molecular probe to determine the therapeutic potential of GLS inhibition. In an attempt to identify more potent GLS inhibitors with improved drug-like molecular properties, a series of BPTES analogs were synthesized and evaluated. Our structure-activity relationship (SAR) studies revealed that some truncated analogs retained the potency of BPTES, presenting an opportunity to improve its aqueous solubility. One of the analogs, N-(5-{2-[2-(5-amino-[1,3,4]thiadiazol-2-yl)-ethylsulfanyl]-ethyl}-[1,3,4]thiadiaz ol-2-yl)-2-phenyl-acetamide 6, exhibited similar potency and better solubility relative to BPTES and attenuated the growth of P493 human lymphoma B cells in vitro as well as in a mouse xenograft model.
Glucose-independent glutamine metabolism via TCA cycling for proliferation and survival in B cells.[Pubmed:22225880]
Cell Metab. 2012 Jan 4;15(1):110-21.
Because MYC plays a causal role in many human cancers, including those with hypoxic and nutrient-poor tumor microenvironments, we have determined the metabolic responses of a MYC-inducible human Burkitt lymphoma model P493 cell line to aerobic and hypoxic conditions, and to glucose deprivation, using stable isotope-resolved metabolomics. Using [U-(13)C]-glucose as the tracer, both glucose consumption and lactate production were increased by MYC expression and hypoxia. Using [U-(13)C,(15)N]-glutamine as the tracer, glutamine import and metabolism through the TCA cycle persisted under hypoxia, and glutamine contributed significantly to citrate carbons. Under glucose deprivation, glutamine-derived fumarate, malate, and citrate were significantly increased. Their (13)C-labeling patterns demonstrate an alternative energy-generating glutaminolysis pathway involving a glucose-independent TCA cycle. The essential role of glutamine metabolism in cell survival and proliferation under hypoxia and glucose deficiency makes them susceptible to the glutaminase inhibitor BPTES and hence could be targeted for cancer therapy.
Full-length human glutaminase in complex with an allosteric inhibitor.[Pubmed:22049910]
Biochemistry. 2011 Dec 20;50(50):10764-70.
Glutaminase (GLS1/2) catalyzes the conversion of L-glutamine to L-glutamate and ammonia. The level of a splice variant of GLS1 (GAC) is elevated in certain cancers, and GAC is specifically inhibited by bis-2-(5-phenylacetimido-1,2,4,thiadiazol-2-yl)ethyl sulfide (BPTES). We report here the first full-length crystal structure of GAC in the presence and absence of BPTES molecules. Two BPTES molecules bind at an interface region of the GAC tetramer in a manner that appears to lock the GAC tetramer into a nonproductive conformation. The importance of these loops with regard to overall enzymatic activity of the tetramer was revealed by a series of GAC point mutants designed to create a BPTES resistant GAC.


