Ibandronate sodiumCAS# 138926-19-9 |
- I-BET-762
Catalog No.:BCC4474
CAS No.:1260907-17-2
- MS436
Catalog No.:BCC4037
CAS No.:1395084-25-9
- PFI-1 (PF-6405761)
Catalog No.:BCC2225
CAS No.:1403764-72-6
- SGC-CBP30
Catalog No.:BCC2419
CAS No.:1613695-14-9
- Bromosporine
Catalog No.:BCC2226
CAS No.:1619994-69-2
- OTX-015
Catalog No.:BCC1829
CAS No.:202590-98-5
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 138926-19-9 | SDF | Download SDF |
| PubChem ID | 23663991 | Appearance | Powder |
| Formula | C9H24NNaO8P2 | M.Wt | 359.2 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 72 mg/mL (199.87 mM) in Water | ||
| Chemical Name | sodium;hydroxy-[1-hydroxy-3-[methyl(pentyl)amino]-1-phosphonopropyl]phosphinate;hydrate | ||
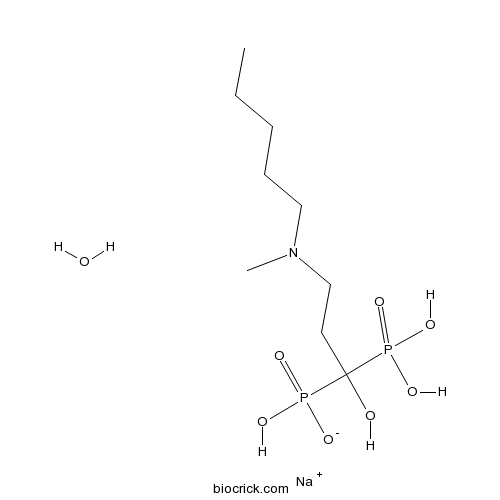
| SMILES | CCCCCN(C)CCC(O)(P(=O)(O)O)P(=O)(O)[O-].O.[Na+] | ||
| Standard InChIKey | VBDRTGFACFYFCT-UHFFFAOYSA-M | ||
| Standard InChI | InChI=1S/C9H23NO7P2.Na.H2O/c1-3-4-5-7-10(2)8-6-9(11,18(12,13)14)19(15,16)17;;/h11H,3-8H2,1-2H3,(H2,12,13,14)(H2,15,16,17);;1H2/q;+1;/p-1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Ibandronate is a highly potent nitrogen-containing bisphosphonate used for the treatment of osteoporosis.
Target: Others
Ibandronate (1.25-2 μM) significantly reduces endothelial cell growth, while ibandronate (2 μM) also significantly reduces capillary-like tube formation and increases apoptosis of endothelial cells. Ibandronate (< 100 μM) dose-dependently increases VEGF expression in endothelial cells [1]. Ibandronate (< 100 μM) inhibits growth of both prostate cancer cell lines (LNCaP and PC-3) in a dose dependent manner [2].
Ibandronate administered either daily (2.5 mg) or intermittently (20 mg every other day for 12 doses every 3 months) significantly reduces the risk of new morphometric vertebral fractures by 62% and 50% (p = 0.0006), respectively, in osteoporotic women after 3 years' treatment. Ibandronate administered either daily (2.5 mg) or intermittently (20 mg every other day for 12 doses every 3 months) significantly and progressively increases BMD of lumbar spine by 6.5% and 5.7%, respectively, in osteoporotic women after 3 years' treatment [3]. Ibandronate (< 125 mg/kg s.c.) results in a dose dependent increase in bone mineral density (BMD), trabecular bone volume and trabecular number, load to failure (Fmax), and yield load in long bones and vertebrae in ovariectomized rats, and increased trabecular separation in ovariectomized rats is fully prevented by all doses [4]. References: | |||||

Ibandronate sodium Dilution Calculator

Ibandronate sodium Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.784 mL | 13.9198 mL | 27.8396 mL | 55.6793 mL | 69.5991 mL |
| 5 mM | 0.5568 mL | 2.784 mL | 5.5679 mL | 11.1359 mL | 13.9198 mL |
| 10 mM | 0.2784 mL | 1.392 mL | 2.784 mL | 5.5679 mL | 6.9599 mL |
| 50 mM | 0.0557 mL | 0.2784 mL | 0.5568 mL | 1.1136 mL | 1.392 mL |
| 100 mM | 0.0278 mL | 0.1392 mL | 0.2784 mL | 0.5568 mL | 0.696 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Ibandronate is a highly potent nitrogen-containing bisphosphonate used for the treatment of osteoporosis.
- Entadamide-A-β-D-glucopyranoside
Catalog No.:BCN8452
CAS No.:138916-58-2
- Brinzolamide
Catalog No.:BCC2313
CAS No.:138890-62-7
- 1-Acetylpiperazine
Catalog No.:BCC8448
CAS No.:13889-98-0
- 5'-Methoxyhexahydrocurcumin
Catalog No.:BCN7049
CAS No.:138870-96-9
- Pasakbumin B
Catalog No.:BCN2991
CAS No.:138809-10-6
- 8'-Oxo-6-hydroxydihydrophaseic acid
Catalog No.:BCN7046
CAS No.:1388075-44-2
- Fmoc-D-Thr(tBu)-OH
Catalog No.:BCC3555
CAS No.:138797-71-4
- Isomangiferolic acid
Catalog No.:BCN4768
CAS No.:13878-92-7
- Fmoc-N-Me-Ile-OH
Catalog No.:BCC3214
CAS No.:138775-22-1
- Fmoc-D-2-Nal-OH
Catalog No.:BCC3290
CAS No.:138774-94-4
- Fmoc-D-1-Nal-OH
Catalog No.:BCC3284
CAS No.:138774-49-3
- Eugenol rutinoside
Catalog No.:BCN6201
CAS No.:138772-01-7
- Rutaretin
Catalog No.:BCN4710
CAS No.:13895-92-6
- Isocoronarin D
Catalog No.:BCN6202
CAS No.:138965-88-5
- Coronarin D ethyl ether
Catalog No.:BCN6203
CAS No.:138965-89-6
- Capsazepine
Catalog No.:BCC1451
CAS No.:138977-28-3
- Ziprasidone hydrochloride monohydrate
Catalog No.:BCC2072
CAS No.:138982-67-9
- 3,4-Dihydroxybenzaldehyde
Catalog No.:BCN6214
CAS No.:139-85-5
- Carmine
Catalog No.:BCN2223
CAS No.:1390-65-4
- Catalpin
Catalog No.:BCN6205
CAS No.:1390-72-3
- Oplodiol
Catalog No.:BCN6204
CAS No.:13902-62-0
- JMV 449
Catalog No.:BCC5863
CAS No.:139026-66-7
- 2-PMDQ
Catalog No.:BCC6726
CAS No.:139047-55-5
- Anemarsaponin B
Catalog No.:BCN6289
CAS No.:139051-27-7
Phase I clinical study to select a novel oral formulation for ibandronate containing the excipient sodium N-[8-(2-hydroxybenzoyl) amino] caprylate (SNAC).[Pubmed:22530305]
Pharmazie. 2012 Mar;67(3):233-41.
The aim of this study was to select a novel oral formulation for ibandronate (IBN, CAS number: 13892619). In four cohorts of 28, 21, 19 and 29 healthy volunteers, the impact of the carrier molecule sodium N-[8-(2-hydroxybenzoyl) amino] caprylate (SNAC, CAS number: 203787-91-1) on the bioavailability of IBN was investigated. Within each cohort different oral formulations with one dose of ibandronate (30 mg) and three different ratios of IBN:SNAC (1:5, 1:10 and 1:20) were compared to the approved oral IBN tablet formulations (150 and 50 mg IBN) in a 4-way cross-over design and a one week washout between the administrations. The highest mean IBN exposure was achieved with a capsule formulation containing drug-coated beadlets and an IBN:SNAC ratio of 1:5. AUC(last) and C(max) of IBN were approximately 1.3- and 2.2-fold higher compared to the reference treatment (150 mg IBN without SNAC). Increasing the post-dose fasting duration from 15 to 30 min resulted in a more than 2-fold increase in AUC(last), while superimposable IBN serum concentration-time profiles were achieved after a 30 and 60 min fast. The tolerability of the IBN/SNAC treatments in all cohorts was similar to that in the IBN reference groups and most adverse events (AEs) were of mild to moderate intensity.
Impurity profiling of ibandronate sodium by HPLC-CAD.[Pubmed:26092222]
J Pharm Biomed Anal. 2015 Oct 10;114:254-64.
The modern bisphosphonate drug Ibandronate sodium, a challenging candidate for impurity profiling, was analyzed using high performance liquid chromatography (HPLC) combined with corona charged aerosol detection (CAD). Separation was achieved on a mixed mode column combining hydrophobic C18 and strong anion exchange retention mechanisms using a mass spectrometer compatible volatile mobile phase consisting of trifluoroacetic acid and acetonitrile while gradient elution was applied. The method was validated following the ICH guideline Q2(R1) and found suitable for the assessment of ibandronate's related substances. The observed CAD-response for all identified impurities was linear (R(2)>0.995) over a small concentration range (0.05-0.25) and a quantification limit of at least 0.03% was found. Four batches of two different manufacturers were tested by means of the method. None of the batches contained a single impurity above 0.05%. The major impurities of all batches were the synthesis by-products N-desmethyl- and N-despentyl ibandronate as well as N,N-dimethyl pamidronate.
Effects of ibandronate sodium, a nitrogen-containing bisphosphonate, on intermediate-conductance calcium-activated potassium channels in osteoclast precursor cells (RAW 264.7).[Pubmed:25362532]
J Membr Biol. 2015 Feb;248(1):103-15.
Ibanonate sodium (Iban), a nitrogen-containing bisphosphonate, is recognized to reduce skeletal complications through an inhibition of osteoclast-mediated bone resorption. However, how this drug interacts with ion channels in osteoclasts and creates anti-osteoclastic activity remains largely unclear. In this study, we investigated the possible effects of Iban and other related compounds on ionic currents in the osteoclast precursor RAW 264.7 cells. Iban suppressed the amplitude of whole-cell K(+) currents (I K) in a concentration-dependent manner with an IC50 value of 28.9 muM. The I K amplitude was sensitive to block by TRAM-34 and Iban-mediated inhibition of I K was reversed by further addition of DCEBIO, an activator of intermediate-conductance Ca(2+)-activated K(+) (IKCa) channels. Intracellular dialysis with Iban diminished I K amplitude and further addition of ionomycin reversed its inhibition. In 17beta-estradiol-treated cells, Iban-mediated inhibition of I K remained effective. In cell-attached current recordings, Iban applied to bath did not modify single-channel conductance of IKCa channels; however, it did reduce channel activity. Iban-induced inhibition of IKCa channels was voltage-dependent. As IKCa-channel activity was suppressed by KN-93, subsequent addition of Iban did not further decrease the channel open probability. Iban could not exert any effect on inwardly rectifying K(+) current in RAW 264.7 cells. Under current-clamp recordings, Iban depolarized the membrane of RAW 264.7 cells and DCEBIO reversed Iban-induced depolarization. Iban also suppressed lipopolysaccharide-stimulated migration of RAW 264.7 cells in a concentration-dependent manner. Therefore, the inhibition by Iban of IKCa channels would be an important mechanism underlying its actions on the functional activity of osteoclasts occurring in vivo.
In vitro osteoclast-suppressing effect of sodium ibandronate.[Pubmed:23422201]
Chin Med J (Engl). 2013 Feb;126(4):751-5.
BACKGROUND: Bisphosphonates (BPs) have been reported to reduce local recurrence in giant cell tumor (GCT) of bone because of their osteoclast-suppressing effect; however, the optimal mode of delivery and the dose and duration of treatment of BPs remain to be established. To address these issues, it is first necessary to clarify the manner of action of BPs on osteoclasts. We herein evaluated the osteoclast-suppressing effect of sodium ibandronate in vitro. METHODS: Mouse osteoclasts (OCLs) were generated in vitro using mouse bone marrow mononuclear cells. First, various concentrations of sodium ibandronate and equal amounts of phosphate-buffered saline were added to cell culture media. The number of multinucleated cells (over three nuclei) was recorded in each group, OCL formation was compared, and the most effective concentration of sodium ibandronate was determined. Then, high concentrations of sodium ibandronate were added to the experimental cell culture media; no ibandronate was given in the control group. Comparisons were made between the two groups in terms of OCL adhesion, migration, and bone resorption. RESULTS: OCL formation was suppressed by sodium ibandronate in vitro; the most pronounced effect was observed at the concentration of 10(-5) mol/L. OCL migration and bone resorption were significantly suppressed at this concentration, though there was no effect on OCL adhesion. CONCLUSIONS: Sodium ibandronate was effective in suppressing OCLs and decreasing resorption in GCT. The strong anti-OCL effectiveness at a high concentration in vitro indicates a topical mode of application.


