N20C hydrochlorideCAS# 1177583-87-7 |
- SU14813
Catalog No.:BCC1971
CAS No.:627908-92-3
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 1177583-87-7 | SDF | Download SDF |
| PubChem ID | 16759164 | Appearance | Powder |
| Formula | C17H21ClN2O | M.Wt | 304.82 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in water | ||
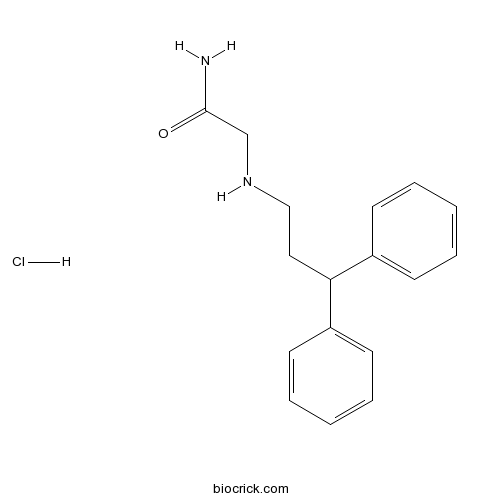
| Chemical Name | 2-(3,3-diphenylpropylamino)acetamide;hydrochloride | ||
| SMILES | C1=CC=C(C=C1)C(CCNCC(=O)N)C2=CC=CC=C2.Cl | ||
| Standard InChIKey | JCJDLPUASJEVRN-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C17H20N2O.ClH/c18-17(20)13-19-12-11-16(14-7-3-1-4-8-14)15-9-5-2-6-10-15;/h1-10,16,19H,11-13H2,(H2,18,20);1H | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Selective, non-competitive NMDA receptor antagonist (IC50 = 5 μM); binds to the receptor-associated ion channel and prevents glutamate-induced Ca2+ influx. Displays neuroprotective activity in vivo. Brain-penetrant. |

N20C hydrochloride Dilution Calculator

N20C hydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.2806 mL | 16.4031 mL | 32.8062 mL | 65.6125 mL | 82.0156 mL |
| 5 mM | 0.6561 mL | 3.2806 mL | 6.5612 mL | 13.1225 mL | 16.4031 mL |
| 10 mM | 0.3281 mL | 1.6403 mL | 3.2806 mL | 6.5612 mL | 8.2016 mL |
| 50 mM | 0.0656 mL | 0.3281 mL | 0.6561 mL | 1.3122 mL | 1.6403 mL |
| 100 mM | 0.0328 mL | 0.164 mL | 0.3281 mL | 0.6561 mL | 0.8202 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Forsythoside I
Catalog No.:BCN6430
CAS No.:1177581-50-8
- Felbamate hydrate
Catalog No.:BCC4160
CAS No.:1177501-39-1
- (R)-(+)-Blebbistatin
Catalog No.:BCC7195
CAS No.:1177356-70-5
- CKI 7 dihydrochloride
Catalog No.:BCC5614
CAS No.:1177141-67-1
- Doramectin
Catalog No.:BCC1536
CAS No.:117704-25-3
- Dexamethasone acetate
Catalog No.:BCC4775
CAS No.:1177-87-3
- Laxogenin
Catalog No.:BCN8434
CAS No.:1177-71-5
- DL-Syringaresinol
Catalog No.:BCN6053
CAS No.:1177-14-6
- LY 255283
Catalog No.:BCC7290
CAS No.:117690-79-6
- Agomelatine hydrochloride
Catalog No.:BCC4210
CAS No.:1176316-99-6
- 3-Cyano-7-ethoxycoumarin
Catalog No.:BCC7979
CAS No.:117620-77-6
- 16-Oxocleroda-3,13E-dien-15-oic acid
Catalog No.:BCN7286
CAS No.:117620-72-1
- SMANT hydrochloride
Catalog No.:BCC6254
CAS No.:1177600-74-6
- CGH 2466 dihydrochloride
Catalog No.:BCC7338
CAS No.:1177618-54-0
- Azithromycin Dihydrate
Catalog No.:BCC4631
CAS No.:117772-70-0
- Decumbenine C
Catalog No.:BCC8314
CAS No.:117772-89-1
- Desmethyl-YM 298198
Catalog No.:BCC7365
CAS No.:1177767-57-5
- AP-III-a4
Catalog No.:BCC5292
CAS No.:1177827-73-4
- NSC 23766
Catalog No.:BCC1149
CAS No.:1177865-17-6
- 3,3',4',5,6,7,8-heptamethoxyflavone
Catalog No.:BCN8203
CAS No.:1178-24-1
- Enterostatin
Catalog No.:BCC6050
CAS No.:117830-79-2
- Ac-Asp(OtBu)-OH
Catalog No.:BCC2880
CAS No.:117833-18-8
- Loreclezole hydrochloride
Catalog No.:BCC7009
CAS No.:117857-45-1
- L-CCG-l
Catalog No.:BCC6609
CAS No.:117857-93-9
Unusual 4-arsonoanilinium cationic species in the hydrochloride salt of (4-aminophenyl)arsonic acid and formed in the reaction of the acid with copper(II) sulfate, copper(II) chloride and cadmium chloride.[Pubmed:28378716]
Acta Crystallogr C Struct Chem. 2017 Apr 1;73(Pt 4):325-330.
Structures having the unusual protonated 4-arsonoanilinium species, namely in the hydrochloride salt, C6H9AsNO3(+).Cl(-), (I), and the complex salts formed from the reaction of (4-aminophenyl)arsonic acid (p-arsanilic acid) with copper(II) sulfate, i.e. hexaaquacopper(II) bis(4-arsonoanilinium) disulfate dihydrate, (C6H9AsNO3)2[Cu(H2O)6](SO4)2.2H2O, (II), with copper(II) chloride, i.e. poly[bis(4-arsonoanilinium) [tetra-mu-chlorido-cuprate(II)]], {(C6H9AsNO3)2[CuCl4]}n, (III), and with cadmium chloride, i.e. poly[bis(4-arsonoanilinium) [tetra-mu-chlorido-cadmate(II)]], {(C6H9AsNO3)2[CdCl4]}n, (IV), have been determined. In (II), the two 4-arsonoanilinium cations are accompanied by [Cu(H2O)6](2+) cations with sulfate anions. In the isotypic complex salts (III) and (IV), they act as counter-cations to the {[CuCl4](2-)}n or {[CdCl4](2-)}n anionic polymer sheets, respectively. In (II), the [Cu(H2O)6](2+) ion sits on a crystallographic centre of symmetry and displays a slightly distorted octahedral coordination geometry. The asymmetric unit for (II) contains, in addition to half the [Cu(H2O)6](2+) ion, one 4-arsonoanilinium cation, a sulfate dianion and a solvent water molecule. Extensive O-H...O and N-H...O hydrogen bonds link all the species, giving an overall three-dimensional structure. In (III), four of the chloride ligands are related by inversion [Cu-Cl = 2.2826 (8) and 2.2990 (9) A], with the other two sites of the tetragonally distorted octahedral CuCl6 unit occupied by symmetry-generated Cl-atom donors [Cu-Cl = 2.9833 (9) A], forming a two-dimensional coordination polymer network substructure lying parallel to (001). In the crystal, the polymer layers are linked across [001] by a number of bridging hydrogen bonds involving N-H...Cl interactions from head-to-head-linked As-O-H...O 4-arsonoanilinium cations. A three-dimensional network structure is formed. Cd(II) compound (IV) is isotypic with Cu(II) complex (III), but with the central CdCl6 complex repeat unit having a more regular M-Cl bond-length range [2.5232 (12)-2.6931 (10) A] compared to that in (III). This series of compounds represents the first reported crystal structures having the protonated 4-arsonoanilinium species.
Lens opacities in children using methylphenidate hydrochloride.[Pubmed:28376677]
Cutan Ocul Toxicol. 2017 Dec;36(4):362-365.
PURPOSE: To assess clinical findings of eye examination in children having attention deficit hyperactivity disorder (ADHD) administered with methylphenidate hydrochloride. METHODS: Fifty-seven consecutive patients diagnosed of ADHD and administered with oral methylphenidate hydrochloride treatment for at least one year were involved in this study (Group 1). Sixty healthy subjects (Group 2) having demographic features similar to group 1 were involved as a control group. All patients underwent detailed ophthalmological examination. RESULTS: One hundred and seventeen consecutive subjects with a mean age of 11.2 +/- 2.4 years (7-18 years) were enrolled. Fifty-seven consecutive patient (32 males, 25 females) under oral methylphenidate hydrochloride treatment (Group 1) and 60 healthy control subjects (30 males, 30 females) (Group 2)) were recruited for this prospective study. The mean methylphenidate hydrochloride dosage was 0.9 +/- 0.1 mg/kg/day and the mean duration of methylphenidate hydrochloride usage was for 2.73 +/- 0.73 years (1-7 years). High intraocular pressure was not observed in any of the patients in our study. We detected lens opacities in five eyes of five patients in group 1 (p = 0.019). The patient with the highest degree of cataract formation had been using MPH for 84 months and this patient's cataract score was P4. CONCLUSION: Long-term use of methylphenidate may cause lens opacities. In particular, patients who have been using methylphenidate for more than two years should go for regular eye examination.
Biophysical Study on the Interaction between Eperisone Hydrochloride and Human Serum Albumin Using Spectroscopic, Calorimetric, and Molecular Docking Analyses.[Pubmed:28380300]
Mol Pharm. 2017 May 1;14(5):1656-1665.
Eperisone hydrochloride (EH) is widely used as a muscle relaxant for patients with muscular contracture, low back pain, or spasticity. Human serum albumin (HSA) is a highly soluble negatively charged, endogenous and abundant plasma protein ascribed with the ligand binding and transport properties. The current study was undertaken to explore the interaction between EH and the serum transport protein, HSA. Study of the interaction between HSA and EH was carried by UV-vis, fluorescence quenching, circular dichroism (CD), Fourier transform infrared (FTIR) spectroscopy, Forster's resonance energy transfer, isothermal titration calorimetry and differential scanning calorimetry. Tryptophan fluorescence intensity of HSA was strongly quenched by EH. The binding constants (Kb) were obtained by fluorescence quenching, and results show that the HSA-EH interaction revealed a static mode of quenching with binding constant Kb approximately 10(4) reflecting high affinity of EH for HSA. The negative DeltaG degrees value for binding indicated that HSA-EH interaction was a spontaneous process. Thermodynamic analysis shows HSA-EH complex formation occurs primarily due to hydrophobic interactions, and hydrogen bonds were facilitated at the binding of EH. EH binding induces alpha-helix of HSA as obtained by far-UV CD and FTIR spectroscopy. In addition, the distance between EH (acceptor) and Trp residue of HSA (donor) was calculated 2.18 nm using Forster's resonance energy transfer theory. Furthermore, molecular docking results revealed EH binds with HSA, and binding site was positioned in Sudlow Site I of HSA (subdomain IIA). This work provides a useful experimental strategy for studying the interaction of myorelaxant with HSA, helping to understand the activity and mechanism of drug binding.
Fabrication yields of serially harvested calf-fed Holstein steers fed zilpaterol hydrochloride.[Pubmed:28380524]
J Anim Sci. 2017 Mar;95(3):1209-1218.
Holstein steers ( = 110) were fed zilpaterol hydrochloride (ZH) for 0 or 20 d before slaughter during a 280-d serial harvest study. Cattle were harvested every 28 d beginning at 254 d on feed (DOF) and concluding at 534 DOF. After slaughter, carcasses were chilled for 48 h and then fabricated into boneless closely trimmed or denuded subprimals, lean trim, trimmable fat, and bone. Inclusion of ZH increased cold side weight (CSW) by 10.3 kg ( < 0.01; 212.7 vs. 202.4 kg [SEM 1.96]) and saleable yield by 10.4 kg ( < 0.01; 131.9 vs. 121.5 kg [SEM 1.16]) in calf-fed Holstein steer carcasses. Additionally, saleable yield as a percentage of CSW increased (
A novel N-methyl-D-aspartate receptor open channel blocker with in vivo neuroprotectant activity.[Pubmed:12065713]
J Pharmacol Exp Ther. 2002 Jul;302(1):163-73.
Excitotoxicity has been implicated in the etiology of ischemic stroke, chronic neurodegenerative disorders, and very recently, in glioma growth. Thus, the development of novel neuroprotectant molecules that reduce excitotoxic brain damage is vigorously pursued. We have used an ionic current block-based cellular assay to screen a synthetic combinatorial library of trimers of N-alkylglycines on the N-methyl-D-aspartate (NMDA) receptor, a well known molecular target involved in excitotoxicity. We report the identification of a family of N-alkylglycines that selectively blocked the NMDA receptor. Notably, compound 3,3-diphenylpropyl-N-glycinamide (referred to as N20C) inhibited NMDA receptor channel activity with micromolar affinity, fast on-off blockade kinetics, and strong voltage dependence. Molecule N20C did not act as a competitive glutamate or glycine antagonist. In contrast, saturation of the blocker binding site with N20C prevented dizolcipine (MK-801) blockade of the NMDA receptor, implying that both drugs bind to the same receptor site. The N-alkylglycine efficiently prevented in vitro excitotoxic neurodegeneration of cerebellar and hippocampal neurons in culture. Attenuation of neuronal glutamate/NMDA-induced Ca(2+) overload and subsequent modulation of the glutamate-nitric oxide-cGMP pathway seems to underlie N20C neuroprotection. Noteworthy, this molecule exhibited significant in vivo neuroprotectant activity against an acute, severe, excitotoxic insult. Taken together, these findings indicate that N-alkylglycine N20C is a novel, low molecular weight, moderate-affinity NMDA receptor open channel blocker with in vitro and in vivo neuroprotective activity, which, in due turn, may become a tolerated drug for the treatment of neurodegenerative diseases and cancer.


