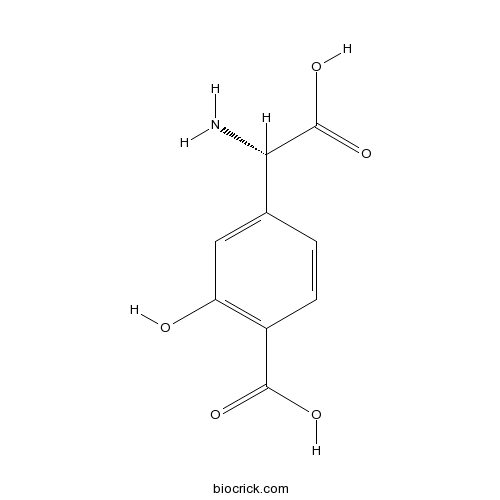
(S)-4-Carboxy-3-hydroxyphenylglycineGroup II agonist/group I antagonist CAS# 85148-82-9 |
- I-BET-762
Catalog No.:BCC4474
CAS No.:1260907-17-2
- Bromodomain Inhibitor, (+)-JQ1
Catalog No.:BCC1132
CAS No.:1268524-70-4
- I-BET151 (GSK1210151A)
Catalog No.:BCC4476
CAS No.:1300031-49-5
- GSK1324726A
Catalog No.:BCC4038
CAS No.:1300031-52-0
- PFI-1 (PF-6405761)
Catalog No.:BCC2225
CAS No.:1403764-72-6
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 85148-82-9 | SDF | Download SDF |
| PubChem ID | 5311455 | Appearance | Powder |
| Formula | C9H9NO5 | M.Wt | 211.17 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | (<em>S</em>)-4C3HPG | ||
| Solubility | Soluble to 100 mM in 1eq. NaOH and to 5 mM in water | ||
| Chemical Name | 4-[(S)-amino(carboxy)methyl]-2-hydroxybenzoic acid | ||
| SMILES | C1=CC(=C(C=C1C(C(=O)O)N)O)C(=O)O | ||
| Standard InChIKey | GXZSAQLJWLCLOX-ZETCQYMHSA-N | ||
| Standard InChI | InChI=1S/C9H9NO5/c10-7(9(14)15)4-1-2-5(8(12)13)6(11)3-4/h1-3,7,11H,10H2,(H,12,13)(H,14,15)/t7-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Competitive antagonist at group I mGlu1a/1a receptors, mixed effect at mGlu5a/5b receptors; agonist at group II metabotropic glutamate receptors. (RS)-4-Carboxy-3-hydroxyphenylglycine also available. |

(S)-4-Carboxy-3-hydroxyphenylglycine Dilution Calculator

(S)-4-Carboxy-3-hydroxyphenylglycine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.7355 mL | 23.6776 mL | 47.3552 mL | 94.7104 mL | 118.388 mL |
| 5 mM | 0.9471 mL | 4.7355 mL | 9.471 mL | 18.9421 mL | 23.6776 mL |
| 10 mM | 0.4736 mL | 2.3678 mL | 4.7355 mL | 9.471 mL | 11.8388 mL |
| 50 mM | 0.0947 mL | 0.4736 mL | 0.9471 mL | 1.8942 mL | 2.3678 mL |
| 100 mM | 0.0474 mL | 0.2368 mL | 0.4736 mL | 0.9471 mL | 1.1839 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Phospho-Glycogen Synthase Peptide-2 (substrate)
Catalog No.:BCC5747
CAS No.:851366-97-7
- PF9 tetrasodium salt
Catalog No.:BCC7854
CAS No.:851265-78-6
- 2-Methoxystypandrone
Catalog No.:BCN4400
CAS No.:85122-21-0
- THIP hydrochloride
Catalog No.:BCC6803
CAS No.:85118-33-8
- Amuvatinib (MP-470, HPK 56)
Catalog No.:BCC2258
CAS No.:850879-09-3
- Danoprevir (RG7227)
Catalog No.:BCC2106
CAS No.:850876-88-9
- Cinchonain IIa
Catalog No.:BCN7716
CAS No.:85081-23-8
- NBI-98782
Catalog No.:BCC4277
CAS No.:85081-18-1
- BX517(PDK1 inhibitor2)
Catalog No.:BCC6391
CAS No.:850717-64-5
- GSK269962A
Catalog No.:BCC5178
CAS No.:850664-21-0
- Alogliptin Benzoate
Catalog No.:BCC1341
CAS No.:850649-62-6
- Alogliptin (SYR-322)
Catalog No.:BCC2113
CAS No.:850649-61-5
- Curculigoside C
Catalog No.:BCN3696
CAS No.:851713-74-1
- OC000459
Catalog No.:BCC4507
CAS No.:851723-84-7, 950688-14-9 (sodium salt)
- PF 514273
Catalog No.:BCC7746
CAS No.:851728-60-4
- 3-(3-Chloropropyl)-1,3-dihydro-7,8-dimethoxy-2H-3-benzazepin-2-one
Catalog No.:BCC8587
CAS No.:85175-59-3
- ADX-47273
Catalog No.:BCC4598
CAS No.:851881-60-2
- RuBi-4AP
Catalog No.:BCC6044
CAS No.:851956-02-0
- TOK-001
Catalog No.:BCC3910
CAS No.:851983-85-2
- BAPTA
Catalog No.:BCC7483
CAS No.:85233-19-8
- 6-Methyl-7-O-methylaromadendrin
Catalog No.:BCN4010
CAS No.:852385-13-8
- Necrostatin 2 racemate
Catalog No.:BCC2077
CAS No.:852391-15-2
- Necrostatin 2
Catalog No.:BCC1793
CAS No.:852391-19-6
- Necrostatin 2 S enantiomer
Catalog No.:BCC2078
CAS No.:852391-20-9
Metabotropic receptors in excitotoxicity: (S)-4-carboxy-3-hydroxyphenylglycine ((S)-4C3HPG) protects against rat striatal quinolinic acid lesions.[Pubmed:8787843]
Neurosci Lett. 1995 Dec 29;202(1-2):109-12.
Striatal quinolinate lesions mimic many of the neuropathological characteristics of Huntington's disease. This excitotoxicity is mediated by combined activity of N-methyl-D-aspartate and metabotropic glutamate receptors (mGluRs). Using recently developed phenylglycine derivatives, (S)-4-Carboxy-3-hydroxyphenylglycine ((S)-4C3HPG) and (+)-alpha-methyl-4-carboxyphenylglycine ((+)-MCPG), we investigated the role of the different sub-classes of mGluRs in the in vivo excitotoxic process. (S)-4C3HPG (500 and 1000 nmol), co-injected with quinolinic acid, significantly reduced lesion volumes by 52 and 89%, respectively, whereas the same doses of (+)-MCPG had no effect on lesion size. The differential actions of these two drugs at Group 1 and Group 2 metabotropic receptors may explain their differential effects. These observations confirm the important role of mGluRs in excitotoxicity and identify them as promising targets for intervention.
(S)-4-carboxy-3-hydroxyphenylglycine, an antagonist of metabotropic glutamate receptor (mGluR) 1a and an agonist of mGluR2, protects against audiogenic seizures in DBA/2 mice.[Pubmed:8189254]
J Neurochem. 1994 Jun;62(6):2492-5.
The in vivo anticonvulsant effects and in vitro metabotropic glutamate receptor selectivity of (S)-4-carboxy-3-hydroxy-phenylglycine [(S)-4C3HPG] were examined. Intracerebroventricular injection of (S)-4C3HPG dose-dependently antagonized audiogenic-induced clonic and tonic convulsions in DBA/2 mice with ED50 values of 76 and 110-nmol per mouse, respectively. (S)-4C3HPG dose-dependently inhibited the spontaneously evoked epileptic spikes in a cingulate cortex-corpus callosum slice preparation. (S)-4C3HPG displaced the binding of [3H]glutamate in membranes prepared from baby hamster kidney (BHK) cells expressing the metabotropic glutamate receptor mGluR1a with an EC50 of 5 +/- 1 microM. (S)-4C3HPG dose-dependently antagonized glutamate-stimulated phosphoinositide hydrolysis in BHK cells expressing mGluR1a with an IC50 of 15 +/- 3 microM. (S)-4C3HPG was, however, an agonist at mGluR2 with an EC50 of 21 +/- 4 microM for inhibition of forskolin-stimulated cyclic AMP formation in BHK cells expressing the mGluR2. (S)-4C3HPG had no effects at mGluR4a. These data suggest that the anticonvulsant action of (S)-4C3HPG is mediated by combined antagonism of mGluR1a and agonism of mGluR2. These results suggest the importance of mGluR1a and/or mGluR2 in the control of epileptic activity.
(S)-4-carboxy-3-hydroxyphenylglycine activates phosphatidyl inositol linked metabotropic glutamate receptors in different brain regions of the neonatal rat.[Pubmed:9460705]
Neurochem Int. 1998 Jan;32(1):77-85.
In the present investigation, effects of several agonists and antagonists of metabotropic glutamate receptors (mGluRs) which are coupled to phosphatidyl inositol (PI) hydrolysis were evaluated in slices of neonatal rat hippocampus, striatum, cortex and cerebellum. The rank order of potency of agonists in the PI hydrolysis assay was identical in all brain regions: quisqualic acid (Quis) > (RS)-3,5-dihydroxyphenylglycine (3,5-DHPG) > 1S, 3R-aminocyclopentane dicarboxylic acid (1S,3R-ACPD) >> L-glutamate (Glu). All agonists were equiefficacious in the four brain regions tested. The responses to 3,5-DHPG, a highly selective Class I mGluR agonist, were attenuated by (S)-4-carboxyphenylglycine ((S)-4CPG), (+)-alpha-methyl-4-carboxyphenylglycine ((+)-MCPG) and 1-aminoindan-1,5-dicarboxylic acid (UPF-523) with a rank order of potency of (+)-MCPG > or = (S)-4CPG > or = UPF-523 in the different brain regions. These results suggest little selectivity among these putative mGluR antagonists in the different brain regions studied. Interestingly, (S)-4-Carboxy-3-hydroxyphenylglycine ((S)-4C3HPG), a compound reported to act as antagonist at Class I mGluRs, produced concentration-dependent increases in PI hydrolysis in all four brain regions suggesting that (S)-4C3HPG acts as an agonist. In striatum, hippocampus and cortex, (S)-4C3HPG was equiefficacious to Quis, 3,5-DHPG, 1S,3R-ACPD and Glu. However, in the cerebellum, (S)-4C3HPG displayed weak agonist activity (37% of that of a maximally effective concentration of Quis). The effects of (S)-4C3HPG in the PI hydrolysis assay appeared to be mediated by the activation of an mGluR subtype since it was significantly blocked by (S)-4CPG, an mGluR antagonist. In addition, the agonistic effects of (S)-4C3HPG appear to be unrelated to inhibition of [3H]-Glu uptake into rat hippocampal or cerebellar synaptosomes. These results demonstrate a unique pharmacological profile of (S)-4C3HPG which can be interpreted as (S)-4C3HPG being a highly selective mGluR5 agonist or alternatively, that the effects of (S)-4C3HPG may be mediated through a novel Class I mGluR subtype(s), yet to be identified.
Structure-activity relationships of new agonists and antagonists of different metabotropic glutamate receptor subtypes.[Pubmed:8730745]
Br J Pharmacol. 1996 Apr;117(7):1493-503.
1. We investigated the agonist and antagonist activities of 22 new phenylglycine and phenylalanine derivatives for metabotropic glutamate receptors (mGluRs) by examining their effects on the signal transduction of mGluR1, mGluR2 and mGluR6 subtypes expressed in Chinese hamster ovary cells. This analysis revealed several structural characteristics that govern receptor subtype specificity of the agonist and antagonist activities of phenylglycine derivatives. 2. Hydroxyphenylglycine derivatives possessed either an agonist activity on mGluR1/mGluR6 or an antagonist activity on mGluR1. 3. Carboxyphenylglycine derivatives showed an agonist activity on mGluR2 but an antagonist activity on mGluR1. 4. alpha-Methylation or alpha-ethylation of the carboxyphenylglycine derivatives converts the agonist property for mGluR2 to an antagonist property, thus producing antagonists at both mGluR1 and mGluR2. 5. Structurally-corresponding phenylalanine derivatives showed little or no agonist or antagonist activity on any subtypes of the receptors. 6. This investigation demonstrates that the nature and positions of side chains and ring substituents incorporated into the phenylglycine structure are critical in determining the agonist and antagonist activities of members of this group of compounds on different subtypes of the mGluR family. 7. We also tested two alpha-methyl derivatives of mGluR agonists. (2S, 1'S, 2'S)-2-(2-Carboxycyclopropyl)glycine (L-CCG-I) is a potent agonist for mGluR2 but alpha-methylation of this compound changes its activity to that of an mGluR2-selective antagonist. In contrast, alpha-methylation of L-2-amino-4-phosphonobutyrate (L-AP4) results in retention of an agonist activity on mGluR6. Thus, alpha-methylation produces different effects, depending on the chemical structures of lead compounds and/or on the subtype of mGluR tested.
Pharmacological analysis of 4-carboxyphenylglycine derivatives: comparison of effects on mGluR1 alpha and mGluR5a subtypes.[Pubmed:8532170]
Neuropharmacology. 1995 Aug;34(8):887-94.
The antagonist effects of the 4-carboxyphenylglycines: (S)-4-carboxy-3hydroxyphenylglycine (4C3HPG), (S)-4-carboxyphenylglycine (4CPG) and (+)-alpha-methyl-4-carboxyphenylglycine (M4CPG) were compared on functional responses of human metabotropic glutamate receptor (mGluR) subtypes mGluR1 alpha and mGluR5a. These receptors both belong to group 1 type mGluRs which couple to the phosphoinositide (PI) hydrolysis/[Ca2+]i mobilization signal transduction pathway and are closely related in both structure and agonist pharmacology. In this study, the IC50 values obtained for quisqualate induced PI hydrolysis responses show that although all the phenylglycines are antagonists for both mGluR1 alpha and mGluR5a, the compounds exhibit differential potencies at these receptor subtypes. The 4C3HPG derivative was the most potent antagonist for both mGluR1 alpha (IC50 range: 19-50 microM) and mGluR5a (IC50 range: 53-280 microM). 4CPG produced an IC50 range of 4r-72 microM for mGluR1 alpha and 150-156 microM for mGluR5a cells. The potency of the M4CPG could not be distinguished from that of 4CPG with IC50 ranges of 29-100 microM and 115-210 microM for mGluR1 alpha and mGluR5a respectively. Further characterization of the dose-response effects of the compounds on quisqualate induced [Ca2+]i mobilization showed that although the magnitude of phenylglycine inhibition was reduced for both mGluR subtypes compared to those observed for stimulation of PI hydrolysis (except for 4C3HPG on mGluR1 alpha), similar differences in the relative potencies of the phenylglycines between mGluR1 alpha (IC50s: 40 +/- 10 microM for 4C3HPG: 300-1000 microM for 4CPG and M4CPG) and mGluR5a (IC50s: > 1000 microM) were evident.(ABSTRACT TRUNCATED AT 250 WORDS)
Analysis of agonist and antagonist activities of phenylglycine derivatives for different cloned metabotropic glutamate receptor subtypes.[Pubmed:8182479]
J Neurosci. 1994 May;14(5 Pt 2):3370-7.
The metabotropic glutamate receptors (mGluRs) consist of at least seven different subtypes and are coupled to intracellular signal transduction via G proteins. However, the lack of specific antagonists for the mGluRs limited the precise characterization of the role of the individual mGluRs. In this study, we investigated the agonist and antagonist activities of a series of phenylglycine derivatives for the mGluRs by examining their effects on the signal transduction of representative mGluR1, mGluR2, and mGluR4 subtypes expressed individually in Chinese hamster ovary cells. The phenylglycine derivatives examined included (S)- and (R)-forms of 3-hydroxyphenylglycine (3HPG), 4-carboxy-phenylglycine (4CPG), 4-carboxy-3-hydroxyphenylglycine (4C3HPG), 3-carboxy-4-hydroxyphenylglycine (3C4HPG), and (+)- and (-)-alpha-methyl-4-carboxyphenylglycine (alpha M4CPG). Among these 10 compounds, (S)-3HPG acted as an agonist for mGluR1, while (S)-4C3HPG, (S)-3C4HPG, and (S)-4CPG served as effective agonists for mGluR2. The rank order of agonist potencies for mGluR2 was L-glutamate > (S)-4C3HPG > (S)-3C4HPG > (S)-4CPG. No other phenylglycine derivatives showed any definite agonist activity on either mGluR1 or mGluR2. Among the phenylglycine derivatives with no mGluR1 agonist activity, (S)-4C3HPG, (S)-3C4HPG, (S)-4CPG, and (+)-alpha M4CPG effectively antagonized the action of L-glutamate on mGluR1. The rank order of antagonist potencies was (S)-4C3HPG > or = (S)-4CPG > or = (+)-alpha M4CPG > (S)-3C4HPG. The Schild plot analysis indicated that (RS)-4C3HPG, (S)-4CPG, and (+)-alpha M4CPG all act as competitive antagonists for mGluR1 with pA2 values of 4.38, 4.46, and 4.38, respectively.(ABSTRACT TRUNCATED AT 250 WORDS)
Phenylglycine derivatives as new pharmacological tools for investigating the role of metabotropic glutamate receptors in the central nervous system.[Pubmed:7680790]
Neuroscience. 1993 Feb;52(3):481-8.
The possible roles of G-protein coupled metabotropic glutamate receptors in central nervous function are currently the focus of intensive investigation. The complexity of effects produced by agonists at these receptors probably reflects the activity of a range of sub-types. The metabotropic glutamate receptors first described are linked to phospholipase C, mediating phosphoinositide hydrolysis and release of Ca2+ from intracellular stores. A substance generally considered to be a selective agonist for these receptors is (1S,3R)-1-aminocyclopentane-1,3-dicarboxylic acid (ACPD). This substance not only stimulates phosphoinositide hydrolysis, but also inhibits cyclic AMP formation. A family of metabotropic glutamate receptors, incorporating both phospholipase C- and adenylcyclase-linked sub-types has been cloned. Various effects of metabotropic glutamate receptor agonists on membrane ion fluxes and synaptic events have been reported, including neuronal depolarization and/or excitation, hyperpolarization, inhibition of Ca(2+)-dependent and voltage-gated K+ currents, potentiation of N-methyl-D-aspartate-induced responses, depression of synaptic excitation and either induction or augmentation of long-term potentiation. To clarify the role of metabotropic glutamate receptors in central nervous activity and to aid the characterization of the various receptor types that may be involved, a range of highly selective agonists and antagonists is required. To date, currently available antagonists such as L-2-amino-3-phosphonopropionate and L-aspartic acid-beta-hydroxamate appear to be unselective and insufficiently potent. We report here the actions of three phenylglycine derivatives, the particular agonist and/or antagonist properties of which may help to elucidate the roles of metabotropic glutamate receptors in central nervous activity.(ABSTRACT TRUNCATED AT 250 WORDS)


