(2,4-Dihydroxyphenyl)acetonitrileCAS# 57576-34-8 |
Quality Control & MSDS
Number of papers citing our products

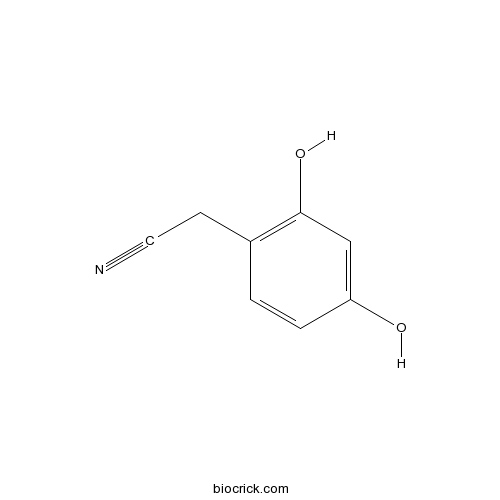
Chemical structure

3D structure
| Cas No. | 57576-34-8 | SDF | Download SDF |
| PubChem ID | 14970422 | Appearance | Powder |
| Formula | C8H7NO2 | M.Wt | 149.2 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 2-(2,4-dihydroxyphenyl)acetonitrile | ||
| SMILES | C1=CC(=C(C=C1O)O)CC#N | ||
| Standard InChIKey | SSUJMKQLZZYAFW-UHFFFAOYSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | (2,4-Dihydroxyphenyl)acetonitrile is a natural product from Diploclisia glaucescens. |
| In vitro | The allelopathic potential of Erica scoparia L.[Reference: WebLink]Oecologia, 1977 , 30 (1) :55-61.The phytochemical action of Erica scoparia L. upon its environment was studied.
|

(2,4-Dihydroxyphenyl)acetonitrile Dilution Calculator

(2,4-Dihydroxyphenyl)acetonitrile Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 6.7024 mL | 33.5121 mL | 67.0241 mL | 134.0483 mL | 167.5603 mL |
| 5 mM | 1.3405 mL | 6.7024 mL | 13.4048 mL | 26.8097 mL | 33.5121 mL |
| 10 mM | 0.6702 mL | 3.3512 mL | 6.7024 mL | 13.4048 mL | 16.756 mL |
| 50 mM | 0.134 mL | 0.6702 mL | 1.3405 mL | 2.681 mL | 3.3512 mL |
| 100 mM | 0.067 mL | 0.3351 mL | 0.6702 mL | 1.3405 mL | 1.6756 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- (24S)-Cycloartane-3,24,25-triol 24,25-acetonide
Catalog No.:BCN1414
CAS No.:57576-31-5
- (3beta,24xi)-Cycloartane-3,24,25-triol
Catalog No.:BCN5782
CAS No.:57576-29-1
- PNU 37883 hydrochloride
Catalog No.:BCC7262
CAS No.:57568-80-6
- Isofuranodiene
Catalog No.:BCN5781
CAS No.:57566-47-9
- SNOG
Catalog No.:BCC6714
CAS No.:57564-91-7
- Notoginsenoside S
Catalog No.:BCN8371
CAS No.:575446-95-6
- H-His(Nτ-Me)-OMe.2HCl
Catalog No.:BCC2958
CAS No.:57519-09-2
- 2-Methylsulfanylpyrimidin-4(3H)-one
Catalog No.:BCC8582
CAS No.:5751-20-2
- 6-Benzyloxypurine
Catalog No.:BCC8770
CAS No.:57500-07-9
- 3-Benzalphthalide
Catalog No.:BCC8621
CAS No.:575-61-1
- 3,4'-Di-O-methylellagic acid
Catalog No.:BCN3710
CAS No.:57499-59-9
- Carpachromene
Catalog No.:BCN5779
CAS No.:57498-96-1
- Norcepharadione B
Catalog No.:BCN5784
CAS No.:57576-41-7
- Aclacinomycin A
Catalog No.:BCC1232
CAS No.:57576-44-0
- Cycloartane-3,24,25-triol
Catalog No.:BCC8922
CAS No.:57586-98-8
- Biocytin
Catalog No.:BCC7659
CAS No.:576-19-2
- Piperenone
Catalog No.:BCN6578
CAS No.:57625-31-7
- Fenobam
Catalog No.:BCC7345
CAS No.:57653-26-6
- Baccatin IV
Catalog No.:BCN5785
CAS No.:57672-77-2
- 1-Dehydroxybaccatin IV
Catalog No.:BCN7211
CAS No.:57672-78-3
- Baccatin VI
Catalog No.:BCN7229
CAS No.:57672-79-4
- Palmitic acid-1-13C
Catalog No.:BCC8229
CAS No.:57677-53-9
- Kansuinine B
Catalog No.:BCN3766
CAS No.:57685-46-8
- Flavanomarein
Catalog No.:BCN6429
CAS No.:577-38-8
The allelopathic potential of Erica scoparia L.
Ballester, A., Albo, J.M. & Vieitez, E. Oecologia (1977) 30: 55
The phytochemical action of Erica scoparia L. upon its environment was studied. The toxicity of biogenic heath products on red clover seedlings was demonstrated by biological tests. Ten phenolic hydrosoluble compounds were identified; p-Hydroxybenzoic, vanillic, syringic, caffeic, ferulic, p-coumaric, protocatechuic, genetisic, and 2-hydroxyphenyl acetic acid, as well as (2,4-Dihydroxyphenyl)acetonitrile. The suppression of herbs, especially grasses, in heathlands can be explained, at least partially, by the allelopathic effects of E. scoparia L.


