TozadenantAdenosine 2a receptor antagonist,novel and selective CAS# 870070-55-6 |
- Preladenant
Catalog No.:BCC1868
CAS No.:377727-87-2
- ANR 94
Catalog No.:BCC7815
CAS No.:634924-89-3
Quality Control & MSDS
Number of papers citing our products

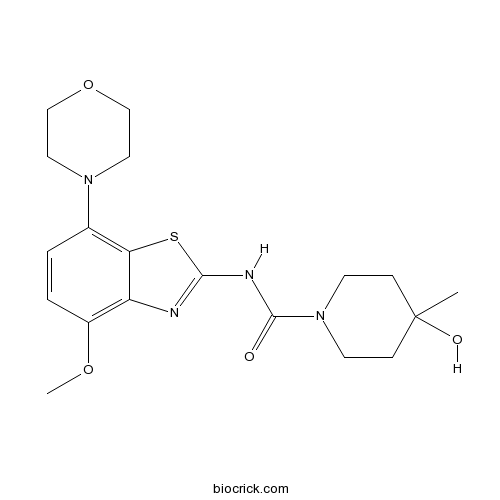
Chemical structure
3D structure
| Cas No. | 870070-55-6 | SDF | Download SDF |
| PubChem ID | 11618368 | Appearance | Powder |
| Formula | C19H26N4O4S | M.Wt | 406.5 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | SYN115 | ||
| Solubility | DMSO : 50 mg/mL (123.00 mM; Need ultrasonic) | ||
| Chemical Name | 4-hydroxy-N-(4-methoxy-7-morpholin-4-yl-1,3-benzothiazol-2-yl)-4-methylpiperidine-1-carboxamide | ||
| SMILES | CC1(CCN(CC1)C(=O)NC2=NC3=C(C=CC(=C3S2)N4CCOCC4)OC)O | ||
| Standard InChIKey | XNBRWUQWSKXMPW-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C19H26N4O4S/c1-19(25)5-7-23(8-6-19)18(24)21-17-20-15-14(26-2)4-3-13(16(15)28-17)22-9-11-27-12-10-22/h3-4,25H,5-12H2,1-2H3,(H,20,21,24) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Tozadenant is an antagonist of adenosine A2A receptor. | |||||
| Targets | adenosine A2A receptor | |||||
| Human experiment [1]: | |
| Patients | Patients with Parkinson’s disease. |
| Dosage form | 60, 120, 180, or 240 mg twice daily for 12 weeks. |
| Application | Tozadenant increases mean daily total on-time and improved total UPDRS scores for parts I-III in the combined tozadenant 120 mg twice-daily and 180 mg twice-daily group and in the 120 mg twice-daily group. Also, tozadenant significantly improved CGI-S and CGI-I scores. |
| Other notes | Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
| References: [1]. Hauser RA, Olanow CW, Kieburtz KD, et al. Tozadenant (SYN115) in patients with Parkinson's disease who have motor fluctuations on levodopa: a phase 2b, double-blind, randomised trial. Lancet Neurol,2014, 13(8): 767-776. | |

Tozadenant Dilution Calculator

Tozadenant Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.46 mL | 12.3001 mL | 24.6002 mL | 49.2005 mL | 61.5006 mL |
| 5 mM | 0.492 mL | 2.46 mL | 4.92 mL | 9.8401 mL | 12.3001 mL |
| 10 mM | 0.246 mL | 1.23 mL | 2.46 mL | 4.92 mL | 6.1501 mL |
| 50 mM | 0.0492 mL | 0.246 mL | 0.492 mL | 0.984 mL | 1.23 mL |
| 100 mM | 0.0246 mL | 0.123 mL | 0.246 mL | 0.492 mL | 0.615 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Tozadenant (SYN115) is an orally active, selective adenosine A2A receptor antagonist.
In animal models of Parkinson’s disease, tozadenant improves motor neuron function.
Tozadenant (60 mg twice daily) was not associated with a signi?cant reduction in o?-time, and tozadenant (240 mg twice daily) was associated with an increased rate of discontinuation because of adverse events (17 of 84 patients [20%]). Tozadenant at dose of 120 or 180 mg twice daily was e?ective at reducing o?-time and was generally well tolerated. Further investigation of tozadenant treatment in phase 3 trials is guaranteed . Perfusion MRI showed that tozadenant induced highly signi?cant suppressed in regional cerebral blood ?ow, with the most important decreases occurring in bilateral thalami.
Reference:
Hauser RA, Olanow CW, Kieburtz KD et al. Tozadenant (SYN115) in patients with Parkinson's disease who have motor fluctuations on levodopa: a phase 2b, double-blind, randomised trial. Lancet Neurol. 2014 Aug;13(8):767-76.
- A 839977
Catalog No.:BCC4290
CAS No.:870061-27-1
- xylitol pentacetate
Catalog No.:BCN6267
CAS No.:13437-68-8
- Inositol
Catalog No.:BCN8471
CAS No.:87-89-8
- D-Mannitol busulfan
Catalog No.:BCN3789
CAS No.:1187-00-4
- Tartaric acid
Catalog No.:BCN3824
CAS No.:87-69-4
- Pyrogallol
Catalog No.:BCN4424
CAS No.:87-66-1
- Gramine
Catalog No.:BCN4959
CAS No.:87-52-5
- trans-Caryophyllene
Catalog No.:BCN2644
CAS No.:87-44-5
- Isosorbide dinitrate
Catalog No.:BCC9004
CAS No.:87-33-2
- Ac-DL-Trp-OH
Catalog No.:BCC3119
CAS No.:87-32-1
- Salicylanilide
Catalog No.:BCC4712
CAS No.:87-17-2
- Thiolutin
Catalog No.:BCC2471
CAS No.:87-11-6
- Apilimod mesylate
Catalog No.:BCC5287
CAS No.:870087-36-8
- Acalisib (GS-9820)
Catalog No.:BCC6384
CAS No.:870281-34-8
- CAL-101 (Idelalisib, GS-1101)
Catalog No.:BCC1270
CAS No.:870281-82-6
- 3,23-Dioxo-9,19-cyclolanost-24-en-26-oic acid
Catalog No.:BCN1322
CAS No.:870456-88-5
- Bis-5,5-nortrachelogenin
Catalog No.:BCN6516
CAS No.:870480-56-1
- GW2580
Catalog No.:BCC1096
CAS No.:870483-87-7
- Ritanserin
Catalog No.:BCC7214
CAS No.:87051-43-2
- Zeylasteral
Catalog No.:BCN3065
CAS No.:87064-16-2
- Euphohelioscopin A
Catalog No.:BCN6501
CAS No.:87064-61-7
- PAP-1
Catalog No.:BCC1836
CAS No.:870653-45-5
- AC 261066
Catalog No.:BCC7848
CAS No.:870773-76-5
- Leptomycin B
Catalog No.:BCC7223
CAS No.:87081-35-4
Pharmacokinetics and metabolism of [(14)C]-tozadenant (SYN-115), a novel A2a receptor antagonist ligand, in healthy volunteers.[Pubmed:27489076]
Xenobiotica. 2017 Aug;47(8):705-718.
1. This phase-I study (NCT02240290) was designed to investigate the human absorption, disposition and mass balance of (14)C-Tozadenant, a novel A2a receptor antagonist in clinical development for Parkinson s disease. 2. Six healthy male subjects received a single oral dose of Tozadenant (240 mg containing 81.47 KBq of [(14)C]-Tozadenant). Blood, urine and feces were collected over 14 days. Radioactivity was determined by liquid scintillation counting or accelerator mass spectrometry (AMS). Tozadenant and metabolites were characterized using HPLC-MS/MS and HPLC-AMS with fraction collection. 3. At 4 h, the Cmax of Tozadenant was 1.74 mug/mL and AUC(0-t) 35.0 h mug/mL, t1/2 15 h, Vz/F 1.82 L/kg and CL/F 1.40 mL/min/kg. For total [(14)C] radioactivity, the Cmax was 2.29 mug eq/mL at 5 h post-dose and AUC(0-t) 43.9 h mug eq/mL. Unchanged Tozadenant amounted to 93% of the radiocarbon AUC(0-48h). At 312 h post-dose, cumulative urinary and fecal excretion of radiocarbon reached 30.5% and 55.1% of the dose, respectively. Unchanged Tozadenant reached 11% in urine and 12% of the dose in feces. Tozadenant was excreted as metabolites, including di-and mono-hydroxylated metabolites, N/O dealkylated metabolites, hydrated metabolites. 4. The only identified species circulating in plasma was unchanged Tozadenant. Tozadenant was primarily excreted in urine and feces in the form of metabolites.
Tozadenant (SYN115) in patients with Parkinson's disease who have motor fluctuations on levodopa: a phase 2b, double-blind, randomised trial.[Pubmed:25008546]
Lancet Neurol. 2014 Aug;13(8):767-76.
BACKGROUND: Many patients with Parkinson's disease have motor fluctuations despite treatment with available drugs. Tozadenant (SYN115) is an oral, selective adenosine A2A receptor antagonist that improves motor function in animal models of Parkinson's disease. We aimed to assess the safety and efficacy of Tozadenant as an adjunct to levodopa in patients with Parkinson's disease who have motor fluctuations on levodopa. METHODS: We did an international, multicentre, phase 2b, randomised, double-blind, placebo-controlled, parallel-group, dose-finding clinical trial of Tozadenant in levodopa-treated patients with Parkinson's disease who had motor fluctuations (at least 2.5 h off-time per day). Eligible patients were randomly assigned via a computer-generated randomisation schedule to receive Tozadenant 60, 120, 180, or 240 mg or matching placebo twice daily for 12 weeks. All study management, site personnel, and patients were masked to treatment assignment. The primary outcome was change from baseline to week 12 in hours per day spent in the off-state (assessed from Parkinson's disease diaries completed by patients). This study is registered at ClinicalTrials.gov, number NCT01283594. FINDINGS: Of 420 randomised patients (mean age 63.3 [SD 8.3] years; mean duration of Parkinson's disease 8.7 [4.7] years), 403 provided post-baseline diary data and 337 completed study treatment. Compared with placebo, mean daily off-time was significantly reduced in the combined Tozadenant 120 mg twice-daily and 180 mg twice-daily group (-1.1 h, 95% CI -1.8 to -0.5; p=0.0006), the Tozadenant 120 mg twice-daily group (-1.1 h, -1.8 to -0.4; p=0.0039), and the Tozadenant 180 mg twice-daily group (-1.2 h, -1.9 to -0.4; p=0.0039). The most common adverse events in these groups were dyskinesia (seven [8%] of 84 patients in the placebo group, 13 [16%] of 82 in the 120 mg twice-daily group, and 17 [20%] of 85 in the 180 mg twice-daily group), nausea (three [4%], 9 [11%], and ten [12%]), and dizziness (one [1%], four [5%], and 11 [13%]). Tozadenant 60 mg twice daily was not associated with a significant reduction in off-time, and Tozadenant 240 mg twice daily was associated with an increased rate of discontinuation because of adverse events (17 [20%] of 84 patients). INTERPRETATION: Tozadenant at 120 or 180 mg twice daily was generally well tolerated and was effective at reducing off-time. Further investigation of Tozadenant treatment in phase 3 trials is warranted. FUNDING: Biotie Therapies.
Adenosine 2A receptor occupancy by tozadenant and preladenant in rhesus monkeys.[Pubmed:25082853]
J Nucl Med. 2014 Oct;55(10):1712-8.
UNLABELLED: Motor symptoms in Parkinson disease (PD) are caused by a loss of dopamine input from the substantia nigra to the striatum. Blockade of adenosine 2A (A(2A)) receptors facilitates dopamine D(2) receptor function. In phase 2 clinical trials, A(2A) antagonists (istradefylline, preladenant, and Tozadenant) improved motor function in PD. We developed a new A(2A) PET radiotracer, (18)F-MNI-444, and used it to investigate the relationship between plasma levels and A(2A) occupancy by preladenant and Tozadenant in nonhuman primates (NHP). METHODS: A series of 20 PET experiments was conducted in 5 adult rhesus macaques. PET data were analyzed with both plasma-input (Logan graphical analysis) and reference-region-based (simplified reference tissue model and noninvasive Logan graphical analysis) methods. Whole-body PET images were acquired for radiation dosimetry estimates. Human pharmacokinetic parameters for Tozadenant and preladenant were used to predict A(2A) occupancy in humans, based on median effective concentration (EC(50)) values estimated from the NHP PET measurements. RESULTS: (18)F-MNI-444 regional uptake was consistent with A(2A) receptor distribution in the brain. Selectivity was demonstrated by dose-dependent blocking by Tozadenant and preladenant. The specific-to-nonspecific ratio was superior to that of other A(2A) PET radiotracers. Pharmacokinetic modeling predicted that Tozadenant and preladenant may have different profiles of A(2A) receptor occupancy in humans. CONCLUSION: (18)F-MNI-444 appears to be a better PET radiotracer for A(2A) imaging than currently available radiotracers. Assuming that EC(50) in humans is similar to that in NHP, it appears that Tozadenant will provide a more sustained A(2A) receptor occupancy than preladenant in humans at clinically tested doses.
Two new adenosine receptor antagonists for the treatment of Parkinson's disease: istradefylline versus tozadenant.[Pubmed:24673462]
Expert Opin Pharmacother. 2014 Jun;15(8):1097-107.
INTRODUCTION: Adenosine A2A receptors are localized in the brain, mainly within the caudate and putamen nuclei of the basal ganglia. Their activation leads to stimulation of the 'indirect' pathway. Conversely, administration of A2A receptor antagonists leads to inhibition of this pathway, which was translated into reduced hypomotility in several animal models of parkinsonism. AREAS COVERED: In this review, the effects of two A2A receptor antagonists, istradefylline and Tozadenant, on parkinsonian symptoms in animal and humans will be discussed. EXPERT OPINION: Animal studies have shown potent antiparkinsonian effects for several A2A receptor antagonists, including istradefylline. In clinical trials, istradefylline reduced OFF time when administered with levodopa, but results are inconclusive. Results with Tozadenant are scarce. Modification of thalamic blood flow compatible with reduced inhibition was noted in one small trial, followed by a significant reduction in OFF time in a larger one. Therefore, both drugs show promising efficacy for the reduction of OFF time in levodopa-treated Parkinson's disease patients, but further research is needed in order to obtain definitive conclusions.


