SalvinoloneCAS# 120278-22-0 |
Quality Control & MSDS
Number of papers citing our products

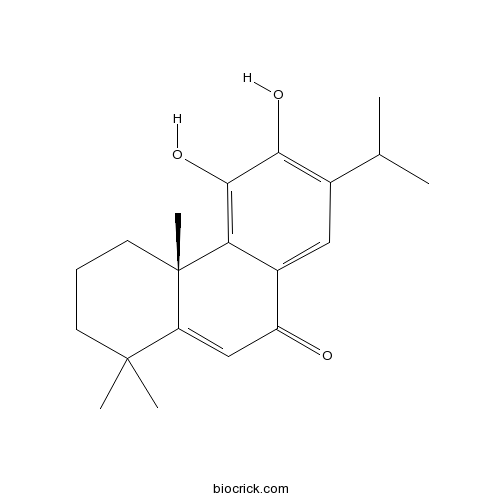
Chemical structure

3D structure
| Cas No. | 120278-22-0 | SDF | Download SDF |
| PubChem ID | 11723174 | Appearance | Powder |
| Formula | C20H26O3 | M.Wt | 314.4 |
| Type of Compound | Diterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (4aS)-5,6-dihydroxy-1,1,4a-trimethyl-7-propan-2-yl-3,4-dihydro-2H-phenanthren-9-one | ||
| SMILES | CC(C)C1=C(C(=C2C(=C1)C(=O)C=C3C2(CCCC3(C)C)C)O)O | ||
| Standard InChIKey | NPADGWOASIJKSB-FQEVSTJZSA-N | ||
| Standard InChI | InChI=1S/C20H26O3/c1-11(2)12-9-13-14(21)10-15-19(3,4)7-6-8-20(15,5)16(13)18(23)17(12)22/h9-11,22-23H,6-8H2,1-5H3/t20-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Salvinolone, demethyl cryptojaponol and taxodione show potent activity with 4-10 microg/mL of MIC against MRSA and 4-16 microg/mL of MIC against VRE. |
| Targets | Antifection |

Salvinolone Dilution Calculator

Salvinolone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.1807 mL | 15.9033 mL | 31.8066 mL | 63.6132 mL | 79.5165 mL |
| 5 mM | 0.6361 mL | 3.1807 mL | 6.3613 mL | 12.7226 mL | 15.9033 mL |
| 10 mM | 0.3181 mL | 1.5903 mL | 3.1807 mL | 6.3613 mL | 7.9517 mL |
| 50 mM | 0.0636 mL | 0.3181 mL | 0.6361 mL | 1.2723 mL | 1.5903 mL |
| 100 mM | 0.0318 mL | 0.159 mL | 0.3181 mL | 0.6361 mL | 0.7952 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- CNX-774
Catalog No.:BCC4394
CAS No.:1202759-32-7
- AVL-292
Catalog No.:BCC1385
CAS No.:1202757-89-8
- CGS 21680
Catalog No.:BCC1475
CAS No.:120225-54-9
- 1beta-Hydroxyeuscaphic acid
Catalog No.:BCN3517
CAS No.:120211-98-5
- Tenidap
Catalog No.:BCC7419
CAS No.:120210-48-2
- NMS-P715
Catalog No.:BCC6373
CAS No.:1202055-34-2
- Clopidogrel Related Compound C
Catalog No.:BCN2689
CAS No.:120202-71-3
- Clopidogrel
Catalog No.:BCC2497
CAS No.:120202-66-6
- 3,4-Dihydroxycinnamamide
Catalog No.:BCN6090
CAS No.:1202-41-1
- Cynoglossamine
Catalog No.:BCN1970
CAS No.:120193-39-7
- TCS 2210
Catalog No.:BCC7798
CAS No.:1201916-31-5
- MLN9708
Catalog No.:BCC2091
CAS No.:1201902-80-8
- 4-Hydroxysapriparaquinone
Catalog No.:BCN4806
CAS No.:120278-25-3
- Dorzolamide
Catalog No.:BCC4287
CAS No.:120279-96-1
- CX-6258
Catalog No.:BCC1504
CAS No.:1202916-90-2
- Citroside A
Catalog No.:BCN7294
CAS No.:120330-44-1
- DASA-58
Catalog No.:BCC6522
CAS No.:1203494-49-8
- Cyclotraxin B
Catalog No.:BCC6357
CAS No.:1203586-72-4
- AS 1949490
Catalog No.:BCC7762
CAS No.:1203680-76-5
- Jionoside B1
Catalog No.:BCN2858
CAS No.:120406-37-3
- Biapenem
Catalog No.:BCC1071
CAS No.:120410-24-4
- AZD1208
Catalog No.:BCC2079
CAS No.:1204144-28-4
- Icotinib Hydrochloride
Catalog No.:BCC1639
CAS No.:1204313-51-8
- TMC647055
Catalog No.:BCC6376
CAS No.:1204416-97-6
Synthesis of variously oxidized abietane diterpenes and their antibacterial activities against MRSA and VRE.[Pubmed:11249127]
Bioorg Med Chem. 2001 Feb;9(2):347-56.
Variously oxidized 12 natural abietanes, 6,7-dehydroferruginol methyl ether (3), ferruginol (5), 11-hydroxy-12-oxo-7,9(11),13-abietatriene (7), royleanone (9), demethyl cryptojaponol (12), Salvinolone (14), sugiol methyl ether (16), sugiol (17), 5,6-dehydrosugiol methyl ether (19), 5,6-dehydrosugiol (20), 6beta-hydroxyferruginol (23), and taxodione (25) were synthesized. Antimicrobial activities of synthesized phenolic diterpenes and their related compounds against MRSA and VRE were evaluated. Phenols (12-hydroxyabieta-8,11,13-trien-6-one 22 and 23), catechols (12 and 14) and taxodione 25 showed potent activity with 4-10 microg/mL of MIC against MRSA and 4-16 microg/mL of MIC against VRE. (-)-Ferruginol showed more potent activity than natural type (+)-ferruginol. Quinone methide 7 showed the most potent activity with 0.5-1 microg/mL of MIC against both MRSA and VRE.
Antitermitic activities of abietane-type diterpenes from Taxodium distichum cones.[Pubmed:19475449]
J Chem Ecol. 2009 Jun;35(6):635-42.
Eight known abietane-type diterpenes were isolated from the weak acidic fraction of the n-hexane extract from cones of Taxodium distichum, one of the extant, living fossil conifers. They were identified as 6,7-dehydroroyleanone (1), taxodal (2), taxodione (3), Salvinolone (4), 14-deoxycoleon U (5), 5,6-dehydrosugiol (6), sandaracopimaric acid (7), and xanthoperol (8). The structures of these compounds were determined by comparison of NMR spectral data with published data. The antitermitic (termicidal and antifeedant) activities of the compounds 1-8 against the subterranean termite, Reticulitermes speratus Kolbe, were evaluated. Compounds 1 and 3 showed potent termicidal activity, and 5 and 8 showed potent antifeedant activity. Compound 1 was found to be one of the representative bioactive compounds in the n-hexane extract of T. distichum cones. Compounds 1-8, with the exception of 7, were oxides of ferruginol (9). Therefore, the presence of various oxidation forms of the abietane-type structure reflects their various bioactivities.
Bioactive diterpenes from Clerodendrum kaichianum.[Pubmed:21366034]
Nat Prod Commun. 2011 Jan;6(1):3-5.
Bioassay-guided isolation studies of the extract of Clerodendrum kaichianum Hsu., a new rearranged abietane diterpene and five known diterpene compounds were isolated by various chromatography methods. Their structures were identified by means of spectroscopic methods, including 1D- and 2D-NMR spectroscopy, as (16R)-12,16-epoxy-11,14,17-trihydroxy-17(15-->16)-abeo-8,11,13-abietatrien-7-one (1), villosin A (2), Salvinolone (3), 14-deoxyloleon U (4), 5,6-dehydrosugiol (5), and coleon U (6). Compounds 1, 2, 3, and 5 are reported for the first time for this genus. Compounds 1, 2, and 6 demonstrated potent cytotoxic activities against the HL-60 tumor cell line.


