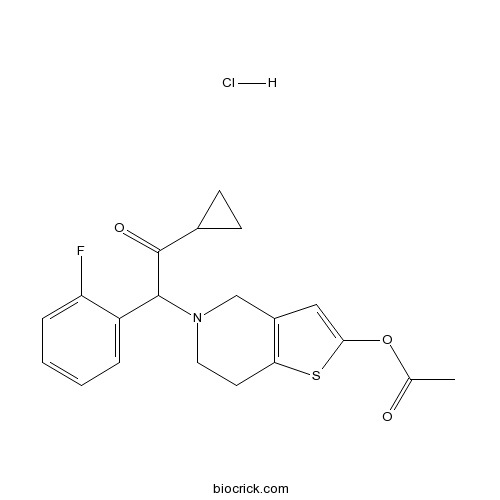
Prasugrel hydrochlorideCAS# 389574-19-0 |
- Dexpramipexole dihydrochloride
Catalog No.:BCC1528
CAS No.:104632-27-1
- Dexpramipexole
Catalog No.:BCC1527
CAS No.:104632-28-2
- Cariprazine hydrochloride
Catalog No.:BCC1454
CAS No.:1083076-69-0
- Cariprazine
Catalog No.:BCC1453
CAS No.:839712-12-8
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 389574-19-0 | SDF | Download SDF |
| PubChem ID | 10158453 | Appearance | Powder |
| Formula | C20H21ClFNO3S | M.Wt | 409.9 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 41.67 mg/mL (101.66 mM; Need ultrasonic) | ||
| Chemical Name | [5-[2-cyclopropyl-1-(2-fluorophenyl)-2-oxoethyl]-6,7-dihydro-4H-thieno[3,2-c]pyridin-2-yl] acetate;hydrochloride | ||
| SMILES | CC(=O)OC1=CC2=C(S1)CCN(C2)C(C3=CC=CC=C3F)C(=O)C4CC4.Cl | ||
| Standard InChIKey | JALHGCPDPSNJNY-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H20FNO3S.ClH/c1-12(23)25-18-10-14-11-22(9-8-17(14)26-18)19(20(24)13-6-7-13)15-4-2-3-5-16(15)21;/h2-5,10,13,19H,6-9,11H2,1H3;1H | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Prasugrel hydrochloride is a platelet inhibitor with IC50 value of 1.8 μM.
Target: P2Y12 receptor
Prasugrel hydrochloride is a novel platelet inhibitor used for the reduction of thrombotic cardiovascular events (including stent thrombosis) in patients with acute coronary syndrome who are to be managed with PCI [2].
Prasugrel hydrochloride reduces the aggregation (clumping) of platelets by irreversibly binding to P2Y12 receptors. In rat platelets, prasugrel hydrochloride AM inhibited in vitro platelet aggregation induced by ADP (10 μm) with an IC50 value of 1.8 Μm [2].
Clinical indications: Acute coronary syndrome Ischemic heart disease Sickle cell anemia Stroke Vascular occlusive disease
FDA Approved Date: February 2009
Toxicity: Hypertension Headache Hypercholesterolemia/hyperlipidemia Nausea Epistaxis References: | |||||

Prasugrel hydrochloride Dilution Calculator

Prasugrel hydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.4396 mL | 12.1981 mL | 24.3962 mL | 48.7924 mL | 60.9905 mL |
| 5 mM | 0.4879 mL | 2.4396 mL | 4.8792 mL | 9.7585 mL | 12.1981 mL |
| 10 mM | 0.244 mL | 1.2198 mL | 2.4396 mL | 4.8792 mL | 6.099 mL |
| 50 mM | 0.0488 mL | 0.244 mL | 0.4879 mL | 0.9758 mL | 1.2198 mL |
| 100 mM | 0.0244 mL | 0.122 mL | 0.244 mL | 0.4879 mL | 0.6099 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Prasugrel hydrochloride is a platelet inhibitor with IC50 value of 1.8 μM.
- Piperlotine A
Catalog No.:BCN6481
CAS No.:389572-70-7
- Icariside D2
Catalog No.:BCN7217
CAS No.:38954-02-8
- Isovitexin
Catalog No.:BCN5441
CAS No.:38953-85-4
- Vitexin 4'-glucoside
Catalog No.:BCC9252
CAS No.:38950-94-6
- Chelerythrine chloride
Catalog No.:BCN8322
CAS No.:3895-92-9
- SBHA
Catalog No.:BCC2425
CAS No.:38937-66-5
- Isodeoxyelephantopin
Catalog No.:BCN4638
CAS No.:38927-54-7
- erythro-Guaiacylglycerol
Catalog No.:BCN5440
CAS No.:38916-91-5
- Halaminol C
Catalog No.:BCN1787
CAS No.:389125-68-2
- Halaminol B
Catalog No.:BCN1748
CAS No.:389125-59-1
- Halaminol A
Catalog No.:BCN1788
CAS No.:389125-56-8
- Z-Glu(OtBu)-OH
Catalog No.:BCC2776
CAS No.:3886-08-6
- Prasugrel Maleic acid
Catalog No.:BCC4292
CAS No.:389574-20-3
- Raspberry ketone glucoside
Catalog No.:BCC8244
CAS No.:38963-94-9
- N-Acetylnorloline
Catalog No.:BCN2005
CAS No.:38964-35-1
- Eriodictyol-7-O-glucoside
Catalog No.:BCN4743
CAS No.:38965-51-4
- 2'-O-Methylperlatolic acid
Catalog No.:BCN5442
CAS No.:38968-07-9
- Ophiopogonin B
Catalog No.:BCN5378
CAS No.:38971-41-4
- α-Tocopherol phosphate
Catalog No.:BCC5420
CAS No.:38976-17-9
- (S)-CPW 399
Catalog No.:BCC7106
CAS No.:389888-02-2
- Gelsevirine
Catalog No.:BCN5443
CAS No.:38990-03-3
- 2-Acetyl-6-methoxynaphthalene
Catalog No.:BCC8514
CAS No.:3900-45-6
- Albiflorin
Catalog No.:BCN1264
CAS No.:39011-90-0
- Oxypaeoniflorin
Catalog No.:BCN6346
CAS No.:39011-91-1
Study of the forced degradation behavior of prasugrel hydrochloride by liquid chromatography with mass spectrometry and liquid chromatography with NMR detection and prediction of the toxicity of the characterized degradation products.[Pubmed:26114771]
J Sep Sci. 2015 Sep;38(17):2995-3005.
Prasugrel was subjected to forced degradation studies under conditions of hydrolysis (acid, base, and neutral), photolysis, oxidation, and thermal stress. The drug showed liability in hydrolytic as well as oxidative conditions, resulting in a total of four degradation products. In order to characterize the latter, initially mass fragmentation pathway of the drug was established with the help of mass spectrometry/time-of-flight, multiple stage mass spectrometry and hydrogen/deuterium exchange data. The degradation products were then separated on a C18 column using a stability-indicating volatile buffer method, which was later extended to liquid chromatography-mass spectrometry studies. The latter highlighted that three degradation products had the same molecular mass, while one was different. To characterize all, their mass fragmentation pathways were established in the same manner as the drug. Subsequently, liquid chromatography-nuclear magnetic resonance (NMR) spectroscopy data were collected. Proton and correlation liquid chromatography with NMR spectroscopy studies highlighted existence of diastereomeric behavior in one pair of degradation products. Lastly, toxicity prediction by computer-assisted technology (TOPKAT) and deductive estimation of risk from existing knowledge (DEREK) software were employed to assess in silico toxicity of the characterized degradation products.
Prasugrel Hydrochloride.[Pubmed:26051687]
Profiles Drug Subst Excip Relat Methodol. 2015;40:195-320.
A comprehensive profile of prasugrel HCl is reported herein with 158 references. A full description including nomenclature, formulae, elemental analysis, and appearance is included. Methods of preparation for prasugrel HCl, its intermediates, and derivatives are fully discussed. In addition, the physical properties, analytical methods, stability, uses and applications, and pharmacology of prasugrel HCl are also discussed.
Prasugrel hydrochloride for the treatment of acute coronary syndromes.[Pubmed:25633751]
Expert Opin Pharmacother. 2015 Mar;16(4):585-96.
INTRODUCTION: P2Y12 receptor antagonists, by inhibiting platelet activation and subsequent aggregation, are critical to prevent ischemic event recurrence after an acute coronary syndrome (ACS). Prasugrel is a third-generation thienopyridine whose metabolites target the P2Y12 receptor. Compared with clopidogrel, prasugrel has a more potent, faster in onset, and more consistent P2Y12 receptor inhibition. AREAS COVERED: This review describes prasugrel chemistry, pharmacokinetics, pharmacodynamics and clinical studies. In a Phase III randomized clopidogrel-controlled trial, prasugrel improved cardiovascular outcome (risk reduction of cardiovascular death, non-fatal heart attack and non-fatal stroke) at the cost of increased major and fatal bleeding complications. Prasugrel, in combination with aspirin, has been approved by European and American regulatory agencies for the prevention of atherothrombotic events in patients with ACS who undergo percutaneous coronary intervention (PCI). EXPERT OPINION: Prasugrel is effective for managing ACS patients with planned PCI and it offers an alternative with potential benefits over clopidogrel. Prasugrel is currently challenged by ticagrelor, a P2Y12 receptor antagonist with different pharmacokinetic/pharmacodynamic properties. The superiority of one drug to the other cannot be reliably estimated from the current trials. Ongoing randomized and observational studies may help to provide valuable information on the safety and efficacy of these two drugs and their respective places with ACS patients.
Prasugrel hydrochloride for the treatment of acute coronary syndrome patients.[Pubmed:27701930]
Expert Rev Cardiovasc Ther. 2016 Nov;14(11):1215-1226.
INTRODUCTION: Dual antiplatelet therapy (DAPT) with aspirin combined with either a thienopyridine (clopidogrel or prasugrel) or acyclopentyl-triazolo-pyrimidine (ticagrelor) plays a vital role in the management of acute coronary syndrome (ACS) especially in those undergoing percutaneous coronary intervention (PCI) but even those being managed medically. Observational studies and some formal studies have shown patients on the standard dual antiplatelet regimen (clopidogrel and aspirin) continue to have further ischemic events and can suffer stent thrombosis. It has been demonstrated that clopidogrel is associated with a delayed onset of action with a considerable inter-individual variation to treatment thus making it difficult to achieve an optimal level of platelet inhibition. Areas covered: This article will review the current evidence that is available regarding the effectiveness and safety of prasugrel in ACS patients undergoing percutaneous coronary intervention (PCI). Expert commentary: Prasugrel is an oral third-generation inhibitor of platelet activation and aggregation. Laboratory studies and early phase clinical trials show prasugrel has a faster onset of action, is more potent and has reduced inter-patient response variability compared to clopidogrel. The published studies so far demonstrated that prasugrel when compared to clopidogrel also shows a higher degree of effectiveness in the prevention of platelet-initiated thrombotic events in patients with ACS undergoing PCI, however these benefits are offset somewhat by an increased bleeding risk.


