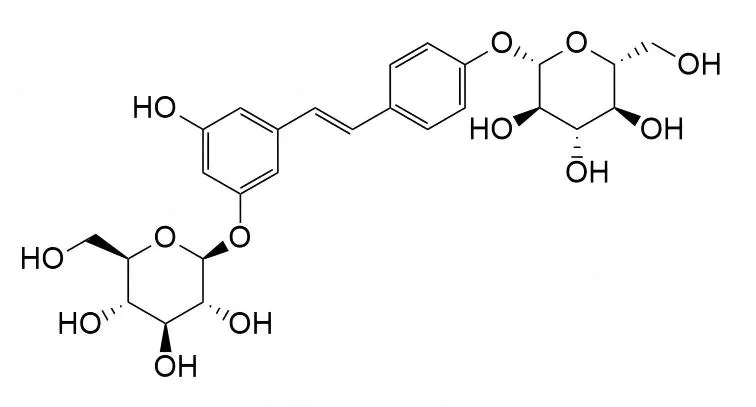
Mulberroside ECAS# 193483-94-2 |
Quality Control & MSDS
Package In Stock
Number of papers citing our products

| Cas No. | 193483-94-2 | SDF | File under preparation. |
| PubChem ID | N/A | Appearance | Powder |
| Formula | C26H32O13 | M.Wt | 552.5 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Mulberroside E Dilution Calculator

Mulberroside E Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.81 mL | 9.0498 mL | 18.0995 mL | 36.1991 mL | 45.2489 mL |
| 5 mM | 0.362 mL | 1.81 mL | 3.6199 mL | 7.2398 mL | 9.0498 mL |
| 10 mM | 0.181 mL | 0.905 mL | 1.81 mL | 3.6199 mL | 4.5249 mL |
| 50 mM | 0.0362 mL | 0.181 mL | 0.362 mL | 0.724 mL | 0.905 mL |
| 100 mM | 0.0181 mL | 0.0905 mL | 0.181 mL | 0.362 mL | 0.4525 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Trollisin I
Catalog No.:BCX2191
CAS No.:911063-04-2
- 6-[2,3-Dihydroxy-1-methoxy-3-methylbutyl]-7-methoxycoumarin)
Catalog No.:BCX2190
CAS No.:425370-70-3
- Ilexosapogenin A
Catalog No.:BCX2189
CAS No.:148031-70-3
- Tricin 4'-O-(erythro-beta-guaiacylglyceryl)ether
Catalog No.:BCX2188
CAS No.:1292294-32-6
- Angelol E
Catalog No.:BCX2187
CAS No.:83156-03-0
- 6-Methyl citrate
Catalog No.:BCX2186
CAS No.:26163-65-5
- Angelol D
Catalog No.:BCX2185
CAS No.:83199-39-7
- Verticilla-4(20),7,11-triene
Catalog No.:BCX2184
CAS No.:386223-19-4
- 7alpha-Hydroxydehydroabietic acid
Catalog No.:BCX2183
CAS No.:76235-98-8
- Pulsatiloside B
Catalog No.:BCX2182
CAS No.:178825-03-1
- Angustone A
Catalog No.:BCX2181
CAS No.:90686-13-8
- 3-O-Feruloyl-5-caffeoylquinic acid
Catalog No.:BCX2180
CAS No.:173658-86-1
- Gymnoside VI
Catalog No.:BCX2193
CAS No.:899430-06-9
- 2',3'-Dihydroxyasarone
Catalog No.:BCX2194
CAS No.:2061275-23-6
- Vernodalol
Catalog No.:BCX2195
CAS No.:65388-17-2
- Hispidulin 7-O-glucuronide
Catalog No.:BCX2196
CAS No.:31105-76-7
- 11alpha,12beta-di-O-tigloyl-tenacigenin B
Catalog No.:BCX2197
CAS No.:154022-56-7
- Diosmetin 6-C-glucoside
Catalog No.:BCX2198
CAS No.:15822-82-9
- 2alpha,3beta,23-trihydroxyurs-20-en-28-oic acid
Catalog No.:BCX2199
CAS No.:948827-08-5
- 4-Methoxy-9,10-dihydrophenanthrene-2,7-di-O-beta-D-glucopyranoside
Catalog No.:BCX2200
CAS No.:1454585-37-5
- 3,3',4-Trimethoxylellagic acid
Catalog No.:BCX2201
CAS No.:124590-44-9
- Orientin 2''-O-(2'''-methylbutyrate)
Catalog No.:BCX2202
CAS No.:681152-44-3
- L-Tryptophan methyl ester
Catalog No.:BCX2203
CAS No.:4299-70-1
- Tenacissoside D
Catalog No.:BCX2204
CAS No.:107343-57-9
Identification of natural inhibitors against prime targets of SARS-CoV-2 using molecular docking, molecular dynamics simulation and MM-PBSA approaches.[Pubmed:33183178]
J Biomol Struct Dyn. 2022 Apr;40(7):3296-3311.
The recently emerged COVID-19 has been declared a pandemic by the World Health Organization as to date; no therapeutic drug/vaccine is available for the treatment. Due to the lack of time and the urgency to contain the pandemic, computational screening appears to be the best tool to find a therapeutic solution. Accumulated evidence suggests that many phyto-compounds possess anti-viral activity. Therefore, we identified possible phyto-compounds that could be developed and used for COVID-19 treatment. In particular, molecular docking was used to prioritize the possible active phyto-compounds against two key targets namely RNA dependent RNA polymerase (RdRp) and main protease (M(pro)) of SARS-CoV-2. In this study, an antiviral drug- Remdesivir (RdRp inhibitor) and Darunavir (M(pro) inhibitor) are used as reference drugs. This study revealed that phyto-molecules- Mulberroside-A/C/E/F, Emblicanin A, Nimbolide, and Punigluconin showed high binding affinity against RdRp while Andrographolides, Mulberrosides, Anolignans, Chebulic acid, Mimusopic acid, and Punigluconin showed better binding affinity against M(pro) as compared with the reference drug. Furthermore, ADME profiles validated the drug-likeness properties of prioritized phyto-compounds. Besides, to assess the stability, MD simulations studies were performed along with reference inhibitors for M(pro) (Darunavir) and RdRp (Remdesivir). Binding free energy calculations (MM-PBSA) revealed the estimated value (DeltaG) of M(pro)_Darunavir; M(pro)_Mulberroside E; RdRp_Remdesivir and RdRp_Emblicanin A were -111.62 +/- 6.788, -141.443 +/- 9.313, 30.782 +/- 5.85 and -89.424 +/- 3.130 kJmol(-1), respectively. Taken together, the study revealed the potential of these phyto-compounds as inhibitors of RdRp and M(pro) inhibitor that could be further validated against SARS-CoV-2 for clinical benefits.Communicated by Ramaswamy H. Sarma.
Inhibition of xanthine and monoamine oxidases by stilbenoids from Veratrum taliense.[Pubmed:11301865]
Planta Med. 2001 Mar;67(2):158-61.
The bioassay guided refractionation of the methanol extract of roots and rhizomes of Veratrum taliense (Liliaceae) yielded five stilbenoids: veraphenol, resveratrol, piceid, isorhapontin, and Mulberroside E, all inhibiting xanthine oxidase (XO, EC 1.2.3.2.) in vitro in a dose-dependent manner with IC50 values of 11.0, 96.7, 66.1, 70.0, and 78.4 microM, respectively. Veraphenol and Mulberroside E were found to be mixed XO inhibitors with the Ki and Ki data of the former being 32.8 and 239.3 microM, and those of latter 32.5 and 13.8 microM, respectively. However, the inhibition on the enzyme by resveratrol, isorhapontin, and piceid was shown to be competitive with their Ki values of 9.7, 19.1, and 14.3 microM, respectively. Among the five stilbenoids, veraphenol and resveratrol were also revealed to inhibit competitively monoamine oxidase A (MAO, EC 1.4.3.4) with IC50 values at 38.0 and 26.6 microM, and Ki data 36.4 and 47.3 microM, respectively. However, none of the stilbenoids was inhibitory on MAO B in our assay. The structure-activity relationship examination showed that glycosylation of the stilbenoids could reduce the inhibition on XO and diminish the activity against MAO A, indicating that the free phenolic hydroxy group of the compounds was most likely essential for these bioactivities.


