LY 334370 hydrochlorideSelective 5-HT1F agonist CAS# 199673-74-0 |
- WYE-354
Catalog No.:BCC1059
CAS No.:1062169-56-5
- GDC-mTOR inhibitor
Catalog No.:BCC1781
CAS No.:1207358-59-5
- GDC-0349
Catalog No.:BCC1094
CAS No.:1207360-89-1
- QL-IX-55
Catalog No.:BCC1876
CAS No.:1223002-54-7
- Nordihydroguaiaretic acid
Catalog No.:BCC1805
CAS No.:500-38-9
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 199673-74-0 | SDF | Download SDF |
| PubChem ID | 19710026 | Appearance | Powder |
| Formula | C21H23ClFN3O | M.Wt | 387.88 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 25 mM in water with gentle warming and to 25 mM in DMSO with gentle warming | ||
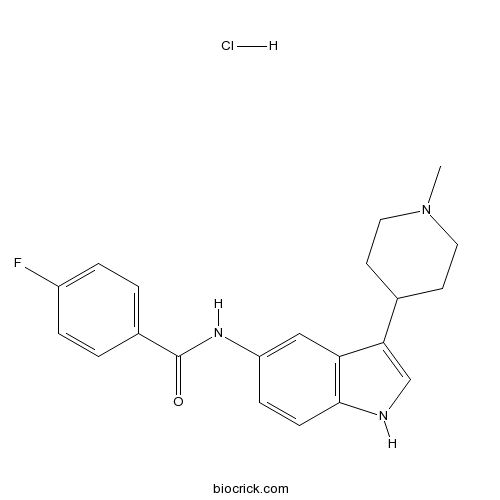
| Chemical Name | 4-fluoro-N-[3-(1-methylpiperidin-4-yl)-1H-indol-5-yl]benzamide;hydrochloride | ||
| SMILES | CN1CCC(CC1)C2=CNC3=C2C=C(C=C3)NC(=O)C4=CC=C(C=C4)F.Cl | ||
| Standard InChIKey | DGPDGAPZTPSHBL-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C21H22FN3O.ClH/c1-25-10-8-14(9-11-25)19-13-23-20-7-6-17(12-18(19)20)24-21(26)15-2-4-16(22)5-3-15;/h2-7,12-14,23H,8-11H2,1H3,(H,24,26);1H | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Selective 5-HT1F receptor agonist (Ki values are 1.87, 16.4, > 100 (IC50), 176, 189, 281, 1280, 1530, 1550, > 3000 (IC50) and 3250 nM for 5-HT1F, 5-HT1A, 5-HT4, 5-HT1E, 5-HT1B, 5-HT1D, 5-HT2B, 5-HT2A, 5-HT7, 5-HT6 and 5-HT2C receptors respectively). Displays antimigraine effects. |

LY 334370 hydrochloride Dilution Calculator

LY 334370 hydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5781 mL | 12.8906 mL | 25.7812 mL | 51.5623 mL | 64.4529 mL |
| 5 mM | 0.5156 mL | 2.5781 mL | 5.1562 mL | 10.3125 mL | 12.8906 mL |
| 10 mM | 0.2578 mL | 1.2891 mL | 2.5781 mL | 5.1562 mL | 6.4453 mL |
| 50 mM | 0.0516 mL | 0.2578 mL | 0.5156 mL | 1.0312 mL | 1.2891 mL |
| 100 mM | 0.0258 mL | 0.1289 mL | 0.2578 mL | 0.5156 mL | 0.6445 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Ro 61-8048
Catalog No.:BCC7619
CAS No.:199666-03-0
- CP 465022 hydrochloride
Catalog No.:BCC7520
CAS No.:199655-36-2
- JIB-04
Catalog No.:BCC4548
CAS No.:199596-05-9
- Methyllucidone
Catalog No.:BCN4877
CAS No.:19956-54-8
- Lucidone
Catalog No.:BCN4876
CAS No.:19956-53-7
- NNC 26-9100
Catalog No.:BCC7361
CAS No.:199522-35-5
- 2-Amino-4-chlorobenzothiazole
Catalog No.:BCC8529
CAS No.:19952-47-7
- Veraguensin
Catalog No.:BCN2163
CAS No.:19950-55-1
- Y-33075
Catalog No.:BCC2064
CAS No.:199433-58-4
- 3-Epicabraleadiol
Catalog No.:BCN4875
CAS No.:19942-04-2
- Gymnestrogenin
Catalog No.:BCN7846
CAS No.:19942-02-0
- Kolavenol
Catalog No.:BCN4680
CAS No.:19941-83-4
- Liguiritigenin-7-O-D-apiosyl-4'-O-D-glucoside
Catalog No.:BCN2840
CAS No.:199796-12-8
- 1-Actamido-3,5-dimethyladmantane
Catalog No.:BCC8449
CAS No.:19982-07-1
- Stattic
Catalog No.:BCC1176
CAS No.:19983-44-9
- PD 166793
Catalog No.:BCC2376
CAS No.:199850-67-4
- Isocudraniaxanthone B
Catalog No.:BCN6887
CAS No.:199851-52-0
- RS 127445
Catalog No.:BCC1909
CAS No.:199864-87-4
- DL-Alanyl-DL-Methionine
Catalog No.:BCC8950
CAS No.:1999-43-5
- Chrysoeriol-7-O-glucoside
Catalog No.:BCN3796
CAS No.:19993-32-9
- UCL 1684
Catalog No.:BCC7016
CAS No.:199934-16-2
- CVT-313
Catalog No.:BCC1503
CAS No.:199986-75-9
- ALX 5407 hydrochloride
Catalog No.:BCC7168
CAS No.:200006-08-2
- Benzyl 4-bromophenyl ketone
Catalog No.:BCC8868
CAS No.:2001-29-8
[Phase I study of gemcitabine hydrochloride (LY 188011) combination therapy with cisplatin in the patients with non-small cell lung cancer].[Pubmed:10396316]
Gan To Kagaku Ryoho. 1999 Jun;26(7):898-907.
The combination Phase I study of gemcitabine hydrochloride with cisplatin was conducted in the patients with non-small cell lung cancer (NSCLC) at 5 investigation sites. Gemcitabine was administrated on day 1, 8 and 15 and cisplatin on day 1 of each 28-day cycle. The dosage of cisplatin was fixed to 80 mg/m2 and the dosage of Gemcitabine was gradually escalated in 3 dosing level from 600, 800 to 1,000 mg/m2. The maximum tolerated dose (MTD) and the recommended dose was determined with Continual Reassessment Method. For each dose level, 6 cases, 3 cases and 6 cases were registered respectively and all 15 cases were evaluable. In the dose level 3 with 1,000 mg/m2 of gemcitabine and 80 mg/m2 of cisplatin, grade 4 neutropenia was observed as DLT in 3 out of 6 cases, thus dose level 3 was considered as MTD and the recommended dose. Major adverse events were leukopenia, neutropenia, nausea/vomiting and anorexia. The incidence of such adverse events seemed to be dose-dependent and especially the grade of neutropenia seemed to be more serious as the dose increased. Also, the grade of liver function tests abnormal seemed to be more serious as the dose increased but the incidence as well as the grade did not have tendency of dose-dependent in another events including renal function tests abnormal. On the other hand, as to the efficacy PR was observed in 4 out of 15 cases. Based upon the results, it is necessary to discuss further the efficacy in the recommended dose in the combination therapy of gemcitabine and cisplatin.
[An early phase II study of gemcitabine hydrochloride (LY 188011). Gemcitabine Cooperative Study Group for Early Phase II].[Pubmed:8937492]
Gan To Kagaku Ryoho. 1996 Nov;23(13):1813-24.
An early phase II cooperative study of Gemcitabine Hydrochloride (abbreviated to "gemcitabine" herewith) was conducted in patients with a variety of solid tumors (i.e., lung cancer, gastric cancer, pancreatic cancer, colon/rectum cancer, cervical cancer, ovarian cancer and breast cancer) at 56 institutions. The aim of the first step (Step I) was to investigate the feasibility of gemcitabine in a variety of different solid tumors, including lung cancer regarding efficacy and safety. The aim of the second step (Step II) was as a result of step I (Responses were observed) to continue to investigate the efficacy and safety of gemcitabine in chemonaive patients with non-small cell lung cancer. As a Step I study, gemcitabine was administered once weekly at a dose of 800 mg/m2 for a consecutive 3-week period followed by a week of rest, in multiple courses. Among the 29 eligible patients with lung cancer, partial response (PR) was achieved in 3 patients (25.0%, 95% confidence interval: 5.5-57.2%) out of 12 chemonaive patients. Adverse reactions (grade 3 or higher) seen in 29 patients with lung cancer were neutropenia (27.6%), leukopenia (13.8%), decreased hemoglobin (13.8%), thrombocytopenia (10.3%), malaise (6.9%), anorexia (3.4%), nausea/vomiting (3.4%), diarrhea (3.4%), dyspnea (3.4%) and interstitial pneumonia (3.4%). In other types of solid tumors, PR was achieved in 2 (8.7%) out of 23 eligible patients with cervical cancer and in 1 (5.3%) of 19 eligible patients with ovarian cancer, while the use of analgesics became unnecessary in 1 patient with pancreatic cancer. Incidence as well as severity of main adverse reactions in these patients were comparable to those seen in patients with lung cancer. A Step II study, in which gemcitabine was administered once weekly at a dose of 1,000 mg/m2 to chemonaive patients with non-small cell lung cancer, was conducted, referring to the results of Step I and clinical studies conducted overseas. The results of the Step II study demonstrated PR in 5 (14.3%, 95% confidence interval: 4.8 - 30.3%) out of 35 eligible patients with non-small cell lung cancer and that the main adverse reactions were comparable to those seen in the Step I study, posing no tolerability problems in particular.
Changes in motor activities induced by microinjections of the selective dopamine agonists LY 171555, quinpirole hydrochloride, and SK&F 38393 into the habenula nucleus.[Pubmed:3495009]
Pharmacol Biochem Behav. 1987 Mar;26(3):643-6.
The effects on behaviour of microinjections into the habenula complex of selective agonists for dopamine D-1 (SK&F 38393) and D-2 (LY 171555) receptors were documented in a holeboard, open-field test. The D-2 agonist reduced grooming responses, locomotor activity and rearing behaviour. In contrast, the D-1 agonist increased rearing and locomotor activity but was without effect on grooming responses. Neither drug produced significant effects on inspective hole exploration. The data extend findings of behavioural consequences of central D-1 receptor activation and provide direct evidence in support of the functional and behavioural importance of intrahabenular dopamine receptor sites. The findings are consistent with suggestions for feedback regulation of habenular efferents to midbrain dopaminergic neurons. Effects of both receptor agonists on some responses but not others indicates potential complex interactions between D-1 and D-2 receptors within the habenula.
Possible antimigraine mechanisms of action of the 5HT1F receptor agonist LY334370.[Pubmed:10668103]
Cephalalgia. 1999 Dec;19(10):851-8.
This study investigated whether the selective 5HT1F receptor agonist LY334370 has other possible antimigraine mechanisms in addition to the proposed inhibition of dural plasma extravasation. LY334370 (up to 10(-5) M) had no vasoconstrictor effects on human cerebral arteries in vitro. It had no effect (up to 10 mg kg-1, i.v.) on neurogenic vasodilation of dural blood vessels produced by electrical stimulation of the dura mater in anesthetized rats. Nor had it any effect (at 3 mg kg-1, i.v.) on the hyperalgesia produced by injection of carrageenan into the paw of conscious rats or on nociceptive reflex responses in the spinalized, decerebrate rabbit (up to 3 mg kg-1, i.v.), indicating that it has no general analgesic properties. However, it significantly inhibited activation of second-order neurons in the trigeminal nucleus caudalis produced by electrical stimulation of the dura mater in anesthetised rats at 3 mg kg-1, i.v. These results provide evidence to suggest that LY334370 has a central mechanism of action in blocking the transmission of nociceptive impulses within the trigeminal nucleus caudalis and that this may represent a mechanism through which it has its antimigraine effect.
G-protein activation at 5-HT1A receptors by the 5-ht1F ligand LY334370 in guinea-pig brain sections and recombinant cell lines.[Pubmed:9641544]
Br J Pharmacol. 1998 May;124(2):283-90.
1. G-protein activation by the 5-ht1F receptor agonist 5-(4-fluorobenzoyl)amino-3-(1-methylpiperidin-4-yl)-1H-indole fumarate (LY334370) was investigated by use of autoradiography of receptor-activated G-proteins in guinea-pig brain sections and [35S]-GTPgammaS binding responses in cell lines stably expressing human 5-HT1A (h 5-HT1A) receptors. 2. LY334370 (10 microM) caused little or no stimulation of [35S]-GTPgammaS binding in guinea-pig brain regions enriched in 5-ht1F binding sites (e.g., claustrum, caudate/putamen and thalamic nuclei), as identified by labelling with 10 nM [3H]-sumatriptan plus 10 nM 5-carboxamidotryptamine (5-CT). 3. Application of LY334370 (10 microM) to guinea-pig brain sections resulted in an increase of [35S]-GTPgammaS binding in hippocampus (123+/-17%), lateral septum (58+/-14%), dorsal raphe (57+/-10%), entorhinal (37+/-11%) and cingulate cortex (28+/-10%). This distribution fits with the G-protein activation mediated by 5-HT1A receptors as found with lisuride (10 microM), and labelling of 5-HT1A receptors by 140 pM [125I]-4-(2'-methoxy-phenyl)- -[2'-(n-2"-pyridinyl)-p-iodobenzamido]-ethyl-piperazine (p-MPPI). 4. The LY334370-mediated [35S]-GTPgammaS response was antagonized by the selective, silent 5-HT1A receptor antagonist N-[2-[4-(2-methoxyphenyl)1-piperazinyl]ethyl]-N-(2-pyridinyl)cyclohex anecarboxa-mide (WAY100635, 1 microM) in each of the brain structures investigated. The distribution pattern of the [35S]-GTPgammaS binding response and the antagonist profile suggest that the LY334370-induced response in guinea-pig brain is mediated by 5-HT1A receptors. 5. The maximal LY334370-induced [35S]-GTPgammaS binding response (83 to 94%) in membranes of recombinant C6-glial/h 5-HT1A and HeLa/h 5-HT1A cells was close to that of 5-HT, suggesting LY334370 to exert high intrinsic activity at h 5-HT1A receptors. 6. In conclusion, in guinea-pig brain sections and recombinant cell lines the 5-ht1F receptor agonist LY334370 causes G-protein activation that is mediated by 5-HT1A receptors. Caution should be taken when employing this ligand as a putative selective 5-ht1F agonist.


