4-MethoxyglucobrassicinCAS# 83327-21-3 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 83327-21-3 | SDF | Download SDF |
| PubChem ID | 49859627 | Appearance | Powder |
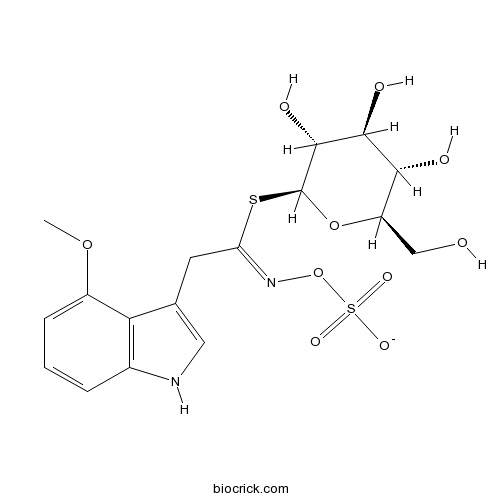
| Formula | C17H21KN2O10S2 | M.Wt | 516.6 |
| Type of Compound | Miscellaneous | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | [(Z)-[2-(4-methoxy-1H-indol-3-yl)-1-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanylethylidene]amino] sulfate | ||
| SMILES | COC1=CC=CC2=C1C(=CN2)CC(=NOS(=O)(=O)[O-])SC3C(C(C(C(O3)CO)O)O)O | ||
| Standard InChIKey | IIAGSABLXRZUSE-KYKLFQSUSA-M | ||
| Standard InChI | InChI=1S/C17H22N2O10S2/c1-27-10-4-2-3-9-13(10)8(6-18-9)5-12(19-29-31(24,25)26)30-17-16(23)15(22)14(21)11(7-20)28-17/h2-4,6,11,14-18,20-23H,5,7H2,1H3,(H,24,25,26)/p-1/b19-12-/t11-,14-,15+,16-,17+/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

4-Methoxyglucobrassicin Dilution Calculator

4-Methoxyglucobrassicin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.9357 mL | 9.6787 mL | 19.3573 mL | 38.7147 mL | 48.3933 mL |
| 5 mM | 0.3871 mL | 1.9357 mL | 3.8715 mL | 7.7429 mL | 9.6787 mL |
| 10 mM | 0.1936 mL | 0.9679 mL | 1.9357 mL | 3.8715 mL | 4.8393 mL |
| 50 mM | 0.0387 mL | 0.1936 mL | 0.3871 mL | 0.7743 mL | 0.9679 mL |
| 100 mM | 0.0194 mL | 0.0968 mL | 0.1936 mL | 0.3871 mL | 0.4839 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Glucoarabin
Catalog No.:BCN8965
CAS No.:67920-64-3
- Glucocamelinin
Catalog No.:BCN8964
CAS No.:67884-10-0
- Glucoraphasatin potassium salt
Catalog No.:BCN8963
CAS No.:245550-64-5
- Glucohirsutin
Catalog No.:BCN8962
CAS No.:21973-60-4
- Gluconasturtiin
Catalog No.:BCN8961
CAS No.:18425-76-8
- Sinalbin potassium salt
Catalog No.:BCN8960
CAS No.:16411-05-5
- Glucocheirolin
Catalog No.:BCN8959
CAS No.:15592-36-6
- Glucobrassicin
Catalog No.:BCN8958
CAS No.:143231-38-3
- Glucoraphasatin
Catalog No.:BCN8957
CAS No.:28463-23-2
- Noratropine
Catalog No.:BCN8955
CAS No.:16839-98-8
- Scopolamine N-oxide hydrobromide
Catalog No.:BCN8953
CAS No.:6106-81-6
- Usaramine N-oxide
Catalog No.:BCN8952
CAS No.:117020-54-9
- 4-Hydroxyglucobrassicin
Catalog No.:BCN8967
CAS No.:83327-20-2
- Epiprogoitrin
Catalog No.:BCN8968
CAS No.:21087-74-1
- Phyllalbine
Catalog No.:BCN8969
CAS No.:4540-25-4
- Glucobarbarin
Catalog No.:BCN8970
CAS No.:21087-78-5
- Glucolimnanthin
Catalog No.:BCN8971
CAS No.:111810-95-8
- Glucocapparin
Catalog No.:BCN8972
CAS No.:15592-33-3
- Sinalbin
Catalog No.:BCN8973
CAS No.:20196-67-2
- Glucobrassicanapin
Catalog No.:BCN8974
CAS No.:245550-58-7
- Glucoiberin
Catalog No.:BCN8975
CAS No.:15592-34-4
- Glucoberteroin
Catalog No.:BCN8976
CAS No.:245550-65-6
- Glucoerucin
Catalog No.:BCN8977
CAS No.:15592-37-7
- Glucotropaeolin
Catalog No.:BCN8978
CAS No.:5115-71-9
Profiling of Individual Desulfo-Glucosinolate Content in Cabbage Head (Brassica oleracea var. capitata) Germplasm.[Pubmed:32316621]
Molecules. 2020 Apr 17;25(8). pii: molecules25081860.
Individual glucosinolates (GSLs) were assessed to select cabbage genotypes for a potential breeding program. One hundred forty-six cabbage genotypes from different origins were grown in an open field from March to June 2019; the cabbage heads were used for GSL analyses. Seven aliphatics [glucoiberin (GIB), progoitrin (PRO), epi-progoitrin (EPI), sinigrin (SIN), glucoraphanin (GRA), glucoerucin (GER) and gluconapin (GNA)], one aromatic [gluconasturtiin (GNS)] and four indolyl GSLs [glucobrassicin (GBS), 4-hydroxyglucobrassicin (4HGBS), 4-Methoxyglucobrassicin (4MGBS), neoglucobrassicin (NGBS)] were found this study. Significant variation was observed in the individual GSL content and in each class of GSLs among the cabbage genotypes. Aliphatic GSLs were predominant (58.5%) among the total GSLs, followed by indolyl GSL (40.7%) and aromatic GSLs (0.8%), showing 46.4, 51.2 and 137.8% coefficients of variation, respectively. GIB, GBS and NGBS were the most common GSLs found in all genotypes. GBS was the most dominant GSL, with an average value of 3.91 micromol g(-1) (0.79 to 13.14 micromol g(-1)). SIN, GIB, PRO and GRA were the other major GSLs, showing average values of 3.45, 1.50, 0.77 and 0.62 micromol g(-1), respectively. The genotypes with relatively high contents of GBS, SIN, GIB and GRA warrant detailed studies for future breeding programs since the hydrolysis products of these GSLs have several anti-cancer properties.
Use of elicitation in the cultivation of Bimi(R) for food and ingredients.[Pubmed:31875967]
J Sci Food Agric. 2020 Mar 30;100(5):2099-2109.
BACKGROUND: Cruciferous foods rich in health-promoting metabolites are of particular interest to consumers as well as being a good source of bioactives-enriched ingredients. Several elicitors have been used to stimulate the biosynthesis and accumulation of secondary metabolites in foods; however, little is known about the response of new hybrid varieties, such as Bimi(R), under field-crop production conditions. Therefore, this study was designed to evaluate the effect of salicylic acid (200 mumol L(-1) , SA), methyl jasmonate (100 mumol L(-1) , MeJA), and their combination on Bimi plant organs (inflorescences and aerial vegetative tissues - stems and leaves). For this, the composition of the glucosinolates present in the tissues was evaluated. Also, aqueous extracts of the plant material, obtained with different times of extraction with boiling water, were studied. RESULTS: The results indicate that the combined treatment (SA + MeJA) significantly increased the content of glucosinolates in the inflorescences and that MeJA was the most effective elicitor in leaves. Regarding the aqueous extracts, the greatest amount of glucosinolates was extracted at 30 min - except for the leaves elicited with MeJA, for which 15 min was optimal. CONCLUSION: The elicitation in the field enriched leaves in glucobrassicin (GB), 4-Methoxyglucobrassicin (MGB), and neoglucobrassicin (NGB) and stems and inflorescences in glucoraphanin, 4-hydroxyglucobrassicin, GB, MGB, and NGB. In this way, this enhanced vegetable material favored the presence of bioactives in the extracts, which is of great interest regarding enriched foods and ingredients with added value obtained from them. (c) 2019 Society of Chemical Industry.
Effect of pretreatment on bioactive compounds in wild rocket juice.[Pubmed:31749470]
J Food Sci Technol. 2019 Dec;56(12):5234-5242.
The aim of the study was to determine the effect of pretreatment with hot water or steaming on glucosinolates, polyphenols contents and antioxidant capacity in obtained raw juices. Moreover, in vitro cytotoxic activity of the raw juice to the cells derived from the gastrointestinal tract, including the small intestine (IEC-6 cell line), colon (Caco-2 cell line) and the liver (HepG2 cell line) were also investigated. The dominant glucosinolates in the wild rocket leaves were glucoraphanin (36%) and dimeric 4-mercaptobutyl (30%), followed by glucosativin and glucoerucin, 11% per each. Glucothiobeinin (6%), glucobrassicin (1%), 4-Methoxyglucobrassicin (1%) and two unidentified compounds (4%) were also detected in rocket leaves. In terms of phenolic compounds, quercetin constituted the majority (55%) and the rest composed of hydroxycinnamic acids. In raw juices produced from steamed, pretreatment with hot water and untreated (control) leaves, glucosinolate contents were lower about 21%, 37% and 53%, respectively, than their levels in the raw material. The highest content of polyphenols among the juices tested (45.4 mg/100 g fresh weight) and antioxidant capacity (5.8 micromol Trolox/1 g f.w.) was recorded in the raw juice from pretreated leaves with hot water. The wild rocket raw juice concentrations responsible for a 50% reduction in Caco-2 and HepG2 cell viability were estimated at 1.87 +/- 0.08 mg/mL and 3.54 +/- 0.29 mg/mL. The viability of the IEC-6 cells was reduced by only 19.04%, at the maximum concentration (3.6 mg/mL) of the raw juice.
Comparative analysis of glucosinolate production in hairy roots of green and red kale (Brassica oleracea var. acephala).[Pubmed:31124740]
Prep Biochem Biotechnol. 2019;49(8):775-782.
Glucosinolates (GSLs) are sulfur- and nitrogen-containing secondary metabolites that function in plant defense and provide benefits to human health. In this study, using Agrobacterium rhizogenes R1000, green and red kale hairy roots were established. The expression levels of GSLs biosynthesis genes and their accumulation in both kale hairy roots were analyzed by quantitative real-time PCR and HPLC. The results showed that the expression of most indolic GSLs biosynthesis genes was higher in the hairy roots of green kale than in that of red kale. In contrast, the expression of BoCYP83A1 and BoSUR1 encoding key enzymes aromatic GSL biosynthesis was significantly higher in red kale hairy root. The HPLC analysis identified six GSLs. The levels of 4-Methoxyglucobrassicin, glucobrassicin, and 4-hydroxyglucobrassicin were 6.21, 5.98, and 2 times higher, respectively, in green kale than in red kale, whereas the levels of neoglucobrassicin and gluconasturtiin were 16.2 and 3.48 times higher, respectively, in red kale than in green kale. Our study provides insights into the underlying mechanisms of GSLs biosynthesis in kale hairy roots and can be potentially used as "biological factories" for producing bioactive substances such as GSLs.
Antimicrobial and Cytotoxic Activities of Lepidium latifolium L. Hydrodistillate, Extract and Its Major Sulfur Volatile Allyl Isothiocyanate.[Pubmed:30714673]
Chem Biodivers. 2019 Apr;16(4):e1800661.
The cultivated Lepidium latifolium L. was investigated to decipher its glucosinolate profile, antimicrobial, and cytotoxic activities. HPLC/ESI-MS analyses of the intact glucosinolates and GC/MS analysis of their hydrolysis products showed the presence of sinigrin (1), glucocochlearin (2), glucotropaeolin (3), and 4-Methoxyglucobrassicin (4). Hydrodistillate, extract, and allyl isothiocyanate, the main volatile resulting from sinigrin degradation, showed antimicrobial activity against all eleven tested pathogenic and food spoilage bacteria and fungi, with highest effect observed against Candida albicans with MIC50 8 and 16 mug/mL. Hydrodistillate and extract showed the best cytotoxic activity on bladder cancer UM-UC-3 cell line during an incubation time of 24 h (IC50 192.9 and 133.8 mug/mL, respectively), while the best effect on glioblastoma LN229 cell line was observed after 48 h (IC50 110.8 and 30.9 mug/mL, respectively). Pure allyl isothiocyanate displayed a similar trend in cytotoxic effect on both cell lines (IC50 23.3 and 36.5 mug/mL after 24 h and 48 h, respectively).
Comparative analysis of glucosinolates and metabolite profiling of green and red mustard (brassica juncea) hairy roots.[Pubmed:30148032]
3 Biotech. 2018 Sep;8(9):382.
Here, accumulation of glucosinolates and expression of glucosinolates biosynthesis genes in green and red mustard hairy roots were identified and quantified by HPLC and qRT-PCR analyses. The total glucosinolates content of green mustard hairy root (10.09 microg/g dry weight) was 3.88 times higher than that of red mustard hairy root. Indolic glucosinolates (glucobrassicin, 4-Methoxyglucobrassicin, and neoglucobrassicin) in green mustard were found at 30.92, 6.95, and 5.29 times higher than in red mustard hairy root, respectively. Conversely, levels of glucotropaeolin (aromatic glucosinolate) was significantly higher in red mustard than in green mustard. Accumulation of glucoraphasatin, an aliphatic glucosinolate, was only observed only in red mustard hairy roots. Quantitative real-time PCR analysis showed that the expression level of genes related to aliphatic and aromatic glucosinolate biosynthesis were higher in red mustard, exception BjCYP83B. The expression of BjCYP79B2, which encodes a key enzyme involved in the indolic glucosinolate biosynthetic pathway, was higher in green mustard than in red mustard. Additionally, to further distinguish between green mustard and red mustard hairy roots, hydrophilic and lipophilic compounds were identified by gas chromatography-mass spectrometry and subjected to principal component analysis. The results indicated that core primary metabolites and glucosinolate levels were higher in the hairy roots of green mustard than in those of red mustard.
Influence of silver nanoparticles on the enhancement and transcriptional changes of glucosinolates and phenolic compounds in genetically transformed root cultures of Brassica rapa ssp. rapa.[Pubmed:30056602]
Bioprocess Biosyst Eng. 2018 Nov;41(11):1665-1677.
Glucosinolates (GSLs) and phenolic compounds (PCs) are biologically active and involved in the defense reaction of plants; these compounds have a beneficial effect on human health. In this study, we described the influence of biologically synthesized silver nanoparticles (Ag NPs) to enhance the phytochemicals (GSLs and PCs), their transcription levels, and their biological activities in genetically transformed root cultures (hairy root cultures) of Brassica rapa. The concentrations of silver and reactive oxygen species (malondialdehyde and hydrogen peroxide) were highly elevated in the Ag NP-elicited hairy roots (HRs). Glucosinolates (glucoallysin, glucobrassicanapin, sinigrin, progoitrin, gluconapin, 4-Methoxyglucobrassicin, 4-hydroxyglucobrassicin, glucobrassicin, neoglucobrassicin, and gluconasturtiin) and their transcripts (MYB34, MYB51, MYB28, and MYB29) were significantly enhanced in the Ag NP-elicited HRs. Moreover, the phenolic compounds (flavonols, hydroxybenzoic, and hydroxycinnamic acids) were significantly enriched in the Ag NP-elicited HRs. Total phenolic and flavonoid concentrations and their transcripts (PAL, CHI, and FLS) were higher in the Ag NP-elicited HRs than in the non-elicited HRs. Additionally, biological (antioxidant, antimicrobial, and anticancer) activities were significantly higher in the Ag NP-elicited HRs than in the non-elicited HRs. The Ag NP-elicited HR cultures offered an efficient and promising in vitro method to increase the production of health-promoting bioactive compounds, which may be useful in nutraceutical and pharmaceutical industries.
Effect of different proportion of sulphur treatments on the contents of glucosinolate in kale (Brassica oleracea var. acephala) commonly consumed in Republic of Korea.[Pubmed:29472789]
Saudi J Biol Sci. 2018 Feb;25(2):349-353.
Kale (Brassica oleracea L. Acephala Group) is the rich source of medicinal value sulphur compounds, glucosinolates (GLSs). The aim of this study was to investigate the effect of different proportion of sulphur (S) supplementation levels on the accumulation of GLSs in the leaves of the kale cultivar ('TBC'). High performance liquid chromatography (HPLC) separation method guided to identify and quantify six GSLs including three aliphatic (progoitrin, sinigrin and gluconapin) and three indolyl (glucobrassicin, 4-Methoxyglucobrassicin and neoglucobrasscin) respectively. Analysis of these distinct levels of S supplementation revealed that the accumulation of individual and total GLSs was directly proportional to the S concentration. The maximum levels of total GLSs (26.8 micromol/g DW) and glucobrassicin (9.98 micromol/g DW) were found in lower and upper parts of the leaves supplemented with 1 mM and 2 mM S, respectively. Interestingly, aliphatic GSLs were noted predominant in all the parts (50.1, 59.3 and 56% of total GSLs). Among the aliphatic and indolyl GSLs, sinigrin and glucobrassicin account 35.3 and 30.88% of the total GSLs. From this study, it is concluded that supply of S enhance the GSLs accumulation in kale.
Molecular characterization of glucosinolates and carotenoid biosynthetic genes in Chinese cabbage (Brassica rapa L. ssp. pekinensis).[Pubmed:29379360]
Saudi J Biol Sci. 2018 Jan;25(1):71-82.
The present study aimed to investigate the contents of glucosinolates (GSLs) and carotenoids in eleven varieties of Chinese cabbage in relation to the expression level of the important transcription factors. MS and HPLC analysis identified the presence of 13 GSLs (progoitrin, sinigrin, glucoalyssin, gluconapoleiferin, gluconapin, glucocochlearin, glucobrassicanapin, glucoerucin, 4-hydroxyglucobrassicin, glucobrassicin, 4-Methoxyglucobrassicin, neoglucobrassicin and gluconasturtiin) and four carotenoids (lutein, zeaxanthin, alpha-carotene and beta-carotene). GSL contents were varied among the different cabbage varieties. The total GSL content ranged from 2.7 to 57.88 mumol/g DW. The proportion of gluconapin (54%) and glucobrassicanapin (22%) was higher in all the varieties, respectively. Results documented the variation in total and individual carotenoid contents that have also been observed among different varieties; however, the total carotenoid contents ranged from 289.12 to 1001.41 mg kg(-1) DW (mean 467.66). Interestingly, the proportion of lutein (66.5) and beta-carotene (25.9) were higher than alpha-carotene (5.1) and zeaxanthin (2.5%). Consequently, the expression level of the regulatory gene, MYB28 was higher in 'K0648' and was directly proportional to GSL content. Similarly, the expression levels of 1-PSY were higher in 'K0112'; however, the expression levels of 2-ZDS, 3-LCYB, 4-LCYE, 5-CHXB and 7-NCED genes showed no significant difference. In addition, the correlation between GSL and carotenoid contents and gene expression level showed moderate significant difference in each Chinese cabbage.
Synthesis of aromatic and indole alpha-glucosinolates.[Pubmed:29169042]
Carbohydr Res. 2018 Jan 2;455:45-53.
Aromatic and indole glucosinolates are important members of the glucosinolate family of compounds du to their potential medicinal properties. They are known to exert antioxidant and anti-carcinogenic activity either by the natural products themselves, or their metabolic products including indole-3-carbinol and isothiocyanates. Natural glucosinolates are all beta-glucosinolates; however, alpha-glucosinolates are also promising compounds for medicinal applications and hence have to be produced synthetically for any bio-activity studies. Here we report on the successful synthesis of a series of alpha-glucosinolates: alpha-neoglucobrassicin, alpha-4-Methoxyglucobrassicin, 2,3-dichlorophenyl-alpha-glucosinolate for the first time. Testing for anti-inflammatory properties of these synthetic GLs, however, did not yield the expected activity.
Rapid Separation of Indole Glucosinolates in Roots of Chinese Cabbage (Brassica rapa Subsp. Pekinensis) by High-Performance Liquid Chromatography with Diode Array Detection.[Pubmed:28694825]
Int J Anal Chem. 2017;2017:5125329.
Glucosinolates are a class of sulphur-containing plant compounds with diverse biological properties. They have been found exclusively in the Brassicaceae family plants and studied exhaustively in biosynthetic and application perspectives. The aim of this current study is to present a simple and updated method to quantify indole glucosinolate content in hairy root cultures of Chinese cabbage by HPLC-DAD-UV/Vis. Method validation controls were performed and recovery experiments were assayed. The data was statically treated and compared with published works. The current method allowed a feasible identification of indole glucosinolates and it was possible to identify accurately three indole glucosinolate compounds (glucobrassicin, 4-Methoxyglucobrassicin, and 1-methoxyglucobrassicin) in roots of Chinese cabbage. The method demonstrated a good linearity (R(2) > 0.99), a good precision, and selectivity sensitivity. In conclusion, this protocol provides an accessible method to extract and quantify glucosinolates in plant samples. Thus, based on our results, the method is valid and can be extended to other plant or food matrices.
Development of a near-infrared spectroscopy method (NIRS) for fast analysis of total, indolic, aliphatic and individual glucosinolates in new bred open pollinating genotypes of broccoli (Brassica oleracea convar. botrytis var. italica).[Pubmed:28490075]
Food Chem. 2017 Oct 1;232:272-277.
This study describes the development of near-infrared spectroscopy (NIRS) calibration to determine individual and total glucosinolates (GSLs) content of 12 new-bred open-pollinating genotypes of broccoli (Brassica oleracea convar. botrytis var. italica). Six individual GSLs were identified using high-performance-liquid chromatography (HPLC). The NIRS calibration was established based on modified partial least squares regression with reference values of HPLC. The calibration was analyzed using coefficient of determination in prediction (R(2)) and ratio of preference of determination (RPD). Large variation occurred in the calibrations, R(2) and RPD due to the variability of the samples. Derived calibrations for total-GSLs, aliphatic-GSLs, glucoraphanin and 4-Methoxyglucobrassicin were quantitative with a high accuracy (RPD=1.36, 1.65, 1.63, 1.11) while, for indole-GSLs, glucosinigrin, glucoiberin, glucobrassicin and 1-methoxyglucobrassicin were more qualitative (RPD=0.95, 0.62, 0.67, 0.81, 0.56). Overall, the results indicated NIRS has a good potential to determine different GSLs in a large sample pool of broccoli quantitatively and qualitatively.
Preferentially enhancing anti-cancer isothiocyanates over glucosinolates in broccoli sprouts: How NaCl and salicylic acid affect their formation.[Pubmed:28419960]
Plant Physiol Biochem. 2017 Jun;115:343-353.
Broccoli (Brassica oleracea L. var. italica) sprouts contain glucosinolates (GLs) that when hydrolysed yield health promoting isothiocyanates such as sulforaphane (SF). SF content can be increased by salt (NaCl) stress, although high salt concentrations negatively impact plant growth. Salicylic acid (SA) treatments can attenuate the negative effects of salt on growth. To test whether sprout isothiocyanate content could be elevated without sprout growth being compromised, broccoli seed were germinated and grown for seven days in salt (0, 80 and 160 mM) alone and in combination with 100 muM SA. Increasing concentrations of salt lowered transcript accumulation of GL biosynthetic genes which was reflected in lowered content of Gluconapin, 4-Methoxyglucobrassicin and neoglucobrassicin glucosinolates. Other glucosinolates such as glucoraphanin did not alter significantly. Salt (160 mM) increased transcript abundance of the GL hydrolytic gene MYROSINASE (BoMYO) and its cofactor EPITHIOSPECIFIER MODIFIER1 (BoESM1) whose encoded product directs MYROSINASE to produce isothiocyanate rather than nitrile forms. SF content was increased 6-fold by the 160 mM salt treatment, but the salt treatment reduced percentage seed germination, slowed seed germination, and reduced sprout hypocotyl elongation. This growth inhibition was prevented if 100 muM SA was included with the salt treatment. These findings suggest that the increase in SF production by salt occurs in part because of increased transcript abundance of genes in the hydrolytic pathway, which occurs independently of the negative impact of salt on sprout growth.
Identification and Quantification of Glucosinolates in Kimchi by Liquid Chromatography-Electrospray Tandem Mass Spectrometry.[Pubmed:28298926]
Int J Anal Chem. 2017;2017:6753481.
A novel and simple method for detecting five glucosinolates (glucoalyssin, gluconapin, glucobrassicanapin, glucobrassicin, and 4-Methoxyglucobrassicin) in kimchi was developed using liquid chromatography-electrospray tandem mass spectrometry (LC-MS/MS). The chromatographic peaks of the five glucosinolates were successfully identified by comparing their retention times, mass spectra. The mobile phase was composed of A (acetonitrile) and B (water). As for glucosinolate, the relative quantities were found through sinigrin, and five different compounds that have not been previously discovered in kimchi were observed. Monitoring was carried out on the glucosinolate in 20 kimchis distributed in markets, and this study examined the various quality and quantity compositions of the five components. The glucoalyssin content ranged from 0.00 to 7.07 mumol/g of day weight (DW), with an average content of 0.86 mumol/g of DW, whereas the gluconapin content ranged from 0.00 to 5.85 mumol/g of DW, with an average of 1.17 mumol/g of DW. The content of glucobrassicanapin varied between 0.00 and 11.87 mumol/g of DW (average = 3.03 mumol/g of DW), whereas that of glucobrassicin varied between 0.00 and 0.42 mumol/g of DW (average = 0.06 mumol/g of DW). The 4-Methoxyglucobrassicin content ranged from 0.12 to 9.36 mumol/g of DW (average = 3.52 mumol/g of DW). A comparison of the contents revealed that, in most cases, the content of 4-Methoxyglucobrassicin was the highest.


