3-AminoquinolineCAS# 580-17-6 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 580-17-6 | SDF | Download SDF |
| PubChem ID | 11375 | Appearance | Powder |
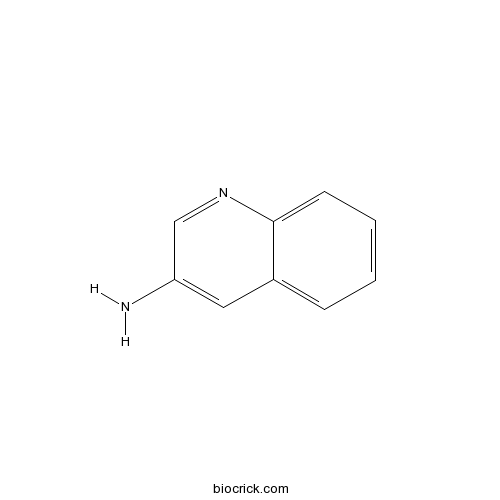
| Formula | C9H8N2 | M.Wt | 144 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | quinolin-3-amine | ||
| SMILES | C1=CC=C2C(=C1)C=C(C=N2)N | ||
| Standard InChIKey | SVNCRRZKBNSMIV-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C9H8N2/c10-8-5-7-3-1-2-4-9(7)11-6-8/h1-6H,10H2 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

3-Aminoquinoline Dilution Calculator

3-Aminoquinoline Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 6.9444 mL | 34.7222 mL | 69.4444 mL | 138.8889 mL | 173.6111 mL |
| 5 mM | 1.3889 mL | 6.9444 mL | 13.8889 mL | 27.7778 mL | 34.7222 mL |
| 10 mM | 0.6944 mL | 3.4722 mL | 6.9444 mL | 13.8889 mL | 17.3611 mL |
| 50 mM | 0.1389 mL | 0.6944 mL | 1.3889 mL | 2.7778 mL | 3.4722 mL |
| 100 mM | 0.0694 mL | 0.3472 mL | 0.6944 mL | 1.3889 mL | 1.7361 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 6-Aminoquinoline
Catalog No.:BCC8766
CAS No.:580-15-4
- Uridine
Catalog No.:BCN4090
CAS No.:58-96-8
- alpha-Tocopherol acetate
Catalog No.:BCN5803
CAS No.:58-95-7
- Chlorothiazide
Catalog No.:BCC3752
CAS No.:58-94-6
- Hydrochlorothiazide
Catalog No.:BCC4786
CAS No.:58-93-5
- D-(+)-Xylose
Catalog No.:BCN1010
CAS No.:58-86-6
- Biotin
Catalog No.:BCC3585
CAS No.:58-85-5
- Papaverine
Catalog No.:BCC8230
CAS No.:58-74-2
- Inosine
Catalog No.:BCN3841
CAS No.:58-63-9
- Adenosine
Catalog No.:BCN5796
CAS No.:58-61-7
- Puromycin aminonucleoside
Catalog No.:BCC1873
CAS No.:58-60-6
- Puromycin dihydrochloride
Catalog No.:BCC7860
CAS No.:58-58-2
- 2-Aminoquinoline
Catalog No.:BCC8555
CAS No.:580-22-3
- Matairesinol
Catalog No.:BCN5789
CAS No.:580-72-3
- Epicorynoxidine
Catalog No.:BCN7554
CAS No.:58000-48-9
- HOKU-81
Catalog No.:BCC1634
CAS No.:58020-43-2
- Averantin
Catalog No.:BCN7027
CAS No.:5803-62-3
- 24, 25-Dihydroxy VD2
Catalog No.:BCC1302
CAS No.:58050-55-8
- Miltefosine
Catalog No.:BCC4360
CAS No.:58066-85-6
- trans-3,4-Methylenedioxycinnamyl alcohol
Catalog No.:BCN1410
CAS No.:58095-76-4
- α-MSH
Catalog No.:BCC7420
CAS No.:581-05-5
- Suberosin
Catalog No.:BCN5791
CAS No.:581-31-7
- Anatabine
Catalog No.:BCN6899
CAS No.:581-49-7
- Isonicoteine
Catalog No.:BCN2152
CAS No.:581-50-0
Engineering the Excited-State Dynamics of 3-Aminoquinoline by Chemical Modification and Temperature Variation.[Pubmed:28002954]
J Phys Chem B. 2016 Dec 22;120(50):12920-12927.
The role of the amino group in the excited-state dynamics of 3-Aminoquinoline (3AQ) has been investigated by comparison with its synthetic derivative 3-(piperidin-1-yl)quinoline (3PQ). The absence of amino hydrogen atoms in 3PQ eliminates, to a large extent, the complexity of the excited-state processes observed in 3AQ. The polarity of the medium is found to be the most important determinant in the nonradiative rate constants of 3PQ, unlike in 3AQ where hydrogen bonding plays the most significant role. The nonradiative rate constants decrease with increase in micropolarity. This trend is opposite to what is usually observed with dipolar states. Temperature dependence of the fluorescence spectra and lifetime has been studied to understand this unexpected observation. An unusual redshift in the emission of 3AQ and 3PQ is observed in nonpolar media at low temperatures. This is surprising, as a process involving a barrier is expected to be hindered at low temperatures and be manifested in a blueshift of the spectra, due to the predominance of the locally excited (LE) state. Moreover, the variation of emission maxima of 3AQ with temperature is sigmoidal in nature, indicating the involvement of two distinct states. The counterintuitive observation of the predominance of the state with comparatively lower emission energy, at low temperatures, establishes the following: the photophysics in 3AQ is dominated by a LE state at room temperature in nonpolar media. This state is associated with rapid flip-flop of the amino group, which provides an efficient nonradiative channel of deactivation. At low temperatures, this flip-flop is hindered and the molecule can undergo intramolecular charge transfer (ICT), whereby the lower energy state is populated. In the case of 3PQ, the ICT state is the only one present, owing to the tertiary amino group.
Synthesis of 1,4-Benzodiazepine-2,5-diones by Base Promoted Ring Expansion of 3-Aminoquinoline-2,4-diones.[Pubmed:27787977]
J Org Chem. 2017 Jan 6;82(1):715-722.
An unprecedented reactivity of 3-Aminoquinoline-2,4-diones is reported. Under basic conditions, these compounds undergo molecular rearrangement to furnish 1,4-benzodiazepine-2,5-diones. The transformations take place under mild reaction conditions by using 1,1,3,3-tetramethylguanidine, NaOEt, or benzyltrimethylammonium hydroxide as a base. A proposed mechanism of the rearrangement and the conformational equilibrium of 1,4-benzodiazepine-2,5-dione rings are discussed.


