VomifoliolCAS# 23526-45-6 |
- Corchoionol C
Catalog No.:BCN1172
CAS No.:189351-15-3
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 23526-45-6 | SDF | Download SDF |
| PubChem ID | 5280462 | Appearance | Powder |
| Formula | C13H20O3 | M.Wt | 224.3 |
| Type of Compound | Sesquiterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
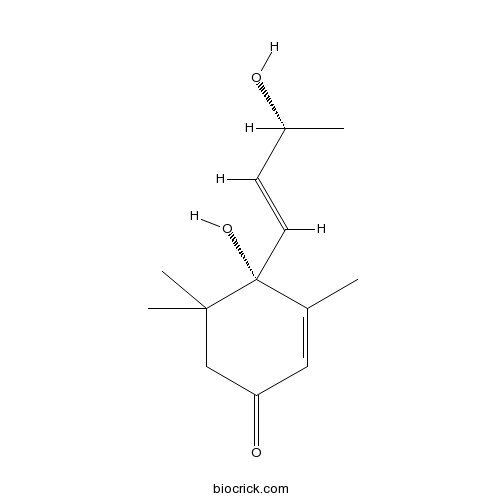
| Chemical Name | (4S)-4-hydroxy-4-[(E,3R)-3-hydroxybut-1-enyl]-3,5,5-trimethylcyclohex-2-en-1-one | ||
| SMILES | CC1=CC(=O)CC(C1(C=CC(C)O)O)(C)C | ||
| Standard InChIKey | KPQMCAKZRXOZLB-KOIHBYQTSA-N | ||
| Standard InChI | InChI=1S/C13H20O3/c1-9-7-11(15)8-12(3,4)13(9,16)6-5-10(2)14/h5-7,10,14,16H,8H2,1-4H3/b6-5+/t10-,13-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Vomifoliol is most likely the immediate precursor of dehydrovomifoliol, because of the organism converted abscisic acid (ABA), to dehydrovomifoliol as the major metabolise, and a cell-free extract exhibits vomifoliol dehydrogenase activity. 2. Vomifoliol and stigmasterol have anti-proliferative activity on human lung cancer cell lines. |
| Targets | NF-kB | HIF | VEGFR |

Vomifoliol Dilution Calculator

Vomifoliol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.4583 mL | 22.2916 mL | 44.5831 mL | 89.1663 mL | 111.4579 mL |
| 5 mM | 0.8917 mL | 4.4583 mL | 8.9166 mL | 17.8333 mL | 22.2916 mL |
| 10 mM | 0.4458 mL | 2.2292 mL | 4.4583 mL | 8.9166 mL | 11.1458 mL |
| 50 mM | 0.0892 mL | 0.4458 mL | 0.8917 mL | 1.7833 mL | 2.2292 mL |
| 100 mM | 0.0446 mL | 0.2229 mL | 0.4458 mL | 0.8917 mL | 1.1146 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- (-)-licarin A
Catalog No.:BCN5087
CAS No.:23518-30-1
- 10-Gingerol
Catalog No.:BCN5922
CAS No.:23513-15-7
- 6-Gingerol
Catalog No.:BCN1030
CAS No.:23513-14-6
- 8-Gingerol
Catalog No.:BCN5921
CAS No.:23513-08-8
- Humulon
Catalog No.:BCC8186
CAS No.:23510-81-8
- MEN 11270
Catalog No.:BCC6094
CAS No.:235082-52-7
- Axillaridine
Catalog No.:BCN2060
CAS No.:23506-96-9
- Peimine
Catalog No.:BCN1094
CAS No.:23496-41-5
- Hoechst 33258 analog 5
Catalog No.:BCC1627
CAS No.:23491-55-6
- Hoechst 33258 analog 2
Catalog No.:BCC1625
CAS No.:23491-54-5
- Hoechst 33342
Catalog No.:BCC1629
CAS No.:23491-52-3
- Hoechst 33258
Catalog No.:BCC1623
CAS No.:23491-45-4
- 5-Aza-2'-deoxycytidine
Catalog No.:BCN2169
CAS No.:2353-33-5
- 8-Debenzoylpaeoniflorin
Catalog No.:BCC8787
CAS No.:23532-11-8
- Daunorubicin HCl
Catalog No.:BCC5083
CAS No.:23541-50-6
- Hoechst 33258 analog 3
Catalog No.:BCC1626
CAS No.:23554-98-5
- HOE 32020
Catalog No.:BCC1620
CAS No.:23554-99-6
- Hoechst 34580
Catalog No.:BCC1632
CAS No.:23555-00-2
- Procyanidin B3
Catalog No.:BCN6316
CAS No.:23567-23-9
- Clotrimazole
Catalog No.:BCC3755
CAS No.:23593-75-1
- Norisoboldine
Catalog No.:BCN6285
CAS No.:23599-69-1
- H-Phe-pNA
Catalog No.:BCC3010
CAS No.:2360-97-6
- [Arg14,Lys15]Nociceptin
Catalog No.:BCC5781
CAS No.:236098-40-1
- HOE 32021
Catalog No.:BCC1621
CAS No.:23623-06-5
Organic extractives from Mentha spp. honey and the bee-stomach: methyl syringate, vomifoliol, terpenediol I, hotrienol and other compounds.[Pubmed:20428087]
Molecules. 2010 Apr 22;15(4):2911-24.
The GC and GC/MS analyses of the solvent organic extractive from the stomach of the bees, having collected Mentha spp. nectar, revealed the presence of methyl syringate (6.6%), terpendiol I (5.0%) and Vomifoliol (3.0%) that can be attributed to the plant origin. Other major compounds from the bee-stomach were related to the composition of cuticular waxes and less to pheromones. Organic extractives from Mentha spp. honey were obtained by solvent-free headspace solid-phase microextraction (HS-SPME) and ultrasonic solvent extraction (USE) and analyzed by GC and GC/MS. The major honey headspace compounds were hotrienol (31.1%-38.5%), 2-methoxy-4-methylphenol (0.5-6.0%), cis- and trans-linalool oxides (0.9-2.8%), linalool (1.0-3.1%) and neroloxide (0.9-1.9%). Methyl syringate was the most abundant compound (38.3-56.2%) in the honey solvent extractives followed by Vomifoliol (7.0-26.6%). Comparison of the honey organic extractives with the corresponding bee-stomach extractive indicated that methyl syringate and vomofoliol were transferred to the honey while terpendiol I was partially transformed to hotrienol in ripened honey.
Screening of natural organic volatiles from Prunus mahaleb L. honey: coumarin and vomifoliol as nonspecific biomarkers.[Pubmed:21407151]
Molecules. 2011 Mar 16;16(3):2507-18.
Headspace solid-phase microextraction (HS-SPME; PDMS/DVB fibre) and ultrasonic solvent extraction (USE; solvent A: pentane and diethyl ether (1:2 v/v), solvent B: dichloromethane) followed by gas chromatography and mass spectrometry (GC, GC-MS) were used for the analysis of Prunus mahaleb L. honey samples. Screening was focused toward chemical composition of natural organic volatiles to determine if it is useful as a method of determining honey-sourcing. A total of 34 compounds were identified in the headspace and 49 in the extracts that included terpenes, norisoprenoids and benzene derivatives, followed by minor percentages of aliphatic compounds and furan derivatives. High Vomifoliol percentages (10.7%-24.2%) in both extracts (dominant in solvent B) and coumarin (0.3%-2.4%) from the extracts (more abundant in solvent A) and headspace (0.9%-1.8%) were considered characteristic for P. mahaleb honey and highlighted as potential nonspecific biomarkers of the honey's botanical origin. In addition, comparison with P. mahaleb flowers, leaves, bark and wood volatiles from our previous research revealed common compounds among norisoprenoids and benzene derivatives.


