L-161,982Selective EP4 receptor antagonist CAS# 147776-06-5 |
- 3-Deazaneplanocin,DZNep
Catalog No.:BCC1129
CAS No.:102052-95-9
- 3-Deazaneplanocin A (DZNep) hydrochloride
Catalog No.:BCC3604
CAS No.:120964-45-6
- CHIR-99021 (CT99021)
Catalog No.:BCC1275
CAS No.:252917-06-9
- PluriSIn #1 (NSC 14613)
Catalog No.:BCC2305
CAS No.:91396-88-2
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 147776-06-5 | SDF | Download SDF |
| PubChem ID | 9961192 | Appearance | Powder |
| Formula | C32H29F3N4O4S2 | M.Wt | 654.72 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in DMSO | ||
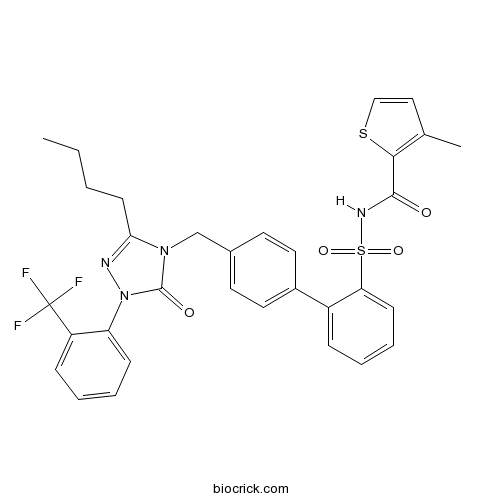
| Chemical Name | N-[2-[4-[[3-butyl-5-oxo-1-[2-(trifluoromethyl)phenyl]-1,2,4-triazol-4-yl]methyl]phenyl]phenyl]sulfonyl-3-methylthiophene-2-carboxamide | ||
| SMILES | CCCCC1=NN(C(=O)N1CC2=CC=C(C=C2)C3=CC=CC=C3S(=O)(=O)NC(=O)C4=C(C=CS4)C)C5=CC=CC=C5C(F)(F)F | ||
| Standard InChIKey | MMDNKTXNUZFVKD-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C32H29F3N4O4S2/c1-3-4-13-28-36-39(26-11-7-6-10-25(26)32(33,34)35)31(41)38(28)20-22-14-16-23(17-15-22)24-9-5-8-12-27(24)45(42,43)37-30(40)29-21(2)18-19-44-29/h5-12,14-19H,3-4,13,20H2,1-2H3,(H,37,40) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | EP4 receptor antagonist that is selective over all other members of the prostanoid receptor family (Ki values are 0.024, 0.71, 1.90, 5.10, 5.63, 6.74, 19 and 23 μM for human EP4, TP, EP3, DP, FP, IP, EP1 and EP2 receptors respectively). Suppresses PGE2-induced bone formation in rats and prevents the nociceptive response induced by misoprostol in formalin-injected mice. |

L-161,982 Dilution Calculator

L-161,982 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.5274 mL | 7.6369 mL | 15.2737 mL | 30.5474 mL | 38.1843 mL |
| 5 mM | 0.3055 mL | 1.5274 mL | 3.0547 mL | 6.1095 mL | 7.6369 mL |
| 10 mM | 0.1527 mL | 0.7637 mL | 1.5274 mL | 3.0547 mL | 3.8184 mL |
| 50 mM | 0.0305 mL | 0.1527 mL | 0.3055 mL | 0.6109 mL | 0.7637 mL |
| 100 mM | 0.0153 mL | 0.0764 mL | 0.1527 mL | 0.3055 mL | 0.3818 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Repaglinide ethyl ester
Catalog No.:BCC9135
CAS No.:147770-06-7
- Fmoc-O-Phospho-Tyr-OH
Catalog No.:BCC3563
CAS No.:147762-53-6
- 3,4-Dimethoxybenzenepropanamine
Catalog No.:BCN1785
CAS No.:14773-42-3
- 6-O-(3'',4''-Dimethoxycinnamoyl)catalpol
Catalog No.:BCN1655
CAS No.:147714-71-4
- 8-(3-Chlorostyryl)caffeine
Catalog No.:BCC7640
CAS No.:147700-11-6
- WIN 18446
Catalog No.:BCC6273
CAS No.:1477-57-2
- 2-Amino-4-methylbenzothiazole
Catalog No.:BCC8533
CAS No.:1477-42-5
- ZM 226600
Catalog No.:BCC6831
CAS No.:147695-92-9
- Magnolianin
Catalog No.:BCN3985
CAS No.:147663-91-0
- Calcipotriol monohydrate
Catalog No.:BCC1445
CAS No.:147657-22-5
- 5-Chloro-4-methoxy-2-oxo-1,2-dihydropyridine-3-carbonitrile
Catalog No.:BCC8744
CAS No.:147619-40-7
- Novobiocin Sodium
Catalog No.:BCC4812
CAS No.:1476-53-5
- DCG IV
Catalog No.:BCC5691
CAS No.:147782-19-2
- ω-Conotoxin MVIIC
Catalog No.:BCC5699
CAS No.:147794-23-8
- Santacruzamate A (CAY10683)
Catalog No.:BCC5488
CAS No.:1477949-42-0
- Cefcapene pivoxil hydrochloride
Catalog No.:BCC8906
CAS No.:147816-24-8
- Siramesine
Catalog No.:BCC4304
CAS No.:147817-50-3
- Niazimicin
Catalog No.:BCN7641
CAS No.:147821-49-6
- Niazinin
Catalog No.:BCN7623
CAS No.:147821-57-6
- Filic-3-en-25-al
Catalog No.:BCN6445
CAS No.:147850-78-0
- CA-074 Me
Catalog No.:BCC3649
CAS No.:147859-80-1
- Isokadsurenin D
Catalog No.:BCN6615
CAS No.:147976-35-0
- Dinitolmide
Catalog No.:BCC8945
CAS No.:148-01-6
- Beta-Tocopherol
Catalog No.:BCN6683
CAS No.:148-03-8
The EP4 receptor antagonist, L-161,982, blocks prostaglandin E2-induced signal transduction and cell proliferation in HCA-7 colon cancer cells.[Pubmed:17631291]
Exp Cell Res. 2007 Aug 15;313(14):2969-79.
Accumulating evidence indicates that elevated levels of prostaglandin E(2) (PGE(2)) can increase intestinal epithelial cell proliferation, and thus play a role in colorectal tumorigenesis. PGE(2) exerts its effects through four G-protein-coupled PGE receptor (EP) subtypes, named the EP1, EP2, EP3, and EP4. Increased phosphorylation of extracellular regulated kinases (ERK1/2) is required for PGE(2) to stimulate cell proliferation of human colon cancer cells. However, the EP receptor(s) that are involved in this process remain unknown. We provide evidence that L-161,982, a selective EP4 receptor antagonist, completely blocks PGE(2)-induced ERK phosphorylation and cell proliferation of HCA-7 cells. In order to identify downstream target genes of ERK1/2 signaling, we found that PGE(2) induces expression of early growth response gene-1 (EGR-1) downstream of ERK1/2 and regulates its expression at the level of transcription. PGE(2) treatment induces phosphorylation of cyclic AMP response element binding protein (CREB) at Ser133 residue and CRE-mediated luciferase activity in HCA-7 cells. Studies with dominant-negative CREB mutant (ACREB) provide clear evidence for the involvement of CREB in PGE(2) driven egr-1 transcription in HCA-7 cells. In conclusion, this study reveals that egr-1 is a target gene of PGE(2) in HCA-7 cells and is regulated via the newly identified EP4/ERK/CREB pathway. Finally our results support the notion that antagonizing EP4 receptors may provide a novel therapeutic approach to the treatment of colon cancer.
The EP4 antagonist, L-161,982, induces apoptosis, cell cycle arrest, and inhibits prostaglandin E2-induced proliferation in oral squamous carcinoma Tca8113 cells.[Pubmed:28342204]
J Oral Pathol Med. 2017 Nov;46(10):991-997.
BACKGROUND: Recent studies suggest that cyclooxygenase 2 (COX-2) inhibitors may enhance the toxic effects of anticancer drugs on tumor cells, including oral squamous cell carcinoma (OSCC), but its long-term use can cause side effects such as stomach ulcers and myocardial infarction. Our aim was to investigate proliferative effects of a downstream product of COX-2, prostaglandin E2 (PGE2), in human oral squamous carcinoma cell line Tca8113 and explore the effects of PGE2 receptors, especially EP4 receptor, on the growth of Tca8113 cells. METHODS: To evaluate the effects of PGE2 and EP receptors on Tca8113 cells, CCK8 assay, Western blotting, cell cycle analysis, and apoptosis assay were performed. RESULTS: We found that the EP4 receptor agonist, PGE1-OH, could mimick PGE2 rescued the inhibitory effect of celecoxib and induced cell growth via ERK phosphorylation, and the EP4 receptor antagonist, L-161,982, completely blocked PGE2-stimulated ERK phosphorylation and proliferation of Tca8113 cells. Furthermore, L-161,982 may induce apoptosis and block cell cycle progression at s phase by upregulating Bax and p21 protein levels and by downregulating Bcl-2, CDK2, and cyclin A2 protein levels. CONCLUSIONS: Our results indicate that EP4 receptor mediates PGE2-induced cell proliferation through ERK signaling, and inhibition of EP4 receptor may represent an alternative therapeutic strategy for the prevention and treatment of OSCC.
Lipopolysaccharide induces epithelium- and prostaglandin E(2)-dependent relaxation of mouse isolated trachea through activation of cyclooxygenase (COX)-1 and COX-2.[Pubmed:16464966]
J Pharmacol Exp Ther. 2006 May;317(2):806-12.
Lipopolysaccharide (LPS), a Toll-like receptor (TLR) 4 agonist, causes airway hyperreactivity through nuclear factor-kappaB (NF-kappaB). Because NF-kappaB induces cyclooxygenase-2 (COX-2) to increase synthesis of prostaglandins (PGs), including the potent airway anti-inflammatory and smooth muscle relaxant PGE(2), we investigated whether LPS causes short-term PGE(2)-dependent relaxation of mouse isolated trachea. In rings of trachea contracted submaximally with carbachol, LPS caused slowly developing, epithelium-dependent relaxations that reached a maximum within 60 min. Fluorescence immunohistochemistry revealed TLR4-like immunoreactivity localized predominantly to the epithelium. The LPS antagonist polymixin B; the nonselective COX inhibitor indomethacin; the selective COX-1 and COX-2 inhibitors 5-(4-chlorophenyl)-1-(4-methoxyphenyl)-3-(trifluoromethyl)-1H-pyrazole (SC560) and 4-[5-(4-chlorophenyl)-1-(trifluoromethyl)-1H-pyrazol-1-yl]-benzenesulfonamide (SC236), respectively; the transcription inhibitor actinomycin D; the translation inhibitor cycloheximide; the p38 mitogen-activated protein kinase (p38 MAPK) inhibitor 4-(4-fluorophenyl)-2-(4-methylsulfinylphenyl)-5-(4-pyridyl)-1H-imadazole (SB203580); and a combination of the mixed DP/EP1/EP2 receptor antagonist 6-isopropoxy-9-xanthone-2-carboxylic acid (AH6809) and the EP4 receptor antagonist 4'-[3-butyl-5-oxo-1-(2-trifluoromethyl-phenyl)-1-5-dihydro-[1,2,4]triazol-4-ylmet hyl]-biphenyl-2-sulfonic acid (3-methyl-thiophene-2-carbonyl)-amide (L-161982) all abolished relaxation to LPS, giving instead slowly developing, small contractions over 60 min. The cytosolic phospholipase A(2) (cPLA(2)) inhibitor 1,1,1-trifluoro-6Z,9Z, 12Z,15Z-heneicosateraen-2-one significantly (p < 0.05) inhibited the relaxation to LPS, whereas the NF-kappaB proteasomal inhibitor Z-Leu-Leu-Leu-aldehyde (MG-132) had no affect on the relaxation in the first 20 min, after which it reversed the response to a contraction. In conclusion, our data indicate that LPS activates airway epithelial TLR4 to cause release of PGE(2) and subsequent EP2 and EP4 receptor-dependent smooth muscle relaxation. Activation of both COX-1 and COX-2 seems to be essential for this novel response to LPS, which also involves cPLA(2), p38 MAPK, NF-kappaB, and an unidentified NF-kappaB-independent, labile regulatory protein.
Role of periaqueductal grey prostaglandin receptors in formalin-induced hyperalgesia.[Pubmed:16360148]
Eur J Pharmacol. 2006 Jan 13;530(1-2):40-7.
In this study we have investigated the role of periaqueductal grey prostaglandin receptors in formalin-induced hyperalgesia in mice. Glutamate and GABA release changes have been monitored by in vivo microdialysis. Intra-periaqueductal grey microinjections of misoprostol, a non-selective prostaglandin receptor agonist, increased nociceptive responses in the formalin test only during the late phase. Prostanoid EP(1) (L-335677), EP(2) (AH 6809), EP(3) (L-826266) and EP(4) (L-161982) receptor antagonists prevented the nociceptive response induced by misoprostol in formalin-injected mice. Prostanoid EP(1), EP(2), EP(3) and EP(4) antagonists reduced, per se, the late hyperalgesic phase. Intra-periaqueductal grey perfusion with misoprostol increased periaqueductal grey glutamate, whereas it produced an increase followed by a decrease in GABA. Likewise, formalin increased glutamate and produced a biphasic response on GABA. When misoprostol was perfused in combination with the peripheral injection of formalin, we observed an increase of glutamate and an increase followed by a stronger decrease in GABA release. These data show that periaqueductal grey prostaglandin receptor stimulation increased formalin-induced nociceptive response in the late phase by increasing glutamate release and by producing a biphasic change in GABA release.
Prostaglandin receptor EP(4) mediates the bone anabolic effects of PGE(2).[Pubmed:11408598]
Mol Pharmacol. 2001 Jul;60(1):36-41.
Prostaglandin (PG) E(2) is a potent inducer of cortical and trabecular bone formation in humans and animals. Although the bone anabolic action of PGE(2) is well documented, the cellular and molecular mechanisms that mediate this effect remain unclear. This study was undertaken to examine the effect of pharmacological inactivation of the prostanoid receptor EP(4), one of the PGE(2) receptors, on PGE(2)-induced bone formation in vivo. We first determined the ability of EP(4)A, an EP(4)-selective ligand, to act as an antagonist. PGE(2) increases intracellular cAMP and suppresses apoptosis in the RP-1 periosteal cell line. Both effects were reversed by EP(4)A, suggesting that EP(4)A acts as an EP(4) antagonist in the cells at concentrations consistent with its in vitro binding to EP(4). We then examined the effect of EP(4) on bone formation induced by PGE(2) in young rats. Five- to 6-week-old rats were treated with PGE(2) (6 mg/kg/day) in the presence or absence of EP(4)A (10 mg/kg/day) for 12 days. We found that treatment with EP(4)A suppresses the increase in trabecular bone volume induced by PGE(2). This effect is accompanied by a suppression of bone formation indices: serum osteocalcin, extent of labeled surface, and extent of trabecular number, suggesting that the reduction in bone volume is due most likely to decreased bone formation. The pharmacological evidence presented here provides strong support for the hypothesis that the bone anabolic effect of PGE(2) in rats is mediated by the EP(4) receptor.


