AM1241Cannabinoid CB2 receptor agonist,potent and selective CAS# 444912-48-5 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 444912-48-5 | SDF | Download SDF |
| PubChem ID | 10141893 | Appearance | Powder |
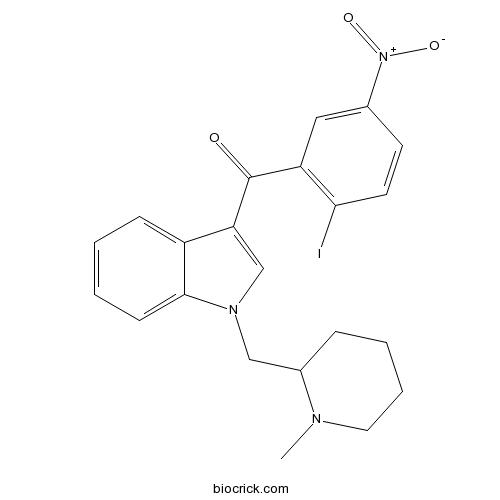
| Formula | C22H22IN3O3 | M.Wt | 503.33 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 101 mg/mL (200.66 mM) in DMSO | ||
| Chemical Name | (2-iodo-5-nitrophenyl)-[1-[(1-methylpiperidin-2-yl)methyl]indol-3-yl]methanone | ||
| SMILES | CN1CCCCC1CN2C=C(C3=CC=CC=C32)C(=O)C4=C(C=CC(=C4)[N+](=O)[O-])I | ||
| Standard InChIKey | ZUHIXXCLLBMBDW-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C22H22IN3O3/c1-24-11-5-4-6-16(24)13-25-14-19(17-7-2-3-8-21(17)25)22(27)18-12-15(26(28)29)9-10-20(18)23/h2-3,7-10,12,14,16H,4-6,11,13H2,1H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | AM-1241 is a selective agonist of cannabinoid CB2 receptor with a Ki value of 3.4 nM. | |||||
| Targets | CB2 | CB1 | ||||
| IC50 | 3.4 nM(Ki) | 280 nM(Ki) | ||||

AM1241 Dilution Calculator

AM1241 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.9868 mL | 9.9338 mL | 19.8677 mL | 39.7354 mL | 49.6692 mL |
| 5 mM | 0.3974 mL | 1.9868 mL | 3.9735 mL | 7.9471 mL | 9.9338 mL |
| 10 mM | 0.1987 mL | 0.9934 mL | 1.9868 mL | 3.9735 mL | 4.9669 mL |
| 50 mM | 0.0397 mL | 0.1987 mL | 0.3974 mL | 0.7947 mL | 0.9934 mL |
| 100 mM | 0.0199 mL | 0.0993 mL | 0.1987 mL | 0.3974 mL | 0.4967 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
IC50: AM-1241 is a selective cannabinoid CB2 receptor agonist with Ki of 3.4 nM, exhibits 82-fold selectivity over CB1 receptor. [1].
The cannabinoid receptor type 2, abbreviated as CB2, is a G protein-coupled receptor from the cannabinoid receptor family that in humans is encoded by the CNR2 gene. CB2 receptors may have possible therapeutic roles in the treatment of neurodegenerative disorders such as Alzheimer's disease. AM-1241, a chemical from the aminoalkylindole family, acts as a potent and selective agonist for the cannabinoid receptor CB2, has analgesic effects in animal studies, particularly against "atypical" pain such as hyperalgesia and allodynia.
In vitro: The purpose of our studies was to provide a characterization of R,S-AM1241 and its resolved enantiomers in vitro and in vivo. In cAMP inhibition assays, R,S-AM1241 was found to be an agonist at human CB2, but an inverse agonist at rat and mouse CB2 receptors. R-AM1241 bound with more than 40-fold higher affinity than S-AM1241, to all three CB2 receptors and displayed a functional profile similar to that of the racemate. In contrast, S-AM1241 was an agonist at all three CB2 receptors. In pain models, S-AM1241 was more efficacious than either R-AM1241 or the racemate. Antagonist blockade demonstrated that the in vivo effects of S-AM1241 were mediated by CB2 receptors. These findings constitute the first in vitro functional assessment of R,S-AM1241 at rodent CB2 receptors and the first characterization of the AM1241 enantiomers in recombinant cell systems and in vivo. The greater antinociceptive efficacy of S-AM1241, the functional CB2 agonist enantiomer of AM1241, is consistent with previous observations that CB2 agonists are effective in relief of pain [2].
In vivo:
Effective treatment or amyotrophic lateral sclerosis (ALS) remains elusive. Motor neuron degeneration is the primary pathology in ALS; however non-neuronal cells contribute to the disease process. In particular, inflammatory processes have been shown to play an important role. AM1241 is a cannabinoid CB2 receptor selective agonist that has been shown to be effective in models of inflammation and hyperalgesia. Here we report that treatment with AM1241 was effective at slowing signs of disease progression when administered after onset of signs in an ALS mouse model (hSOD1G93A transgenic mice). Administration at the onset of tremors delayed motor impairment in treated mice when compared to vehicle controls. Three conditions of ALS, the loss of motor function, paralysis scoring and weight loss, were analyzed using a mathematical model. Loss of motor function (as assessed by performance on a rotarod) was delayed by 12.5 days in male mice by AM1241. In female mice, AM1241 extended rotarod performance by 3 days, although this was not statistically significant. In male mice, AM1241 also extended by 5 days the time to reach the 50%point on a visually-assessed performance scale.AM1241 did not affectweight loss or survival (129.8±1.7 days, vehicle; 129.1±7.0 days, AM1241, n=16). As AM1241 was well tolerated by the animals, cannabinoid CB2 receptor-selective compounds may be the basis for developing new drugs for the treatment of ALS and other chronic neurodegenerative diseases [3].
Clinical trial: AM1241 is still in preclinical development phase and no clinical study has been found.
Reference:
[1] Ibrahim MM, Deng H, Zvonok A, Cockayne DA, Kwan J, Mata HP, Vanderah TW, Lai J, Porreca F, Makriyannis A, Malan TP Jr. Activation of CB2 cannabinoid receptors by AM1241 inhibits experimental neuropathic pain: pain inhibition by receptors not present in the CNS. Proc Natl Acad Sci U S A. 2003;100(18):10529-33.
[2] B Bingham, PG Jones, AJ Uveges, S Kotnis, P Lu, VA Smith, S-C Sun, L Resnick, M Chlenov, Y He. Species-specific in vitro pharmacological effects of the cannabinoid receptor 2 (CB2) selective ligand AM1241 and its resolved enantiomers. British Journal of Pharmacology (2007) 151, 1061–1070
[3] Kathline Kim, Dan H. Moore, Alexandros Makriyannis, Mary E. Abood. AM1241, a cannabinoid CB2 receptor selective compound, delays disease progression in a mouse model of amyotrophic lateral sclerosis. European Journal of Pharmacology 542 (2006) 100-105
- Warangalone
Catalog No.:BCN4788
CAS No.:4449-55-2
- Cyclopamine
Catalog No.:BCN2964
CAS No.:4449-51-8
- CMX001
Catalog No.:BCC4106
CAS No.:444805-28-1
- Pazopanib (GW-786034)
Catalog No.:BCC1286
CAS No.:444731-52-6
- Norwogonin
Catalog No.:BCN3190
CAS No.:4443-09-8
- 2-Cyclohexylethylamine
Catalog No.:BCN1794
CAS No.:4442-85-7
- 1,4-Benzodioxane-6-carboxylic acid
Catalog No.:BCC8422
CAS No.:4442-54-0
- Vandetanib (ZD6474)
Catalog No.:BCC3883
CAS No.:443913-73-3
- Glycosmisic acid
Catalog No.:BCN3650
CAS No.:443908-19-8
- Norkhellol
Catalog No.:BCN5497
CAS No.:4439-68-3
- 3-Chloro-4-(3-fluorobenzyloxy)nitrobenzene
Catalog No.:BCC8626
CAS No.:443882-99-3
- JNJ-7706621
Catalog No.:BCC2171
CAS No.:443797-96-4
- TC-O 9311
Catalog No.:BCC7900
CAS No.:444932-31-4
- EBPC
Catalog No.:BCC6677
CAS No.:4450-98-0
- 1,4:3,6-Dianhydro-α-D-glucopyranose
Catalog No.:BCC8420
CAS No.:4451-30-3
- ZM 449829
Catalog No.:BCC2444
CAS No.:4452-06-6
- BMS-345541(free base)
Catalog No.:BCC5374
CAS No.:445430-58-0
- SGS 518 oxalate
Catalog No.:BCC7750
CAS No.:445441-27-0
- BMS CCR2 22
Catalog No.:BCC7572
CAS No.:445479-97-0
- Homoeriodictyol
Catalog No.:BCN6804
CAS No.:446-71-9
- Genistein
Catalog No.:BCN5499
CAS No.:446-72-0
- Azathioprine
Catalog No.:BCC4762
CAS No.:446-86-6
- 2,4,5-Trimethoxybenzaldehyde
Catalog No.:BCN5498
CAS No.:4460-86-0
- YM 230888
Catalog No.:BCC5956
CAS No.:446257-23-4
The cannabinoid CB2 receptor-specific agonist AM1241 increases pentylenetetrazole-induced seizure severity in Wistar rats.[Pubmed:27608434]
Epilepsy Res. 2016 Nov;127:160-167.
The potential efficacy of cannabinoid receptor ligands for the treatment of epilepsy remains controversial; cannabis components that act via cannabinoid type 1 (CB1) receptors produce anticonvulsant effects in animal models despite treatment with the CB receptor agonist reliably inducing convulsions in various species. Moreover, the potential role of cannabinoid receptor type 2 (CB2) to modulate seizures remains under-investigated. This study assessed the effects of the selective CB2 receptor agonist, AM1241, on pentylenetetrazole (PTZ)-induced seizures in rats. A stereotactically placed guide cannula was surgically implanted into the right lateral ventricle in adult Wistar rats which, 5-6days later, received an acute intracerebroventricular (i.c.v.) microinfusion of AM1241 (0.01, 1 or 10mug/2mul or vehicle) 5min before intraperitoneal (i.p.) injection of PTZ (70mg/kg). Rats were observed for 30min and the seizure severity behavior measured using a modified Racine's scale. Additional groups of rats were pretreated with a single low dose of the selective CB2 receptor antagonist, AM630 (dose 1mg/kg; i.p.), or vehicle, 30min prior to i.c.v. microinfusion of AM1241 (1mug/2mul). AM1241 administration significantly increased tonic-clonic seizure incidence and severity while also decreasing the onset of generalized seizures (AM1241 1 and 10mug/2mul). Pretreatment with AM630 prevented the proconvulsant effects of AM1241. This study shows, for the first time, that selective activation of CB2 receptors can increase generalized seizure susceptibility and suggests that pathological hyperexcitability phenomena can be differentially regulated by targeting CB1 and CB2 receptors.
The cannabinoid CB(2) receptor agonist AM1241 enhances neurogenesis in GFAP/Gp120 transgenic mice displaying deficits in neurogenesis.[Pubmed:24148086]
Br J Pharmacol. 2014 Jan;171(2):468-79.
BACKGROUND AND PURPOSE: HIV-1 glycoprotein Gp120 induces apoptosis in rodent and human neurons in vitro and in vivo. HIV-1/Gp120 is involved in the pathogenesis of HIV-associated dementia (HAD) and inhibits proliferation of adult neural progenitor cells (NPCs) in glial fibrillary acidic protein (GFAP)/Gp120 transgenic (Tg) mice. As cannabinoids exert neuroprotective effects in several model systems, we examined the protective effects of the CB(2) receptor agonist AM1241 on Gp120-mediated insults on neurogenesis. EXPERIMENTAL APPROACH: We assessed the effects of AM1241 on survival and apoptosis in cultures of human and murine NPCs with immunohistochemical and TUNEL techniques. Neurogenesis in the hippocampus of GFAP/Gp120 transgenic mice in vivo was also assessed by immunohistochemistry. KEY RESULTS: AM1241 inhibited in vitro Gp120-mediated neurotoxicity and apoptosis of primary human and murine NPCs and increased their survival. AM1241 also promoted differentiation of NPCs to neuronal cells. While GFAP/Gp120 Tg mice exhibited impaired neurogenesis, as indicated by reduction in BrdU(+) cells and doublecortin(+) (DCX(+)) cells, and a decrease in cells with proliferating cell nuclear antigen (PCNA), administration of AM1241 to GFAP/Gp120 Tg mice resulted in enhanced in vivo neurogenesis in the hippocampus as indicated by increase in neuroblasts, neuronal cells, BrdU(+) cells and PCNA(+) cells. Astrogliosis and gliogenesis were decreased in GFAP/Gp120 Tg mice treated with AM1241, compared with those treated with vehicle. CONCLUSIONS AND IMPLICATIONS: The CB(2) receptor agonist rescued impaired neurogenesis caused by HIV-1/Gp120 insult. Thus, CB(2) receptor agonists may act as neuroprotective agents, restoring impaired neurogenesis in patients with HAD.
Immunofluorescent spectral analysis reveals the intrathecal cannabinoid agonist, AM1241, produces spinal anti-inflammatory cytokine responses in neuropathic rats exhibiting relief from allodynia.[Pubmed:22574283]
Brain Behav. 2012 Mar;2(2):155-77.
During pathological pain, the actions of the endocannabinoid system, including the cannabinoid 2 receptor (CB(2)R), leads to effective anti-allodynia and modifies a variety of spinal microglial and astrocyte responses. Here, following spinal administration of the CB(2)R compound, AM1241, we examined immunoreactive alterations in markers for activated p38 mitogen-activated protein kinase, interleukin-1beta (IL-1beta), the anti-inflammatory cytokine, interleukin-10 (IL-10) as well as degradative endocannabinoid enzymes, and markers for altered glial responses in neuropathic rats. In these studies, the dorsal horn of the spinal cord and dorsal root ganglia were examined. AM1241 produced profound anti-allodynia with corresponding immunoreactive levels of p38 mitogen-activated kinase, IL-1beta, IL-10, the endocannabinoid enzyme monoacylglycerol lipase, and astrocyte activation markers that were similar to nonneuropathic controls. In contrast, spinal AM1241 did not suppress the increased microglial responses observed in neuropathic rats. The differences in fluorescent markers were determined within discrete anatomical regions by applying spectral analysis methods, which virtually eliminated nonspecific signal during the quantification of specific immunofluorescent intensity. These data reveal expression profiles that support the actions of intrathecal AM1241 control pathological pain through anti-inflammatory mechanisms by modulating critical glial factors, and additionally decrease expression levels of endocannabinoid degradative enzymes.
Activation of Cannabinoid Receptor Type II by AM1241 Ameliorates Myocardial Fibrosis via Nrf2-Mediated Inhibition of TGF-beta1/Smad3 Pathway in Myocardial Infarction Mice.[Pubmed:27614871]
Cell Physiol Biochem. 2016;39(4):1521-36.
AIMS: Myocardial interstitial fibrosis is a major histologic landmark resulting in cardiac dysfunction after myocardial infarction (MI). Activation of cannabinoid receptor type II (CB2 receptor) have been demonstrated to reduce fibrosis in hepatic cirrhotic rat. However, the anti-fibrotic effect of CB2 receptor activation in infarcted hearts was still unclear. In this study, we aimed to investigate the effects of a CB2 receptor selective agonist AM1241 on myocardial fibrosis post MI in mice. METHODS: Echocardiograph was conducted to assess cardiac function. Fibrosis markers such as type I and type III collagen, fibronectin, Plasminogen activator inhibitor(PAI)-1 and tissue inhibitor of metalloprotease(TIMP)-1 were examined by Western blot, while collagens were directly observed by Sirius-red staining. Primary cultured cardiac fibroblasts(CFs) were subjected to hypoxia/serum deprivation (H/SD) injury to simulate ischemic conditions in vivo. Nrf2 siRNA were applied to explore the role of Nrf2 and TGF-beta1/Smad3 pathway in this process. RESULTS: Echocardiography showed that AM1241 significantly improved cardiac function, suppressed the expression of fibrosis markers such as collagen I and collagen III, fibronectin, PAI-1 and TIMP-1 in mice with MI. In cardiac fibroblasts subjected to H/SD injury, AM1241 reduced the elevated levels of alpha-SMA, collagen I and collagen III, which were partially abrogated by the Nrf2 siRNA transfection. Furthermore, AM1241 not only activated and accelerated the translocation of Nrf2 to nucleus, but also inhibited TGF-beta1/ Smad3 pathway in an Nrf2 dependent manner. CONCLUSION: CB2 receptor agonist AM1241 alleviated myocardial interstitial fibrosis via Nrf2 -mediated down-regulation of TGF-beta1/Smad3 pathway, which suggested that CB2 receptor activation might represent a promising target for retarding cardiac fibrosis after MI.


