VitexilactoneCAS# 61263-49-8 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 61263-49-8 | SDF | Download SDF |
| PubChem ID | 21636178 | Appearance | Powder |
| Formula | C22H34O5 | M.Wt | 378.5 |
| Type of Compound | Diterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
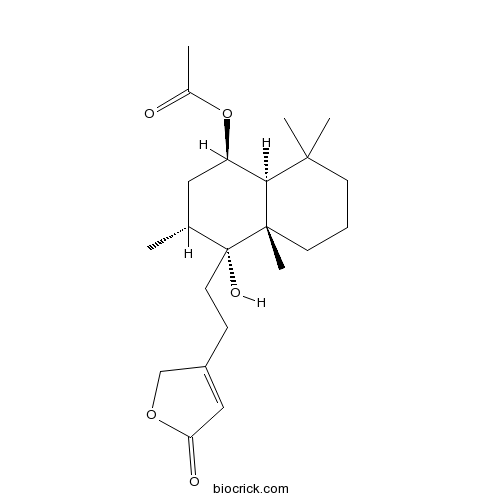
| Chemical Name | [(1R,3R,4R,4aS,8aS)-4-hydroxy-3,4a,8,8-tetramethyl-4-[2-(5-oxo-2H-furan-3-yl)ethyl]-2,3,5,6,7,8a-hexahydro-1H-naphthalen-1-yl] acetate | ||
| SMILES | CC1CC(C2C(CCCC2(C1(CCC3=CC(=O)OC3)O)C)(C)C)OC(=O)C | ||
| Standard InChIKey | FBWWXAGANVJTLU-HEXLTJKYSA-N | ||
| Standard InChI | InChI=1S/C22H34O5/c1-14-11-17(27-15(2)23)19-20(3,4)8-6-9-21(19,5)22(14,25)10-7-16-12-18(24)26-13-16/h12,14,17,19,25H,6-11,13H2,1-5H3/t14-,17-,19+,21+,22-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Vitexilactone dramatically induces apoptosis both on tsFT210 and K562 cells at higher concentrations while at lower concentrations it inhibits the cell cycle progression of both tsFT210 and K562 cells at the G0/G1 phase; it is one anticancer component of Vitex trifolia L., which exerts its anti-proliferative effect on cancer cells through inducing apoptosis and inhibiting the cell cycle. 2. Vitexilactone has trypanocidal activity, the minimum lethal concentration against epimastigotes of Trypanosoma cruzi is 66 microM . 3. Vitexilactone is a HIV-1 reverse transcriptase inhibitor in virtual screening against Indonesian Herbal Database using AutoDock4 performed on HIV-1 reverse transcriptase. |
| Targets | HIV | NADPH-oxidase | Antifection |

Vitexilactone Dilution Calculator

Vitexilactone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.642 mL | 13.21 mL | 26.4201 mL | 52.8402 mL | 66.0502 mL |
| 5 mM | 0.5284 mL | 2.642 mL | 5.284 mL | 10.568 mL | 13.21 mL |
| 10 mM | 0.2642 mL | 1.321 mL | 2.642 mL | 5.284 mL | 6.605 mL |
| 50 mM | 0.0528 mL | 0.2642 mL | 0.5284 mL | 1.0568 mL | 1.321 mL |
| 100 mM | 0.0264 mL | 0.1321 mL | 0.2642 mL | 0.5284 mL | 0.6605 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Cannabispiran
Catalog No.:BCN4134
CAS No.:61262-81-5
- PTP1B-IN-1
Catalog No.:BCC5506
CAS No.:612530-44-6
- AZD1080
Catalog No.:BCC4508
CAS No.:612487-72-6
- Denudadione C
Catalog No.:BCN6608
CAS No.:61240-34-4
- 11-Hydroxybisabola-1,3,5-trien-9-one
Catalog No.:BCN7530
CAS No.:61235-23-2
- 6alpha-Hydroxymaackiain
Catalog No.:BCN3947
CAS No.:61218-44-8
- Uzarigenin digitaloside
Catalog No.:BCN4613
CAS No.:61217-80-9
- Quinine HCl Dihydrate
Catalog No.:BCC4933
CAS No.:6119-47-7
- Grandifloroside
Catalog No.:BCN4133
CAS No.:61186-24-1
- 4-(3,4-Dihydroxyphenyl)-2-butanone
Catalog No.:BCN4132
CAS No.:61152-62-3
- 6alpha-Hydroxymedicarpin
Catalog No.:BCN3939
CAS No.:61135-92-0
- 3,9-Dihydroxypterocarpan
Catalog No.:BCN4131
CAS No.:61135-91-9
- Acteoside
Catalog No.:BCN4136
CAS No.:61276-17-3
- Boc-D-Phe(4-NO2)-OH
Catalog No.:BCC3276
CAS No.:61280-75-9
- Schizandrin B
Catalog No.:BCN1022
CAS No.:61281-37-6
- Schizandrin A
Catalog No.:BCN1021
CAS No.:61281-38-7
- AKT inhibitor VIII
Catalog No.:BCC1334
CAS No.:612847-09-3
- 2-(Phenylmethoxy)-naphthalene
Catalog No.:BCC8485
CAS No.:613-62-7
- 2-Aminoacetophenone
Catalog No.:BCC8546
CAS No.:613-89-8
- Schisandrin C
Catalog No.:BCN1198
CAS No.:61301-33-5
- Isoacteoside
Catalog No.:BCN4137
CAS No.:61303-13-7
- Boc-D-Arg(Tos)-OH
Catalog No.:BCC3070
CAS No.:61315-61-5
- Sulconazole Nitrate
Catalog No.:BCC4853
CAS No.:61318-91-0
- Neoschaftoside
Catalog No.:BCN3053
CAS No.:61328-41-4
New norditerpenoids with trypanocidal activity from Vitex trifolia.[Pubmed:15577254]
Chem Pharm Bull (Tokyo). 2004 Dec;52(12):1492-4.
Trypanocidal constituents of the fruits of Vitex trifolia were investigated. Activity-guided isolation of the acetone extract resulted in the isolation of two new norditerpene aldehydes, 1 and 2, together with five known diterpenes: vitexifolin E (3), vitexifolin F (4), Vitexilactone (5), 6-acetoxy-9-hydroxy-13(14)-labden-16,15-olide (6), and preVitexilactone (7). In vitro minimum lethal concentrations of the isolated compounds against epimastigotes of Trypanosoma cruzi were 11 microM (1), 36 microM (2), 34 microM (3), 34 microM (4), 66 microM (5), 66 microM (6), and >265 microM (7).
Labdane-type diterpenes as new cell cycle inhibitors and apoptosis inducers from Vitex trifolia L.[Pubmed:15621610]
J Asian Nat Prod Res. 2005 Apr;7(2):95-105.
Five labdane-type diterpenes, Vitexilactone (1), (rel 5S,6R,8R,9R,10S)-6-acetoxy-9-hydroxy-13(14)-labden-16,15-olide (2), rotundifuran (3), vitetrifolin D (4), and vitetrifolin E (5), have been isolated from Vitex trifolia L., a Chinese folk medicine used to treat cancers, as new cell cycle inhibitors and apoptosis inducers through a bioassay-guided separation procedure and were identified by spectroscopic methods. Compounds 1-5 dramatically induced apoptosis both on tsFT210 and K562 cells at higher concentrations while at lower concentrations they inhibited the cell cycle progression of both tsFT210 and K562 cells at the G0/G1 phase. MIC values for 1-5 for inducing apoptosis and concentration regions for 1-5 for inhibiting cell cycle both on tsFT210 and K562 cells have also been determined. Furthermore, the inhibitory effects of 1-5 on the proliferation of tsFT210 and K562 cells have been evaluated by MTT assay to obtain IC50 values to confirm that 1-5 are anticancer components of Vitex trifolia L., which exert their anti-proliferative effect on cancer cells through inducing apoptosis and inhibiting the cell cycle. The present results provide labdane-type diterpenes, 1-5, as a new class of cell cycle inhibitors and compounds 1, 2, 4, and 5 as new apoptosis inducers, which also explains, for the first time, the usage of Vitex trifolia L. by Chinese people to treat cancers.
Virtual screening of Indonesian herbal database as HIV-1 reverse transcriptase inhibitor.[Pubmed:23275721]
Bioinformation. 2012;8(24):1206-10.
HIV-1 (Human immunodeficiency virus type 1) is a member of retrovirus family that could infect human and causing AIDS disease. AIDS epidemic is one of most destructive diseases in modern era. There were more than 33 million people infected by HIV until 2010. Various studies have been widely employed to design drugs that target the essential enzymes of HIV-1 that is, reverse transcriptase, protease and integrase. In this study, in silico virtual screening approach is used to find lead molecules from the library or database of natural compounds as HIV-1 reverse transcriptase inhibitor. Virtual screening against Indonesian Herbal Database using AutoDock4 performed on HIV-1 reverse transcriptase. From the virtual screening, top ten compounds were mulberrin, plucheoside A, Vitexilactone, brucine N-oxide, cyanidin 3-arabinoside, alpha-mangostin, guaijaverin, erycristagallin, morusin and sanggenol N.
Development and validation of a rapid ultra-high performance liquid chromatography diode array detector method for Vitex agnus-castus.[Pubmed:23522912]
J Chromatogr B Analyt Technol Biomed Life Sci. 2013 May 15;927:181-90.
A rapid ultra-high performance liquid chromatography diode array detector (UHPLC-DAD) method was developed and validated for the simultaneous determination of all classes of non-volatile phytochemicals (iridoids, flavonoids and diterpenes) in Vitex agnus-castus (Lamiaceae) fruits, a traditional medicinal plant used against premenstrual symptoms (PMS) and other disorders. Seven marker compounds, 3,4-dihydroxybenzoic acid, p-hydroxybenzoic acid, agnuside, 5-hydroxykaempferol-3,6,7,4'-tetramethylether, 1,2-dibenzoic acid glucose, methoxy-Vitexilactone, and vitetrifolin D were isolated from the methanol extract of V. agnus-castus to be used as reference substances. Chromatographic separation was performed on a Zorbax Eclipse XDB-C18 (50mmx2.1mm) UHPLC column with 1.8mum particle size, within 20min. A solvent gradient from 0.5% acetic acid to acetonitrile at a flow rate of 0.6mL/min was used as mobile phase. Analyte detection and quantification was realized at 210nm and 260nm. The UHPLC-DAD assay was validated for the quantitative analysis of agnuside, isovitexin, casticin, 5-hydroxykaempferol-3,6,7,4'-tetramethylether and vitetrifolin D. It was found to be specific, accurate, precise, and reproducible for the quantification of these compound within a concentration range of 0.7-500.0mug/mL for casticin and 5-hydroxykaempferol-3,6,7,4'-tetramethylether, 1.4-1000.0mug/mL for isovitexin and agnuside, and 12.4-1000.0mug/mL for vitetrifolin D. Intra- and inter-day variations showed relative standard deviations (RSD) of less than 3.9% and 6.4%, respectively. Tentatively assignment of 62 chromatographic features found in the UHPLC-DAD assay was carried out by coupling the UHPLC instrument to a quadrupole time-of-flight mass spectrometer via an electrospray ionization interface (ESI-QTOF-MS) operated in positive and negative ion mode. By using the established quantitative UHPLC-DAD assay to asses agnuside, isovitexin, casticin, 5-hydroxykaempferol-3,6,7,4'-tetramethylether and vitetrifolin D in V. agnus-castus derived preparations as extracts, tinctures and tablets, the applicability of the developed assay to phytopharmaceuticals was successfully proven.
Compounds from the Fruits of the Popular European Medicinal Plant Vitex agnus-castus in Chemoprevention via NADP(H):Quinone Oxidoreductase Type 1 Induction.[Pubmed:23662135]
Evid Based Complement Alternat Med. 2013;2013:432829.
As part of our continuing efforts in the search for potential biologically active compounds from medicinal plants, we have isolated 18 compounds including two novel nitrogen containing diterpenes from extracts of the fruits of Vitex agnus-castus. These isolates, along with our previously obtained novel compound vitexlactam A (1), were evaluated for potential biological effects, including cancer chemoprevention. Chemically, the nitrogenous isolates were found to be two labdane diterpene alkaloids, each containing an alpha , beta -unsaturated gamma -lactam moiety. Structurally, they were elucidated to be 9 alpha -hydroxy-13(14)-labden-16,15-amide (2) and 6 beta -acetoxy-9 alpha -hydroxy-13(14)-labden-15,16-amide (3), which were named vitexlactams B and C, respectively. The 15 known isolates were identified as Vitexilactone (4), rotundifuran (5), 8-epi-manoyl oxide (6), vitetrifolin D (7), spathulenol (8), cis-dihydro-dehydro-diconiferylalcohol-9-O- beta -D-glucoside (9), luteolin-7-O-glucoside (10), 5-hydroxy-3,6,7,4'-tetramethoxyflavone (11), casticin (12), artemetin (13), aucubin (14), agnuside (15), beta -sitosterol (16), p-hydroxybenzoic acid (17), and p-hydroxybenzoic acid glucose ester (18). All compound structures were determined/identified on the basis of 1D and/or 2D NMR and mass spectrometry techniques. Compounds 6, 8, 9, and 18 were reported from a Vitex spieces for the first time. The cancer chemopreventive potentials of these isolates were evaluated for NADP(H):quinone oxidoreductase type 1 (QR1) induction activity. Compound 7 demonstrated promising QR1 induction effect, while the new compound vitexlactam (3) was only slightly active.


