PimavanserinCAS# 706779-91-1 |
- IPI-145 (INK1197)
Catalog No.:BCC1104
CAS No.:1201438-56-3
- IC-87114
Catalog No.:BCC1161
CAS No.:371242-69-2
- PI-103
Catalog No.:BCC1162
CAS No.:371935-74-9
- PIK-75
Catalog No.:BCC1163
CAS No.:372196-77-5
- TGX-221
Catalog No.:BCC1244
CAS No.:663619-89-4
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 706779-91-1 | SDF | Download SDF |
| PubChem ID | 10071196 | Appearance | Powder |
| Formula | C25H34FN3O2 | M.Wt | 427.55 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | ACP-103 | ||
| Solubility | DMSO : 50 mg/mL (116.95 mM; Need ultrasonic) | ||
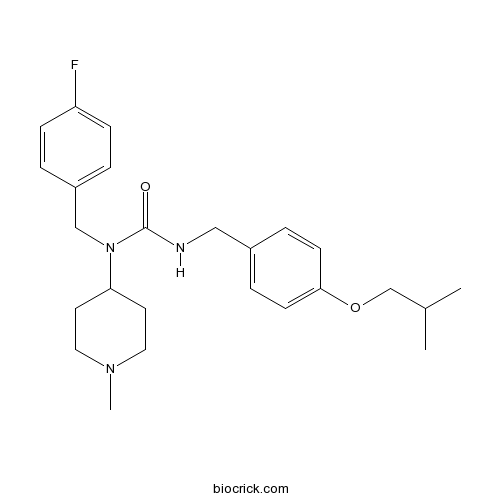
| Chemical Name | 1-[(4-fluorophenyl)methyl]-1-(1-methylpiperidin-4-yl)-3-[[4-(2-methylpropoxy)phenyl]methyl]urea | ||
| SMILES | CC(C)COC1=CC=C(C=C1)CNC(=O)N(CC2=CC=C(C=C2)F)C3CCN(CC3)C | ||
| Standard InChIKey | RKEWSXXUOLRFBX-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C25H34FN3O2/c1-19(2)18-31-24-10-6-20(7-11-24)16-27-25(30)29(23-12-14-28(3)15-13-23)17-21-4-8-22(26)9-5-21/h4-11,19,23H,12-18H2,1-3H3,(H,27,30) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Pimavanserin is a potent 5-hydroxytryptamine (5-HT)2A receptor inverse agonist, displays potent inverse agonist activity in the cell-based functional assay receptor selection and amplification technology (R-SAT), with a mean pIC50 of 8.7.In Vitro:Pimavanserin (ACP-103) competitively antagonizes the binding of [3H]ketanserin to heterologously expressed human 5-HT2A receptors with a mean pKi of 9.3 in membranes and 9.70 in whole cells. Pimavanserin demonstrates lesser affinity (mean pKi of 8.80 in membranes and 8.00 in whole cells, as determined by radioligand binding) and potency as an inverse agonist (mean pIC50 7.1 in R-SAT) at human 5-HT2C receptors, and lacked affinity and functional activity at 5-HT2B receptors, dopamine D2 receptors, and other human monoaminergic receptors[1]. Pimavanserin (ACP-103) is highly selective for 5-HT2A receptors, lacking affinity for other receptors in a broad profile screen including 65 different molecular targets; the only other receptor for which Pimavanserin demonstrates affinity is 5-HT2C, and Pimavanserin is approximately 30-fold selective for 5-HT2A receptors over 5-HT2C receptors depending on the assay[2].In Vivo:Pimavanserin (ACP-103) is a potent, efficacious, orally active 5-HT2A receptor inverse agonist with a behavioral pharmacological profile consistent with utility as an antipsychotic agent. Pimavanserin attenuates head-twitch behavior (3 mg/kg p.o.), and prepulse inhibition deficits (1-10 mg/kg s.c.) induced by the 5-HT2A receptor agonist (±)-2,5-dimethoxy-4-iodoamphetamine hydrochloride in rats and reduces the hyperactivity induced in mice by the N-methyl-D-aspartate receptor noncompetitive antagonist 5H-dibenzo[a,d]cyclohepten-5,10-imine (dizocilpine maleate; MK-801) (0.1 and 0.3 mg/kg s.c.; 3 mg/kg p.o.), consistent with a 5-HT2A receptor mechanism of action in vivo and antipsychotic-like efficacy. Pimavanserin demonstrates >42.6% oral bioavailability in rats[1]. References: | |||||

Pimavanserin Dilution Calculator

Pimavanserin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.3389 mL | 11.6945 mL | 23.3891 mL | 46.7782 mL | 58.4727 mL |
| 5 mM | 0.4678 mL | 2.3389 mL | 4.6778 mL | 9.3556 mL | 11.6945 mL |
| 10 mM | 0.2339 mL | 1.1695 mL | 2.3389 mL | 4.6778 mL | 5.8473 mL |
| 50 mM | 0.0468 mL | 0.2339 mL | 0.4678 mL | 0.9356 mL | 1.1695 mL |
| 100 mM | 0.0234 mL | 0.1169 mL | 0.2339 mL | 0.4678 mL | 0.5847 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Pimavanserin(ACP-103) is a potent and selective 5-HT2A receptor inverse agonist with mean pIC50 of with 8.7 in the cell-based functional assay.
- Canniprene
Catalog No.:BCN4271
CAS No.:70677-47-3
- Catharanthine Sulfate
Catalog No.:BCN3859
CAS No.:70674-90-7
- Z-D-Lys-OH
Catalog No.:BCC2761
CAS No.:70671-54-4
- Anisotropine Methylbromide; Octatropine Methylbromide
Catalog No.:BCC8120
CAS No.:70642-90-9
- Boc-D-Tyr-OH
Catalog No.:BCC3463
CAS No.:70642-86-3
- Phlorizin dihydrate
Catalog No.:BCN2584
CAS No.:7061-54-3
- 14beta-Benzoyloxy-2-deacetylbaccatin VI
Catalog No.:BCN1373
CAS No.:705973-69-9
- 19alpha-Hydroxyfern-7-ene
Catalog No.:BCN7405
CAS No.:70588-12-4
- Chrysoobtusin
Catalog No.:BCC8309
CAS No.:70588-06-6
- Obtusin
Catalog No.:BCC8223
CAS No.:70588-05-5
- Shizukanolide A
Catalog No.:BCN8021
CAS No.:70578-36-8
- Yunaconitine
Catalog No.:BCN6261
CAS No.:70578-24-4
- 3-Chlorotyrosine
Catalog No.:BCC2639
CAS No.:70680-93-2
- H-D-Ala(3-pyridyl)-OH.HCl
Catalog No.:BCC3323
CAS No.:70702-47-5
- Flurofamide
Catalog No.:BCC5660
CAS No.:70788-28-2
- 2',3'-Dihydroxy-4'-methoxyacetophenone
Catalog No.:BCN7168
CAS No.:708-53-2
- Doxapram hydrochloride monohydrate
Catalog No.:BCC8953
CAS No.:7081-53-0
- FPH1 (BRD-6125)
Catalog No.:BCC5342
CAS No.:708219-39-0
- Panaxydiol
Catalog No.:BCN3702
CAS No.:708257-91-4
- JNJ 10397049
Catalog No.:BCC6139
CAS No.:708275-58-5
- Chicoric acid
Catalog No.:BCN1215
CAS No.:70831-56-0
- Cyanidin-3-O-glucoside chloride
Catalog No.:BCN1230
CAS No.:7084-24-4
- Chlorothiazide Sodium
Catalog No.:BCC5628
CAS No.:7085-44-1
- Troxerutin
Catalog No.:BCN3828
CAS No.:7085-55-4
Pimavanserin: A Novel Antipsychotic for Parkinson's Disease Psychosis.[Pubmed:28375643]
Ann Pharmacother. 2017 Jun;51(6):479-487.
OBJECTIVE: To review the pharmacology, pharmacokinetics, efficacy, safety, and place in therapy of Pimavanserin for the treatment of hallucinations and delusions of Parkinson's disease psychosis (PDP). DATA SOURCES: A comprehensive PubMed search (1966 to January 2017) was conducted using the search terms Parkinson's disease psychosis, hallucinations, delusions, Pimavanserin, and ACP-103. Additional data were obtained from references of identified articles, governmental sources, manufacturer product labeling and website, and Clinicaltrials.gov. STUDY SELECTION AND DATA EXTRACTION: All English-language trials evaluating Pimavanserin in PDP were included. Data from review articles were included if relevant to clinical practice. One phase II and 3 phase III trials are discussed. DATA SYNTHESIS: Pimavanserin was approved in April 2016 for the treatment of delusions and hallucinations of PDP. One phase II and 2 phase III trials reported no difference for primary outcomes when Pimavanserin was compared with placebo. The pivotal phase III ACP-103-020 trial adapted a scale to target more specific symptoms prevalent in PDP and showed that least-squares mean differences of the total PD-adapted Scale for the Assessment of Positive Symptoms score were significantly improved for Pimavanserin-treated patients as compared with placebo-treated patients (difference = -3.06; 95% CI [-4.91 to -1.20]; P = 0.0014]). Pimavanserin's adverse effect profile includes urinary tract infections, falls, peripheral edema, hallucinations, confusion, nausea, and headaches. CONCLUSION: Pimavanserin is a novel 5-HT2A inverse agonist that has shown promising results for managing hallucinations and delusions in patients with PDP without worsening motor effects or orthostasis. Yet its high cost and specialty pharmacy access may limit use in clinical practice.
Evidence for the use of pimavanserin in the treatment of Parkinson's disease psychosis.[Pubmed:27800022]
Ther Adv Neurol Disord. 2016 Nov;9(6):462-473.
Parkinson's disease (PD) is a progressive neurodegenerative disorder with both motor and nonmotor symptoms (NMS), leading to significant morbidity and caregiver burden. Psychosis is common but is under recognized by physicians. When present, it increases the patient's risk of hospitalization and nursing home placement and caregiver burden. Although the atypical antipsychotic agent, clozapine, has been considered the gold standard treatment, severe agranulocytosis in 0.38% of patients and more commonly milder leukopenia, resulting in frequent blood testing, limit its use. Pimavanserin, a 5HT2A receptor inverse agonist, has been shown to reduce psychosis in PD without worsening motor symptoms. It is therefore a welcome therapeutic option for this devastating NMS.
Update on the treatment of Parkinson's disease psychosis: role of pimavanserin.[Pubmed:28331324]
Neuropsychiatr Dis Treat. 2017 Mar 8;13:737-744.
Parkinson's disease (PD) has a prevalence of nearly 1 million people in the USA, with increasing incidence in the elderly population. Generally, the age of presentation is between 55 and 65 years, with the likelihood of diagnosis increasing as patients reach the age of 80 years or above. Some of the common treatments for PD increase dopamine levels in the brain. Dopaminergic therapy helps to improve motor and non-motor symptoms, but it is not without risks. Dopaminergic therapy can cause confusion, delirium, and psychotic-like behavior. It is recommended that these agents are used cautiously in patients with a history of psychosis due to the risk of exacerbation. It is unclear whether Parkinson's disease psychosis (PDP) is due to the disease itself, the treatment, or a combination of both, but it is clear that a safe, effective treatment is necessary. Second-generation (atypical) antipsychotics are the current choice of therapy for PDP. All of these agents have a black box warning from the US Food and Drug Administration (FDA) for elevated risk of mortality in elderly patients with dementia-related psychosis. Pimavanserin (Nuplazid((R))) received its novel drug approval by the FDA on April 29, 2016, to treat hallucinations and delusions associated with psychosis experienced by some people with PD. We review in this article the new research that led to this approval as well as its potential place in therapy.
Repeated 7-Day Treatment with the 5-HT2C Agonist Lorcaserin or the 5-HT2A Antagonist Pimavanserin Alone or in Combination Fails to Reduce Cocaine vs Food Choice in Male Rhesus Monkeys.[Pubmed:27857126]
Neuropsychopharmacology. 2017 Apr;42(5):1082-1092.
Cocaine use disorder is a global public health problem for which there are no Food and Drug Administration-approved pharmacotherapies. Emerging preclinical evidence has implicated both serotonin (5-HT) 2C and 2A receptors as potential mechanisms for mediating serotonergic attenuation of cocaine abuse-related neurochemical and behavioral effects. Therefore, the present study aim was to determine whether repeated 7-day treatment with the 5-HT2C agonist lorcaserin (0.1-1.0 mg/kg per day, intramuscular; 0.032-0.1 mg/kg/h, intravenous) or the 5-HT2A inverse agonist/antagonist Pimavanserin (0.32-10 mg/kg per day, intramuscular) attenuated cocaine reinforcement under a concurrent 'choice' schedule of cocaine and food availability in rhesus monkeys. During saline treatment, cocaine maintained a dose-dependent increase in cocaine vs food choice. Repeated Pimavanserin (3.2 mg/kg per day) treatments significantly increased small unit cocaine dose choice. Larger lorcaserin (1.0 mg/kg per day and 0.1 mg/kg/h) and Pimavanserin (10 mg/kg per day) doses primarily decreased rates of operant behavior. Coadministration of ineffective lorcaserin (0.1 mg/kg per day) and Pimavanserin (0.32 mg/kg per day) doses also failed to significantly alter cocaine choice. These results suggest that neither 5-HT2C receptor activation nor 5-HT2A receptor blockade are sufficient to produce a therapeutic-like decrease in cocaine choice and a complementary increase in food choice. Overall, these results do not support the clinical utility of 5-HT2C agonists and 5-HT2A inverse agonists/antagonists alone or in combination as candidate anti-cocaine use disorder pharmacotherapies.


