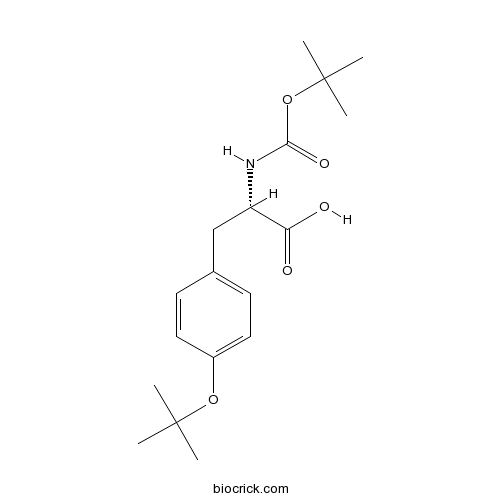
Boc-Tyr(tBu)-OHCAS# 47375-34-8 |
- Cisplatin
Catalog No.:BCN1552
CAS No.:14283-03-5
- Z-VAD-FMK
Catalog No.:BCC1126
CAS No.:187389-52-2
- Z-WEHD-FMK
Catalog No.:BCC1139
CAS No.:210345-00-9
- Z-LEHD-FMK
Catalog No.:BCC5117
CAS No.:210345-04-3
- AZ 10417808
Catalog No.:BCC2356
CAS No.:331645-84-2
- Apoptosis Activator 2
Catalog No.:BCC2099
CAS No.:79183-19-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 47375-34-8 | SDF | Download SDF |
| PubChem ID | 7017901 | Appearance | Powder |
| Formula | C18H27NO5 | M.Wt | 337.4 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (2S)-2-[(2-methylpropan-2-yl)oxycarbonylamino]-3-[4-[(2-methylpropan-2-yl)oxy]phenyl]propanoic acid | ||
| SMILES | CC(C)(C)OC1=CC=C(C=C1)CC(C(=O)O)NC(=O)OC(C)(C)C | ||
| Standard InChIKey | ZEQLLMOXFVKKCN-AWEZNQCLSA-N | ||
| Standard InChI | InChI=1S/C18H27NO5/c1-17(2,3)23-13-9-7-12(8-10-13)11-14(15(20)21)19-16(22)24-18(4,5)6/h7-10,14H,11H2,1-6H3,(H,19,22)(H,20,21)/t14-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Boc-Tyr(tBu)-OH Dilution Calculator

Boc-Tyr(tBu)-OH Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.9638 mL | 14.8192 mL | 29.6384 mL | 59.2768 mL | 74.096 mL |
| 5 mM | 0.5928 mL | 2.9638 mL | 5.9277 mL | 11.8554 mL | 14.8192 mL |
| 10 mM | 0.2964 mL | 1.4819 mL | 2.9638 mL | 5.9277 mL | 7.4096 mL |
| 50 mM | 0.0593 mL | 0.2964 mL | 0.5928 mL | 1.1855 mL | 1.4819 mL |
| 100 mM | 0.0296 mL | 0.1482 mL | 0.2964 mL | 0.5928 mL | 0.741 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Boc-Tyr(tBu)-OH
- SCH 563705
Catalog No.:BCC1933
CAS No.:473728-58-4
- SCH 527123
Catalog No.:BCC1932
CAS No.:473727-83-2
- AMG 487
Catalog No.:BCC5140
CAS No.:473719-41-4
- Boc-Trp(For)-OH
Catalog No.:BCC3456
CAS No.:47355-10-2
- Carpinontriol B
Catalog No.:BCN8113
CAS No.:473451-73-9
- Spegatrine
Catalog No.:BCN4068
CAS No.:47326-53-4
- Betulin
Catalog No.:BCN5528
CAS No.:473-98-3
- Tolbutamide Sodium
Catalog No.:BCC5632
CAS No.:473-41-6
- beta-Eudesmol
Catalog No.:BCN6294
CAS No.:473-15-4
- alpha-Cyperone
Catalog No.:BCN1193
CAS No.:473-08-5
- SB 366791
Catalog No.:BCC7128
CAS No.:472981-92-3
- 8(14),15-Isopimaradien-3-ol
Catalog No.:BCN5526
CAS No.:4728-30-7
- Lersivirine
Catalog No.:BCC1698
CAS No.:473921-12-9
- Forskolin G
Catalog No.:BCN5527
CAS No.:473981-11-2
- Brazilin
Catalog No.:BCN5529
CAS No.:474-07-7
- Chenodeoxycholic acid
Catalog No.:BCN2620
CAS No.:474-25-9
- Citrostadienol
Catalog No.:BCN7357
CAS No.:474-40-8
- Reserpin N-oxide
Catalog No.:BCN3493
CAS No.:474-48-6
- Daucosterol
Catalog No.:BCN5531
CAS No.:474-58-8
- Campestanol
Catalog No.:BCN3890
CAS No.:474-60-2
- Campesterol
Catalog No.:BCN3181
CAS No.:474-62-4
- Brassicasterol
Catalog No.:BCN2613
CAS No.:474-67-9
- CAL-130 Racemate
Catalog No.:BCC1442
CAS No.:474012-90-3
- N-Methylsarpagine methosalt
Catalog No.:BCN5530
CAS No.:47418-70-2
Cyclization studies with tetra- and pentapeptide sequences corresponding to beta-casomorphins.[Pubmed:1917307]
Int J Pept Protein Res. 1991 Jun;37(6):502-7.
The tetrapeptide Boc-D-Orn-Phe-D-Pro-Gly-OH and the pentapeptide sequence Boc-Tyr(tBu)-D-Orn-Phe-D-Pro-Gly-OH were used to study the influence of different coupling reagents on the yield and purity of these model peptides. The simple structure prevented racemization and cyclodimerization and facilitated the ring formation. The most favorable effects on yield and purity were obtained in both reactions using diphenyl-phosphoryl azide (DPPA) and norborn-5-ene-2,3-dicarboximidodiphenylphosphate (NDPP), while the cyclizations with the powerful activating reagents benzotriazol-1-yl-oxy-tris(dimethylamino)-phosphonium hexafluorophosphate (BOP) and 2-(1-H-benzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate (HBTU) with the exception of the cyclopentapeptide reaction with HBTU/4-dimethylaminopyridine gave unsatisfactory results.
A convenient incorporation of conformationally constrained 5,5-dimethylproline into the ribonuclease A 89-124 sequence by condensation of synthetic peptide fragments.[Pubmed:12558949]
J Pept Res. 2003 Mar;61(3):140-51.
The presence of l-5,5-dimethylproline (dmP) within an amino acid sequence results in the formation of an X-dmP peptide bond predominantly locked in a cis conformation. However, the common use of this unnatural amino acid has been hampered by the difficulty of the economical incorporation of the dmP residue into longer peptide segments due to the steric hindrance imposed by the dimethyl moieties. Here, we describe synthesis of the C-terminal 36-residue peptide, corresponding to the 89-124 sequence of bovine pancreatic ribonuclease A (RNase A), in which dmP is incorporated as a substitute for Pro93. The peptide was assembled by condensation of protected 5- and 31-residue peptide fragments, which were synthesized by solid-phase peptide methodology using fluorenylmethyloxycarbonyl chemistry. We focused on optimizing the synthesis of the Fmoc-Ser(tBu)-Ser(tBu)-Lys(Boc)-Tyr(tBu)-dmP-OH pentapeptide (residues 89-93) with efficient acylation of the sterically hindered dmP residue. In a comparative study, the reagent O-(7-azabenzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate was found to be superior to bromo-tris-pyrrolidino-phosphonium hexafluorophosphate and tetramethylfluoroformamidinium hexafluorophosphate for the synthesis of the -Tyr(tBu)-dmP- peptide bond in solution as well as on a resin.
A structure-activity relationship study and combinatorial synthetic approach of C-terminal modified bifunctional peptides that are delta/mu opioid receptor agonists and neurokinin 1 receptor antagonists.[Pubmed:18266313]
J Med Chem. 2008 Mar 13;51(5):1369-76.
A series of bifunctional peptides with opioid agonist and substance P antagonist bioactivities were designed with the concept of overlapping pharmacophores. In this concept, the bifunctional peptides were expected to interact with each receptor separately in the spinal dorsal horn where both the opioid receptors and the NK1 receptors were found to be expressed, to show an enhanced analgesic effect, no opioid-induced tolerance, and to provide better compliance than coadministration of two drugs. Compounds were synthesized using a two-step combinatorial method for C-terminal modification. In the method, the protected C-terminal-free carboxyl peptide, Boc-Tyr( tBu)- d-Ala-Gly Phe-Pro-Leu-Trp(Boc)-OH, was synthesized as a shared intermediate using Fmoc solid phase chemistry on a 2-chlorotrityl resin. This intermediate was esterified or amidated in solution phase. The structure-activity relationships (SAR) showed that the C-terminus acted as not only a critical pharmacophore for the substance P antagonist activities, but as an address region for the opioid agonist pharmacophore that is structurally distant from the C-terminal. Among the peptides, H-Tyr- d -Ala-Gly-Phe-Pro-Leu-Trp-NH-Bzl ( 3) demonstrated high binding affinities at both delta and mu receptors ( K i = 10 and 0.65 nM, respectively) with efficient agonist functional activity in the mouse isolated vas deferens (MVD) and guinea pig isolated ileum (GPI) assays (IC 50 = 50 and 13 nM, respectively). Compound 3 also showed a good antagonist activity in the GPI assay with substance P stimulation ( K e = 26 nM) and good affinity for the hNK1 receptor ( K i = 14 nM). Consequently, compound 3 is expected to be a promising and novel type of analgesic with bifunctional activities.
Cyclic morphiceptin analogs: cyclization studies and opioid activities in vitro.[Pubmed:8985782]
Int J Pept Protein Res. 1996 Dec;48(6):495-502.
Attempts were undertaken to develop cyclic beta-casomorphin-5 analogs with improved opioid activity profiles by deletion of the glycine residue in position 5, leading to analogs structurally related to the opioid peptide morphiceptin (H-Tyr-Pro-Phe-Pro-NH2). The tetrapeptide sequence Boc-Tyr(tBu)-D-Xaa-Phe-Yaa-OH (Xaa = Lys, Orn, A2bu; Yaa = Pro in L- or D-configuration) was used to study the influence of ring size and chirality on the yield of cyclization between the omega-amino group of Xaa and the C-terminal carboxyl group. In all cases the cyclization reaction was performed under identical experimental conditions to allow a direct comparison with regard to yield and homogeneity. The reaction products were purified by crystallization and liquid chromatography, and were characterized by HPLC, TLC, electrospray mass spectrometry and 1H-NMR spectroscopy. In none of the reactions performed with the cyclization precursors containing proline in the L-configuration could a cyclic monomer be detected, and the cyclodimer (7-9) was the exclusive product in each case. Cyclodimerization was also the favored reaction in the attempted formation of the 11-membered ring of the cyclic [D-A2bu2, D-Pro4]-morphiceptin analog 12, since only traces of the monomer were found. In the case of both the [D-Lys2, D-Pro4]-analog 10 and the [D-Orn2, D-Pro4]-analog 11, the cyclomonomer/cyclodimer ratio was about 80:20. The cyclic monomers 10 and 11 showed high opioid activity in the mu-receptor-representative guinea pig ileum assay (IC50 = 2-5 nM) and in the delta-receptor representative mouse vas deferens assay (IC50 = 50-60 nM), whereas the potency of the cyclodimers was 2-3 orders of magnitude lower in both in vitro bioassays.


