Axitinib (AG 013736)VEGFR1/ c-Kit inhibitor CAS# 319460-85-0 |
- Ki8751
Catalog No.:BCC1116
CAS No.:228559-41-9
- Telatinib (BAY 57-9352)
Catalog No.:BCC3879
CAS No.:332012-40-5
- Pazopanib (GW-786034)
Catalog No.:BCC1286
CAS No.:444731-52-6
- Dovitinib Dilactic acid
Catalog No.:BCC3771
CAS No.:852433-84-2
- RAF265
Catalog No.:BCC3677
CAS No.:927880-90-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 319460-85-0 | SDF | Download SDF |
| PubChem ID | 6450551 | Appearance | Powder |
| Formula | C22H18N4OS | M.Wt | 386.47 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | AG-013736 | ||
| Solubility | DMSO : 20.83 mg/mL (53.90 mM; Need ultrasonic) H2O : < 0.1 mg/mL (insoluble) | ||
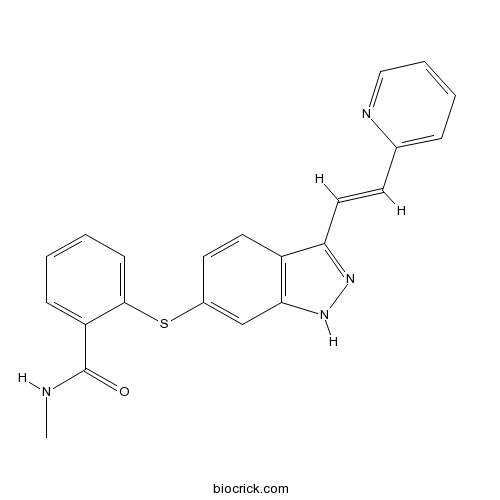
| Chemical Name | N-methyl-2-[[3-[(E)-2-pyridin-2-ylethenyl]-1H-indazol-6-yl]sulfanyl]benzamide | ||
| SMILES | CNC(=O)C1=CC=CC=C1SC2=CC3=C(C=C2)C(=NN3)C=CC4=CC=CC=N4 | ||
| Standard InChIKey | RITAVMQDGBJQJZ-FMIVXFBMSA-N | ||
| Standard InChI | InChI=1S/C22H18N4OS/c1-23-22(27)18-7-2-3-8-21(18)28-16-10-11-17-19(25-26-20(17)14-16)12-9-15-6-4-5-13-24-15/h2-14H,1H3,(H,23,27)(H,25,26)/b12-9+ | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Potent inhibitor of VEGFR-2, -3, and -1 (IC50 values are 0.2, 0.1-0.3, and 1.2 nM respectively). Exhibits minimal activity against a panel of ~100 protein kinase. Inhibits angiogenesis and vascular permeability. Also a high affinity BCR-ABL1 (T315I) inhibitor (Ki = 149 pM for autophosphorylated ABL1 (T315I)). Blocks proliferation of Ba/F3 cells expressing BCR-ABL1 (T315I). Orally available. |

Axitinib (AG 013736) Dilution Calculator

Axitinib (AG 013736) Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5875 mL | 12.9376 mL | 25.8752 mL | 51.7505 mL | 64.6881 mL |
| 5 mM | 0.5175 mL | 2.5875 mL | 5.175 mL | 10.3501 mL | 12.9376 mL |
| 10 mM | 0.2588 mL | 1.2938 mL | 2.5875 mL | 5.175 mL | 6.4688 mL |
| 50 mM | 0.0518 mL | 0.2588 mL | 0.5175 mL | 1.035 mL | 1.2938 mL |
| 100 mM | 0.0259 mL | 0.1294 mL | 0.2588 mL | 0.5175 mL | 0.6469 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Axitinib is a selective and oral inhibitor of vascular endothelial growth factor (VEGF) receptor tyrosine kinases 1, 2 and 3 with [1].
Axitinib inhibited the phosphorylation of VEGFR-1, 2 and 3 with IC50 values of 1.2 nM, 0.2 nM and 0.1 to 0.3 nM in cells, respectively. In HUVEC cells, Axitinib inhibited VEGFR-2 stimulated cell survival with about 1000-fold selectivity against FGFR-1. Axitinib also significantly suppressed the phosphorylation of VEGF downstream signaling molecules including Akt, eNOS and ERK1/2. Besides that, axitinib inhibited VEGFR-2 phosphorylation with EC50 value of 0.49 nM in vivo. It delayed tumore growth of human xenograft tumors in mice such as M24met, HCT-116 and SN12C [1].
References:
[1] Hu-Lowe D D, Zou H Y, Grazzini M L, et al. Nonclinical antiangiogenesis and antitumor activities of axitinib (AG-013736), an oral, potent, and selective inhibitor of vascular endothelial growth factor receptor tyrosine kinases 1, 2, 3. Clinical Cancer Research, 2008, 14(22): 7272-7283.
- (RS)-3-Hydroxyphenylglycine
Catalog No.:BCC6604
CAS No.:31932-87-3
- H-D-Allo-Ile-OH
Catalog No.:BCC2683
CAS No.:319-78-8
- Moricizine
Catalog No.:BCC5235
CAS No.:31883-05-3
- Methyl orsellinate
Catalog No.:BCN5238
CAS No.:3187-58-4
- H-Orn-OH. HCl
Catalog No.:BCC3000
CAS No.:3184-13-2
- H-Phenylalaninol
Catalog No.:BCC2719
CAS No.:3182-95-4
- H-Phe-OEt.HCl
Catalog No.:BCC3007
CAS No.:3182-93-2
- Propranolol HCl
Catalog No.:BCC4336
CAS No.:318-98-9
- H-Ala-pNA.HCl
Catalog No.:BCC3195
CAS No.:31796-55-1
- (-)-Lyoniresinol
Catalog No.:BCN3488
CAS No.:31768-94-2
- 3-Deoxyaconitine
Catalog No.:BCN2797
CAS No.:3175-95-9
- 2-Methyl-4-(2-methylbenzoylamino)benzoic acid
Catalog No.:BCC8579
CAS No.:317374-08-6
- Cornucervine
Catalog No.:BCN1962
CAS No.:31948-48-8
- Norarmepavine
Catalog No.:BCN7077
CAS No.:3195-01-5
- H-β-Ala-OMe.HCl
Catalog No.:BCC2853
CAS No.:3196-73-4
- Ethyl glucoside
Catalog No.:BCN5239
CAS No.:3198-49-0
- SIB 1757
Catalog No.:BCC6971
CAS No.:31993-01-8
- 5-Azacytidine
Catalog No.:BCC1130
CAS No.:320-67-2
- Aclidinium Bromide
Catalog No.:BCC4575
CAS No.:320345-99-1
- Marmin acetonide
Catalog No.:BCN5240
CAS No.:320624-68-8
- Adenine sulfate
Catalog No.:BCC4451
CAS No.:321-30-2
- Fluoronaphthalene
Catalog No.:BCC8987
CAS No.:321-38-0
- EO 1428
Catalog No.:BCC7511
CAS No.:321351-00-2
- [D-Trp8]-γ-MSH
Catalog No.:BCC7902
CAS No.:321351-81-9
Phase I study of axitinib (AG-013736) in combination with gemcitabine in patients with advanced pancreatic cancer.[Pubmed:21670972]
Invest New Drugs. 2012 Aug;30(4):1531-9.
PURPOSE: Axitinib (AG-013736), an oral, potent, and selective inhibitor of vascular endothelial growth factor (VEGF) receptors 1, 2, and 3, is under investigation for treatment of various solid tumors. The safety and pharmacokinetics of axitinib in combination with gemcitabine in patients with advanced pancreatic cancer was evaluated in the phase I portion of this trial. The randomized phase II portion was reported separately. PATIENTS AND METHODS: Patients with advanced pancreatic cancer who had received no prior chemotherapy were eligible for this study. Pharmacokinetic profiles of the drugs were obtained on cycle (C) 1 day (D) 1 (gemcitabine alone 1,000 mg/m(2)), C1D14 (steady state, axitinib alone 5 mg twice daily [BID]), and C1D15 (gemcitabine plus steady-state axitinib). Adverse events were monitored weekly at the clinic. RESULTS: Eight patients participated in the phase IB portion of the trial. Patients received gemcitabine on D1, D8, and D15 and continuous axitinib in a 28 day-cycle beginning C1D3. There was no dose-limiting toxicity. Common treatment-related adverse events included fatigue, diarrhea, dysphonia, and hypertension. Myelosuppression was similar to gemcitabine monotherapy. No apparent major pharmacokinetic interactions between gemcitabine and axitinib were observed. Of six patients evaluable for efficacy, three had confirmed partial responses. CONCLUSIONS: Axitinib (5 mg BID) and gemcitabine (1,000 mg/m(2)) were well tolerated when administered together, without any pharmacokinetic interactions, and showed encouraging antitumor activity.
Key predictive factors of axitinib (AG-013736)-induced proteinuria and efficacy: a phase II study in Japanese patients with cytokine-refractory metastatic renal cell Carcinoma.[Pubmed:21889330]
Eur J Cancer. 2011 Nov;47(17):2592-602.
BACKGROUND: Axitinib (AG-013736) is an oral, selective and potent inhibitor of vascular endothelial growth factor receptors (VEGFR)-1, 2 and 3. This phase II study investigated axitinib efficacy, safety and biomarkers in Japanese patients with cytokine-refractory metastatic renal cell carcinoma (mRCC). PATIENTS AND METHODS: In an open-label, multicentre study, 64 patients received an axitinib starting dose of 5mg twice daily. RESULTS: Objective response rate (ORR) was 50.0% and median progression-free survival (PFS) was 11.0 months per independent review committee. Common treatment-related adverse events were hypertension (84%; 70% grade >/=3), hand-foot syndrome (75%; 22% grade >/=3) and diarrhoea (64%; 5% grade >/=3). Eighteen patients (28%) developed proteinuria >/=2g/24h and required dose reduction or treatment interruption/discontinuation. Proteinuria was a major cause for treatment discontinuation. Baseline urine protein levels were associated with development of proteinuria >/=2g/24h (hazard ratio [HR]=5.457, P=0.0035 in patients with baseline proteinuria >/=1+ versus <1+). Baseline urine protein levels correlated more strongly with axitinib-related proteinuria than other baseline renal function test values or blood pressure. Patients with greater decreases in soluble VEGFR-2 concentrations had significantly higher ORR and longer PFS than those with smaller decreases (ORR: 64.5% versus 37.5%, P=0.045; median PFS: 12.9 months versus 9.2 months, HR=0.42, P=0.01). CONCLUSIONS: Axitinib showed significant antitumour activity and was well tolerated in Japanese mRCC patients. Baseline proteinuria and soluble VEGFR-2 levels may be key indicators of axitinib-induced proteinuria and efficacy, respectively.
A phase II study of axitinib (AG-013736) in patients with incurable adenoid cystic carcinoma.[Pubmed:27566443]
Ann Oncol. 2016 Oct;27(10):1902-8.
BACKGROUND: Recurrent/metastatic adenoid cystic carcinoma (ACC) is an incurable disease with no standard treatments. The majority of ACCs express the oncogenic transcription factor MYB (also c-myb), often in the context of a MYB gene rearrangement. This phase II trial of the tyrosine kinase inhibitor (TKI) axitinib (Pfizer) tested the hypothesis that targeting pathways activated by MYB can be therapeutically effective for ACC. PATIENTS AND METHODS: This is a minimax two-stage, phase II trial that enrolled patients with incurable ACC of any primary site. Progressive or symptomatic disease was required. Patients were treated with axitinib 5 mg oral twice daily; dose escalation was allowed. The primary end point was best overall response (BOR). An exploratory analysis correlating biomarkers to drug benefit was conducted, including next-generation sequencing (NGS) in 11 patients. RESULTS: Thirty-three patients were registered and evaluable for response. Fifteen patients had the axitinib dose increased. Tumor shrinkage was achieved in 22 (66.7%); 3 (9.1%) had confirmed partial responses. Twenty-five (75.8%) patients had stable disease, 10 of whom had disease stability for >6 months. The median progression-free survival (PFS) was 5.7 months (range 0.92-21.8 months). Grade 3 axitinib-related toxicities included hypertension, oral pain and fatigue. A trend toward superior PFS was noted with the MYB/NFIB rearrangement, although this was not statistically significant. NGS revealed three tumors with 4q12 amplification, producing increased copies of axitinib-targeted genes PDGFR/KDR/KIT. Two 4q12 amplified patients achieved stable disease for >6 months, including one with significant tumor reduction and the longest PFS on study (21.8 months). CONCLUSIONS: Although the primary end point was not met, axitinib exhibited clinical activity with tumor shrinkage achieved in the majority of patients with progressive disease before trial enrollment. Analysis of MYB biomarkers and genomic profiling suggests the hypothesis that 4q12 amplified ACCs are a disease subset that benefit from TKI therapy.
Axitinib (AG-013736), an oral specific VEGFR TKI, shows potential therapeutic utility against cholangiocarcinoma.[Pubmed:24755544]
Jpn J Clin Oncol. 2014 Jun;44(6):570-8.
OBJECTIVE: Cholangiocarcinoma is a refractory cancer whose incidence has been increasing worldwide in recent years. Neoangiogenesis plays an important role in the growth of various solid cancers, including cholangiocarcinoma. Vascular endothelial growth factor plays an important role in tumor-induced angiogenesis and its expression is associated with the progression and prognosis of cholangiocarcinoma. This study examined whether axitinib (AG-013736, INLYTA((R))), a potent and selective second-generation inhibitor of vascular endothelial growth factor receptors 1, 2 and 3, could be a potentially useful therapeutic agent for cholangiocarcinoma. METHODS: We performed expression profiling of angiogenesis-related molecules in eight cholangiocarcinoma cell lines and found that three of them showed high vascular endothelial growth factor expression. Among them, we examined the in vivo anti-tumor effect of axitinib on NCC-BD1 (a gemcitabine-sensitive extra-hepatic cholangiocarcinoma cell line) and TKKK (a gemcitabine-resistant intra-hepatic cholangiocarcinoma cell line) using subcutaneous xenograft models. RESULTS: Oral administration of axitinib significantly inhibited the growth of TKKK xenografts at a dose of 6 mg kg(-1) day(-1) (P<0.05), and the growth of NCC-BD1 xenografts at 30 mg kg(-1)day(-1) (P<0.05). Treated tumors showed a significant decrease of microvessel density and the tumor cell proliferation index and a mild but significant increase of the apoptotic index in comparison with untreated tumors. CONCLUSIONS: Our results suggest that axitinib should be a promising therapy for vascular endothelial growth factor-expressing cholangiocarcinoma, irrespective of tumor origin and gemcitabine sensitivity.
Axitinib effectively inhibits BCR-ABL1(T315I) with a distinct binding conformation.[Pubmed:25686603]
Nature. 2015 Mar 5;519(7541):102-5.
The BCR-ABL1 fusion gene is a driver oncogene in chronic myeloid leukaemia and 30-50% of cases of adult acute lymphoblastic leukaemia. Introduction of ABL1 kinase inhibitors (for example, imatinib) has markedly improved patient survival, but acquired drug resistance remains a challenge. Point mutations in the ABL1 kinase domain weaken inhibitor binding and represent the most common clinical resistance mechanism. The BCR-ABL1 kinase domain gatekeeper mutation Thr315Ile (T315I) confers resistance to all approved ABL1 inhibitors except ponatinib, which has toxicity limitations. Here we combine comprehensive drug sensitivity and resistance profiling of patient cells ex vivo with structural analysis to establish the VEGFR tyrosine kinase inhibitor axitinib as a selective and effective inhibitor for T315I-mutant BCR-ABL1-driven leukaemia. Axitinib potently inhibited BCR-ABL1(T315I), at both biochemical and cellular levels, by binding to the active form of ABL1(T315I) in a mutation-selective binding mode. These findings suggest that the T315I mutation shifts the conformational equilibrium of the kinase in favour of an active (DFG-in) A-loop conformation, which has more optimal binding interactions with axitinib. Treatment of a T315I chronic myeloid leukaemia patient with axitinib resulted in a rapid reduction of T315I-positive cells from bone marrow. Taken together, our findings demonstrate an unexpected opportunity to repurpose axitinib, an anti-angiogenic drug approved for renal cancer, as an inhibitor for ABL1 gatekeeper mutant drug-resistant leukaemia patients. This study shows that wild-type proteins do not always sample the conformations available to disease-relevant mutant proteins and that comprehensive drug testing of patient-derived cells can identify unpredictable, clinically significant drug-repositioning opportunities.
Nonclinical antiangiogenesis and antitumor activities of axitinib (AG-013736), an oral, potent, and selective inhibitor of vascular endothelial growth factor receptor tyrosine kinases 1, 2, 3.[Pubmed:19010843]
Clin Cancer Res. 2008 Nov 15;14(22):7272-83.
PURPOSE: Axitinib (AG-013736) is a potent and selective inhibitor of vascular endothelial growth factor (VEGF) receptor tyrosine kinases 1 to 3 that is in clinical development for the treatment of solid tumors. We provide a comprehensive description of its in vitro characteristics and activities, in vivo antiangiogenesis, and antitumor efficacy and translational pharmacology data. EXPERIMENTAL DESIGN: The potency, kinase selectivity, pharmacologic activity, and antitumor efficacy of axitinib were assessed in various nonclinical models. RESULTS: Axitinib inhibits cellular autophosphorylation of VEGF receptors (VEGFR) with picomolar IC(50) values. Counterscreening across multiple kinase and protein panels shows it is selective for VEGFRs. Axitinib blocks VEGF-mediated endothelial cell survival, tube formation, and downstream signaling through endothelial nitric oxide synthase, Akt and extracellular signal-regulated kinase. Following twice daily oral administration, axitinib produces consistent and dose-dependent antitumor efficacy that is associated with blocking VEGFR-2 phosphorylation, vascular permeability, angiogenesis, and concomitant induction of tumor cell apoptosis. Axitinib in combination with chemotherapeutic or targeted agents enhances antitumor efficacy in many tumor models compared with single agent alone. Dose scheduling studies in a human pancreatic tumor xenograft model show that simultaneous administration of axitinib and gemcitabine without prolonged dose interruption or truncation of axitinib produces the greatest antitumor efficacy. The efficacious drug concentrations predicted in nonclinical studies are consistent with the range achieved in the clinic. Although axitinib inhibits platelet-derived growth factor receptors and KIT with nanomolar in vitro potencies, based on pharmacokinetic/pharmacodynamic analysis, axitinib acts primarily as a VEGFR tyrosine kinase inhibitor at the current clinical exposure. CONCLUSIONS: The selectivity, potency for VEGFRs, and robust nonclinical activity may afford broad opportunities for axitinib to improve cancer therapy.
Modulation of the antitumor activity of metronomic cyclophosphamide by the angiogenesis inhibitor axitinib.[Pubmed:18202011]
Mol Cancer Ther. 2008 Jan;7(1):79-89.
The promising but still limited efficacy of angiogenesis inhibitors as monotherapies for cancer treatment indicates a need to integrate these agents into existing therapeutic regimens. Presently, we investigate the antitumor activity of the small-molecule angiogenesis inhibitor axitinib (AG-013736) and its potential for combination with metronomic cyclophosphamide. Axitinib significantly inhibited angiogenesis in rat 9L tumors grown s.c. in scid mice but only moderately delayed tumor growth. Combination of axitinib with metronomic cyclophosphamide fully blocked 9L tumor growth on initiation of drug treatment. In contrast, metronomic cyclophosphamide alone required multiple treatment cycles to halt tumor growth. However, in contrast to the substantial tumor regression that is ultimately induced by metronomic cyclophosphamide, the axitinib/cyclophosphamide combination was tumor growth static. Axitinib did not inhibit hepatic activation of cyclophosphamide or export of its activated metabolite, 4-hydroxy-cyclophosphamide (4-OH-CPA), to extrahepatic tissues; rather, axitinib selectively decreased 9L tumor uptake of 4-OH-CPA by 30% to 40%. The reduced tumor penetration of 4-OH-CPA was associated with a decrease in cyclophosphamide-induced tumor cell apoptosis and a block in the induction of the endogenous angiogenesis inhibitor thrombospondin-1 in tumor-associated host cells, which may contribute to the absence of tumor regression with the axitinib/cyclophosphamide combination. Finally, axitinib transiently increased 9L tumor cell apoptosis, indicating that its effects are not limited to the endothelial cell population. These findings highlight the multiple effects that may characterize antiangiogenic agent/metronomic chemotherapy combinations and suggest that careful optimization of drug scheduling and dosages will be required to maximize antitumor responses.


