AurantiamideCAS# 58115-31-4 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 58115-31-4 | SDF | Download SDF |
| PubChem ID | 185904 | Appearance | Powder |
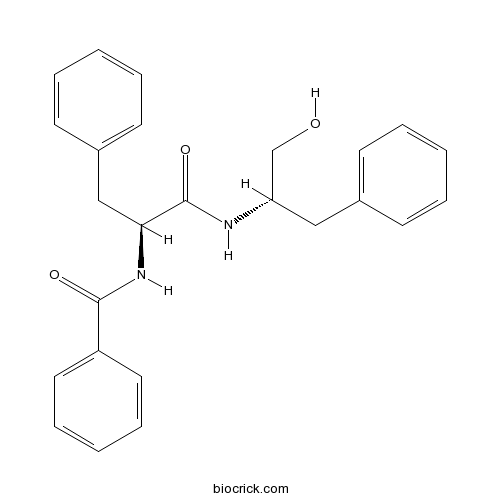
| Formula | C25H26N2O3 | M.Wt | 402.5 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | N-[(2S)-1-[[(2S)-1-hydroxy-3-phenylpropan-2-yl]amino]-1-oxo-3-phenylpropan-2-yl]benzamide | ||
| SMILES | C1=CC=C(C=C1)CC(CO)NC(=O)C(CC2=CC=CC=C2)NC(=O)C3=CC=CC=C3 | ||
| Standard InChIKey | KSVKECXWDNCRTM-GOTSBHOMSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Aurantiamide acetate may suppress the growth of malignant gliomas by blocking autophagic flux. 2. Aurantiamide acetate has an anti-neuroinflammatory effect on LPS stimulation through its inhibition of the NF-κB, JNK and p38 pathways. 3. Aurantiamide acetate inhibits cysteine proteinases, in particular, cathepsin L (3.4.22.15) and B (3.4.22.1) with IC50 of 12 microM and 49 microM, respectively . 4. Aurantiamide acetate is the main active substance of EECTD, which has significant anti-inflammatory and antinociceptive activities. |
| Targets | Autophagy | NO | PGE | NOS | COX | TNF-α | IL Receptor | NF-kB | IkB | JNK | p38MAPK | IKK |

Aurantiamide Dilution Calculator

Aurantiamide Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.4845 mL | 12.4224 mL | 24.8447 mL | 49.6894 mL | 62.1118 mL |
| 5 mM | 0.4969 mL | 2.4845 mL | 4.9689 mL | 9.9379 mL | 12.4224 mL |
| 10 mM | 0.2484 mL | 1.2422 mL | 2.4845 mL | 4.9689 mL | 6.2112 mL |
| 50 mM | 0.0497 mL | 0.2484 mL | 0.4969 mL | 0.9938 mL | 1.2422 mL |
| 100 mM | 0.0248 mL | 0.1242 mL | 0.2484 mL | 0.4969 mL | 0.6211 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 1-(4-(3-Chloropropoxy)-3-methoxyphenyl)ethanone
Catalog No.:BCC8406
CAS No.:58113-30-7
- Fmoc-Arg(NO2)-OH
Catalog No.:BCC2596
CAS No.:58111-94-7
- Undulatoside A
Catalog No.:BCN6773
CAS No.:58108-99-9
- Isonicoteine
Catalog No.:BCN2152
CAS No.:581-50-0
- Anatabine
Catalog No.:BCN6899
CAS No.:581-49-7
- Suberosin
Catalog No.:BCN5791
CAS No.:581-31-7
- α-MSH
Catalog No.:BCC7420
CAS No.:581-05-5
- trans-3,4-Methylenedioxycinnamyl alcohol
Catalog No.:BCN1410
CAS No.:58095-76-4
- Miltefosine
Catalog No.:BCC4360
CAS No.:58066-85-6
- 24, 25-Dihydroxy VD2
Catalog No.:BCC1302
CAS No.:58050-55-8
- Averantin
Catalog No.:BCN7027
CAS No.:5803-62-3
- HOKU-81
Catalog No.:BCC1634
CAS No.:58020-43-2
- SU 4312
Catalog No.:BCC7073
CAS No.:5812-07-7
- 2-Hydroxynaringenin
Catalog No.:BCN4820
CAS No.:58124-18-8
- NSC 207895 (XI-006)
Catalog No.:BCC2243
CAS No.:58131-57-0
- Militarine
Catalog No.:BCN2551
CAS No.:58139-23-4
- Ethyl 5-amino-4-cyano-3-(2-ethoxy-2-oxoethyl)thiophene-2-carboxylate
Catalog No.:BCC8975
CAS No.:58168-20-0
- Idebenone
Catalog No.:BCC4913
CAS No.:58186-27-9
- H-DL-Ser-OMe.HCl
Catalog No.:BCC3100
CAS No.:5819-04-5
- Tetraethyl ranelate
Catalog No.:BCC9177
CAS No.:58194-26-6
- Betulin palmitate
Catalog No.:BCN5792
CAS No.:582315-55-7
- BMS265246
Catalog No.:BCC3741
CAS No.:582315-72-8
- ICA 069673
Catalog No.:BCC7911
CAS No.:582323-16-8
- Kakkalide
Catalog No.:BCN8263
CAS No.:58274-56-9
Aurantiamide acetate suppresses the growth of malignant gliomas in vitro and in vivo by inhibiting autophagic flux.[Pubmed:25704599]
J Cell Mol Med. 2015 May;19(5):1055-64.
We aim to investigate the effect of Aurantiamide acetate isolated from the aerial parts of Clematis terniflora DC against gliomas. Human malignant glioma U87 and U251 cells were incubated with different concentrations (0-100 muM) of Aurantiamide acetate. Aurantiamide acetate greatly decreased the cell viability in a dose- and time-dependent manner. It induced moderate mitochondrial fragmentation and the loss of mitochondrial membrane potential. No significant difference was found in the alternation of other intracellular organelles, although F-actin structure was slightly disturbed. Apparent ultrastructure alternation with increased autophagosome and autolysosome accumulation was observed in Aurantiamide acetate-treated cells. The expression of LC3-II was greatly up-regulated in cells exposed to Aurantiamide acetate (P < 0.05 compared with control). The cytoplasmic accumulation of autophagosomes and autolysosomes induced by Aurantiamide acetate treatment was confirmed by fluorescent reporter protein labelling. Administration of chloroquine (CQ), which inhibits the fusion step of autophagosomes, further increased the accumulation of autophagosomes in the cytoplasm of U87 cells. Autophagy inhibition by 3-methyladenine, Bafilomycin A1 or CQ had no influence on Aurantiamide acetate-induced cytotoxicity, whereas autophagy stimulator rapamycin significantly suppressed Aurantiamide acetate-induced cell death. The anti-tumour effects of Aurantiamide acetate were further evaluated in tumour-bearing nude mice. Intratumoural injection of Aurantiamide acetate obviously suppressed tumour growth, and increased number of autophagic vacuoles was observed in tumour tissues of animals receiving Aurantiamide acetate. Our findings suggest that Aurantiamide acetate may suppress the growth of malignant gliomas by blocking autophagic flux.
An in vivo and in vitro assessment of the anti-inflammatory, antinociceptive, and immunomodulatory activities of Clematis terniflora DC. extract, participation of aurantiamide acetate.[Pubmed:25910534]
J Ethnopharmacol. 2015 Jul 1;169:287-94.
AIM: Clematis terniflora DC. has been widely used as a traditional Chinese medicine for the treatment of tonsillitis, rheumatoid arthritis, and prostatitis. Despite its widespread use in China, there are currently no studies systematically examined its therapeutic effects and mechanism of action. As such, the present study was conducted to evaluate the anti-inflammatory, antinociceptive, and immunomodulatory effects of C. terniflora DC. using rodent and cellular models. METHODS: The anti-inflammatory properties of the 70% ethanol eluted fraction of the 70% ethanol extract of C. terniflora DC. (EECTD) were evaluated using the xylene-induced ear swelling test, the carrageenan-induced edema model, and the cotton pellet granuloma method. Its antinociceptive activities were determined using both the acetic acid-induced writhing test and the hot plate assay. In parallel, we conducted an in vitro assay in LPS-induced RAW264.7 cells to examine the anti-inflammatory effects of EECTD and its purified form, Aurantiamide acetate (AA) on inhibition of nitric oxide (NO) and prostaglandin E2 (PGE2) release. RESULTS: EECTD (300mg/kg) significantly reduced the number of writhing, extended the pain response latency, and suppressed xylene-induced ear swelling. Each EECTD treatment group also had significant inhibition of cotton granulation formation in addition to reduced carrageenan-induced paw edema. EECTD was also shown to alleviate signs of inflammation in histopathological paw sections. However, it had a less noticeable effect on mouse ear swelling in the delayed type hypersensitivity test. A purified compound was isolated from EECTD and its structure was identified as AA. In vitro experimental results showed that both EECTD and AA were able to significantly inhibit the release of pro-inflammatory cytokines NO and PGE2 on LPS-induced RAW264.7 cells. CONCLUSION: These results suggest that EECTD has significant anti-inflammatory and antinociceptive activities, partially related to one of the active substances identified as AA. We hypothesize that these effects are related to its ability to inhibit the production of cytokines NO and PGE2. However, further work will be needed to determine its exact mechanism of action.
Aurantiamide acetate, a selective cathepsin inhibitor, produced by Aspergillus penicilloides.[Pubmed:11440138]
Biosci Biotechnol Biochem. 2001 May;65(5):1195-7.
Aurantiamide acetate was isolated from the fermentation broth of Aspergillus penicilloides for the first time. Aurantiamide acetate inhibited cysteine proteinases, in particular, cathepsin L (3.4.22.15) and B (3.4.22.1) with IC50 of 12 microM and 49 microM, respectively. In the adjuvant-arthritic rat model, subcutaneously administered 10 mg/kg body weight of this compound suppressed hind paw swelling.
Anti-neuroinflammatory effect of aurantiamide acetate from the marine fungus Aspergillus sp. SF-5921: inhibition of NF-kappaB and MAPK pathways in lipopolysaccharide-induced mouse BV2 microglial cells.[Pubmed:25448500]
Int Immunopharmacol. 2014 Dec;23(2):568-74.
In the course of a search for anti-neuroinflammatory metabolites from marine fungi, Aurantiamide acetate (1) was isolated from marine-derived Aspergillus sp. as an anti-neuroinflammatory component. Compound 1 dose-dependently inhibited the production of nitric oxide (NO) and prostaglandin E2 (PGE2) in BV2 microglial cells. It also attenuated inducible NO synthase (iNOS), cyclooxygenase-2 (COX-2), and other pro-inflammatory cytokines, such as interleukin-1beta (IL-1beta) and tumor necrosis factor-alpha (TNF-alpha). In a further study designed to elucidate the mechanism of its anti-neuroinflammatory effect, compound 1 was shown to block the activation of nuclear factor-kappa B (NF-kappaB) in lipopolysaccharide (LPS)-induced BV2 microglial cells by inhibiting the phosphorylation of the inhibitor kappa B-alpha (IkappaB)-alpha. In addition, compound 1 decreased the phosphorylation levels of c-Jun N-terminal kinase (JNK) and p38 mitogen-activated protein kinases (MAPKs). These results suggest that compound 1 has an anti-neuroinflammatory effect on LPS stimulation through its inhibition of the NF-kappaB, JNK and p38 pathways.


