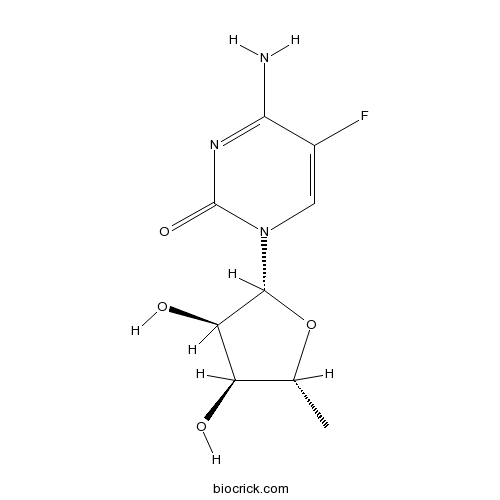
5'-Deoxy-5-fluorocytidineCAS# 66335-38-4 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 66335-38-4 | SDF | Download SDF |
| PubChem ID | 10037499 | Appearance | Powder |
| Formula | C9H12FN3O4 | M.Wt | 245 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 4-amino-1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-methyloxolan-2-yl]-5-fluoropyrimidin-2-one | ||
| SMILES | CC1C(C(C(O1)N2C=C(C(=NC2=O)N)F)O)O | ||
| Standard InChIKey | YSNABXSEHNLERR-ZIYNGMLESA-N | ||
| Standard InChI | InChI=1S/C9H12FN3O4/c1-3-5(14)6(15)8(17-3)13-2-4(10)7(11)12-9(13)16/h2-3,5-6,8,14-15H,1H3,(H2,11,12,16)/t3-,5-,6-,8-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

5'-Deoxy-5-fluorocytidine Dilution Calculator

5'-Deoxy-5-fluorocytidine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.0816 mL | 20.4082 mL | 40.8163 mL | 81.6327 mL | 102.0408 mL |
| 5 mM | 0.8163 mL | 4.0816 mL | 8.1633 mL | 16.3265 mL | 20.4082 mL |
| 10 mM | 0.4082 mL | 2.0408 mL | 4.0816 mL | 8.1633 mL | 10.2041 mL |
| 50 mM | 0.0816 mL | 0.4082 mL | 0.8163 mL | 1.6327 mL | 2.0408 mL |
| 100 mM | 0.0408 mL | 0.2041 mL | 0.4082 mL | 0.8163 mL | 1.0204 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Dihydroguaiaretic acid
Catalog No.:BCN4212
CAS No.:66322-34-7
- ML 171
Catalog No.:BCC6252
CAS No.:6631-94-3
- 3H-1,2-Benzodithiol-3-one-1,1-dioxide
Catalog No.:BCC8633
CAS No.:66304-01-6
- Pregomisin
Catalog No.:BCN7000
CAS No.:66280-26-0
- Gomisin J
Catalog No.:BCN2270
CAS No.:66280-25-9
- 4-Amino-2-methylquinoline
Catalog No.:BCC8677
CAS No.:6628-04-2
- 6'-O-Acetylpaniculoside II
Catalog No.:BCC8316
CAS No.:
- Stemonidine
Catalog No.:BCC8253
CAS No.:66267-46-7
- N-dotriacontanol
Catalog No.:BCC8218
CAS No.:6624-79-9
- N-Benzylanthranilic acid
Catalog No.:BCC9093
CAS No.:6622-55-5
- Cyromazine
Catalog No.:BCC5328
CAS No.:66215-27-8
- Deacetylxylopic acid
Catalog No.:BCN4210
CAS No.:6619-95-0
- Ranitidine
Catalog No.:BCC9134
CAS No.:66357-35-5
- Ranitidine Hydrochloride
Catalog No.:BCC4533
CAS No.:66357-59-3
- TGX-221
Catalog No.:BCC1244
CAS No.:663619-89-4
- Methylsyringol
Catalog No.:BCN3535
CAS No.:6638-05-7
- (+)-Apogossypol
Catalog No.:BCC5585
CAS No.:66389-74-0
- Chloranthalactone A
Catalog No.:BCN8022
CAS No.:66395-02-6
- Chloranthalactone B
Catalog No.:BCN8020
CAS No.:66395-03-7
- 5alpha-Hydroxychloranthalactone A
Catalog No.:BCN7469
CAS No.:66395-04-8
- Pamoic acid disodium salt
Catalog No.:BCC7909
CAS No.:6640-22-8
- 13-Hydroxyoxyberberine
Catalog No.:BCN3355
CAS No.:66408-27-3
- 6-Amino-1,3-dimethyluracil
Catalog No.:BCC8755
CAS No.:6642-31-5
- 2-(2,4-Diaminophenoxy)ethanol dihydrochloride
Catalog No.:BCN8497
CAS No.:66422-95-5
Rabeprazole intake does not affect systemic exposure to capecitabine and its metabolites, 5'-deoxy-5-fluorocytidine, 5'-deoxy-5-fluorouridine, and 5-fluorouracil.[Pubmed:30972456]
Cancer Chemother Pharmacol. 2019 Apr 9. pii: 10.1007/s00280-019-03837-y.
PURPOSE: Several retrospective studies have shown that the antitumor efficacy of capecitabine-containing chemotherapy decreases when co-administered with a proton pump inhibitor (PPI). Although a reduction in capecitabine absorption by PPIs was proposed as the underlying mechanism, the effects of PPIs on capecitabine pharmacokinetics remain unclear. We prospectively examined the effects of rabeprazole on the pharmacokinetics of capecitabine and its metabolites. METHODS: We enrolled patients administered adjuvant capecitabine plus oxaliplatin (CapeOX) for postoperative colorectal cancer (CRC) patients and metastatic CRC patients receiving CapeOX with/without bevacizumab. Patients receiving a PPI before registration were allocated to the rabeprazole group, and the PPI was changed to rabeprazole (20 mg/day) at least 1 week before the initiation of capecitabine treatment. On day 1, oral capecitabine (1000 mg/m(2)) was administered 1 h after rabeprazole intake. Oxaliplatin (and bevacizumab) administration on day 1 was shifted to day 2 for pharmacokinetic analysis of the first capecitabine dose. Plasma concentrations of capecitabine, 5'-deoxy-5-fluorocytidine, 5'-deoxy-5-fluorouridine, and 5-fluorouracil were analyzed by high-performance liquid chromatography. Effects of rabeprazole on inhibition of cell proliferation by each capecitabine metabolite were examined with colon cancer cells (COLO205 and HCT116). RESULTS: Five and 9 patients enrolled between September 2017 and July 2018 were allocated to rabeprazole and control groups, respectively. No significant effects of rabeprazole on area under the plasma concentration-time curve divided by capecitabine dose for capecitabine and its three metabolites were observed. Rabeprazole did not affect the proliferation inhibition of colon cancer cells by the respective capecitabine metabolites. CONCLUSION: Rabeprazole does not affect capecitabine pharmacokinetics.
Pharmacokinetic analysis of metronomic capecitabine in refractory metastatic colorectal cancer patients.[Pubmed:29488048]
Invest New Drugs. 2018 Aug;36(4):709-714.
The aim of the present study was to assess the pharmacokinetics (PK) of metronomic capecitabine and its metabolites in a population of refractory metastatic colorectal cancer (mCRC) patients. Thirty-four patients (M/F, 22/12) with a diagnosis of mCRC received capecitabine 800 mg p.o. twice a day and cyclophosphamide 50 mg/day p.o. Blood samples were collected at baseline, 15 min, 30 min, 1 h, 1.5 h, 2 h, 3 h and 5 h at day 1 after capecitabine administration. Plasma concentrations of capecitabine and its metabolites were measured by high performance liquid chromatography and the main PK parameters were calculated. Maximum plasma concentrations (Cmax) of capecitabine (11.51 +/- 9.73 mug/ml) occurred at 0.5 h, whereas the Cmax of 5'-deoxy-5-fluorocytidine (5'-DFCR; 2.45 +/- 2.93 mug/ml), 5'-deoxy-5-fluorouridine (5'-DFUR; 6.43 +/- 8.2 mug/ml), and 5-fluorouracil (5-FU; 0.24 +/- 0.16 mug/ml) were found at 1 h, 1.5 h and 1 h, respectively. Capecitabine, 5'-DFCR, 5'-DFUR and 5-FU AUCs at day 1 were 21.30 +/- 10.78, 5.2 +/- 4.6, 19.59 +/- 3.83 and 0.66 +/- 0.77 hxmug/ml, respectively. In conclusion, low doses of capecitabine were rapidly absorbed and extensively metabolized, achieving measurable plasma concentrations in a heavily pretreated population of patients.
Circadian variations in the pharmacokinetics of capecitabine and its metabolites in rats.[Pubmed:29175408]
Eur J Pharm Sci. 2018 Jan 15;112:152-158.
Capecitabine, an orally available prodrug of 5-fluorouracil, is widely used to treat patients with colorectal cancer. Although various studies have shown circadian variations in plasma 5-fluorouracil concentrations during long-term infusion, it is still unknown whether circadian variations also exist following administration of capecitabine. The present study aimed to investigate whether the pharmacokinetics of capecitabine and its metabolites, including 5-fluorouracil, vary according to administration time in rats. Rats were orally administered capecitabine (180mg/kg) at 07:00 (23h after light onset, HALO), 13:00 (5 HALO), or 19:00h (11 HALO). Plasma concentrations of capecitabine and its metabolites, such as 5'-deoxy-5-fluorocytidine (5'-DFCR), 5'-deoxy-5-fluorouridine (5'-DFUR), and 5-fluorouracil, were determined after capecitabine administration. The results showed that the t1/2 and AUC0-infinity values of 5-fluorouracil differed as a function of the dosing time of capecitabine. The maximum and minimum mean t1/2 values of 5-fluorouracil were obtained when the drug was administered at 07:00h (23 HALO: 3.1+/-1.2h) and 13:00h (5 HALO: 1.5+/-0.6h), respectively. The AUC0-infinity value of 5-fluorouracil at 07:00h (23 HALO: 533.9+/-195.7mumolh/L) was 1.8-fold higher than the value at 13:00h (5 HALO: 302.5+/-157.1mumolh/L). The clearance of 5-fluorouracil followed a cosine circadian curve, and the simulated population mean clearance was highest at rest times and lowest during active times in rats. The results for the plasma 5'-DFCR and 5'-DFUR levels indicated that circadian variations in the sequential metabolism of capecitabine to 5-fluorouracil would also affect plasma 5-fluorouracil levels following capecitabine administration. In conclusion, the pharmacokinetics of capecitabine and its metabolites, including 5-fluorouracil, varied according to time of dosing, suggesting that the capecitabine administration time is an important factor in achieving sufficient efficacy and reducing toxicity in patients.
Assessment of metabolism-dependent drug efficacy and toxicity on a multilayer organs-on-a-chip.[Pubmed:27605158]
Integr Biol (Camb). 2016 Oct 10;8(10):1022-1029.
Pharmaceutical development is greatly hindered by the poor predictive power of existing in vitro models for drug efficacy and toxicity testing. In this work, we present a new and multilayer organs-on-a-chip device that allows for the assessment of drug metabolism, and its resultant drug efficacy and cytotoxicity in different organ-specific cells simultaneously. Four cell lines representing the liver, tumor (breast cancer and lung cancer), and normal tissue (gastric cells) were cultured in the compartmentalized micro-chambers of the multilayer microdevice. We adopted the prodrug capecitabine (CAP) as a model drug. The intermediate metabolites 5'-deoxy-5-fluorocytidine (DFUR) of CAP that were metabolized from liver and its active metabolite 5-fluorouracil (5-FU) from the targeted cancer cells and normal tissue cells were identified using mass spectrometry. CAP exhibited strong cytoxicity on breast cancer and lung cancer cells, but not in normal gastric cells. Moreover, the drug-induced cytotoxicity on cells varied in various target tissues, suggesting the metabolism-dependent drug efficacy in different tissues as exisits in vivo. This in vitro model can not only allow for characterizing the dynamic metabolism of anti-cancer drugs in different tissues simultaneously, but also facilitate the assessment of drug bioactivity on various target tissues in a simple way, indicating the utility of this organs-on-chip for applications in pharmacodynamics/pharmacokinetics studies, drug efficacy and toxicity testing.
Simultaneous determination of capecitabine and its three nucleoside metabolites in human plasma by high performance liquid chromatography-tandem mass spectrometry.[Pubmed:25808944]
J Chromatogr B Analyt Technol Biomed Life Sci. 2015 May 1;989:71-9.
Capecitabine (Cape) is a prodrug that is metabolized into 5'-deoxy-5-fluorocytidine (DFCR), 5'-deoxy-5-fluorouridine (DFUR), and 5-fluorouracil (5-FU) after oral administration. A liquid chromatography-tandem mass spectrometry method for the simultaneous determination of capecitabine and its three metabolites in human plasma was developed and validated. The ex vivo conversion of DFCR to DFUR in human blood was investigated and an appropriate blood sample handling condition was recommended. Capecitabine and its metabolites were extracted from 100 muL of plasma by protein precipitation. Adequate chromatographic retention and efficient separation were achieved on an Atlantis dC18 column under gradient elution. Interferences from endogenous matrix and the naturally occurring heavy isotopic species were avoided. Detection was performed in electrospray ionization mode using a polarity-switching strategy. The method was linear in the range of 10.0-5000 ng/mL for Cape, DFCR, and DFUR, and 2.00-200 ng/mL for 5-FU. The LLOQ was established at 10.0 ng/mL for Cape, DFCR, and DFUR, and 2.00 ng/mL for 5-FU. The inter- and intra-day precisions were less than 13.5%, 11.1%, 9.7%, and 11.4%, and the accuracy was in the range of -13.2% to 1.6%, -2.4% to 2.5%, -7.1% to 8.2%, and -2.0% to 3.8% for Cape, DFCR, DFUR, and 5-FU, respectively. The matrix effect was negligible under the current conditions. The mean extraction recoveries were within 105-115%, 92.6-101%, 94.0-100%, and 85.1-99.9% for Cape, DFCR, DFUR, and 5-FU, respectively. Stability testing showed that the four analytes remained stable under all relevant analytical conditions. This method has been applied to a clinical bioequivalence study.
Capecitabine and lapatinib uptake in surgically resected brain metastases from metastatic breast cancer patients: a prospective study.[Pubmed:25015089]
Neuro Oncol. 2015 Feb;17(2):289-95.
BACKGROUND: Breast cancer brain metastases (BCBM) are challenging complications that respond poorly to systemic therapy. The role of the blood-tumor barrier in limiting BCBM drug delivery and efficacy has been debated. Herein, we determined tissue and serum levels of capecitabine, its prodrug metabolites, and lapatinib in women with BCBM resected via medically indicated craniotomy. METHODS: Study patients with BCBM requiring surgical resection received either single-dose capecitabine (1250 mg/m(2)) 2-3 h before surgery or 2-5 doses of lapatinib (1250 mg) daily, the last dose 2-3 h before surgery. Serum samples were collected serially on the day of surgery. Drug concentrations were determined in serum and BCBM using liquid chromatography tandem mass spectrometry. RESULTS: Twelve patients were enrolled: 8 for capecitabine and 4 for lapatinib. Measurable drug levels of capecitabine and metabolites, 5'-deoxy-5-fluorocytidine, 5'-deoxy-5-fluorouridine, and 5-fluorouracil, were detected in all BCBM. The ratio of BCBM to serum was higher for 5-fluorouracil than for capecitabine. As for lapatinib, the median BCBM concentrations ranged from 1.0 to 6.5 microM. A high variability (0.19-9.8) was noted for lapatinib BCBM-to-serum ratio. CONCLUSIONS: This is the first study to demonstrate that capecitabine and lapatinib penetrate to a significant though variable degree in human BCBM. Drug delivery to BCBM is variable and in many cases appears partially limiting. Elucidating mechanisms that limit drug concentration and innovative approaches to overcome limited drug uptake will be important to improve clinical efficacy of these agents in the central nervous system. Trial registration ID: NCT00795678.
Pharmacokinetics and exposure-effect relationships of capecitabine in elderly patients with breast or colorectal cancer.[Pubmed:24801171]
Cancer Chemother Pharmacol. 2014 Jun;73(6):1285-93.
PURPOSE: The aims of the present study were (1) to investigate the impact of great age on pharmacokinetics of capecitabine and its metabolites and (2) to evaluate the exposure-effect relationship of capecitabine in elderly patients. METHODS: Data collected from 20 elderly patients (75-92 years old) with breast or colorectal cancer who received oral capecitabine were analyzed. In order to study the old age effect on pharmacokinetics, data collected from two phase I studies involving 40 younger adults (<75 years old) with metastatic cancer who received oral capecitabine were added in the database. The population pharmacokinetic analysis was based on a four-compartment model describing the sequence of capecitabine and three of its metabolites. RESULTS: The absorption rate constant was found lower in the oldest patient group (>/=75 years) compared with the youngest group, and the constant rate elimination of the 5-fluorouracil metabolite was found decreased over time (i.e., after 2 consecutive weeks of capecitabine administration). This time effect was not found different between the two age groups. In elderly patients, the exposure-safety analysis showed, from the second cycle of chemotherapy, significantly higher median exposures of capecitabine and its metabolites (5'-deoxy-5-fluorocytidine, 5'-deoxy-5-fluorouridine and 5-fluorouracil) in patients who experienced hand-foot syndrome compared with patients who did not. CONCLUSION: This study puts forward new arguments for the treatment of elderly cancer patients who could benefit from capecitabine chemotherapy with acceptable toxicity.
Safety and pharmacology of gemcitabine and capecitabine in patients with advanced pancreatico-biliary cancer and hepatic dysfunction.[Pubmed:24166106]
Cancer Chemother Pharmacol. 2014 Jan;73(1):113-24.
PURPOSE: We assessed the impact of hepatic dysfunction on the safety and pharmacology of gemcitabine/capecitabine in patients with advanced pancreatico-biliary cancer. METHODS: We included 12 patients receiving 3 weekly gemcitabine 1,000 mg/m(2) day 1, 8 and oral capecitabine 650 mg/m(2) b.i.d. over 2 weeks until disease progression or intolerable toxicity. Patients were included into one normal hepatic function cohort [total bilirubin (TB) 80 mumol/L). Three patients with a creatinine clearance <60 ml/min were also included. Patients were sampled for gemcitabine, difluoro-deoxy uridine, intracellular gemcitabine triphosphates, capecitabine, 5'-deoxy-5-fluorocytidine, 5'-deoxy-5-fluorouridine and 5-fluorouracil up to 4 h after initiation of chemotherapy on day 1, and up to 90 min on day 8. All compounds were analyzed using validated liquid chromatography-tandem mass spectrometry. Nonlinear mixed-effect modeling was used for population analysis. RESULTS: Hepatic dysfunction was caused by intrahepatic cholestasis in 4 out of 8 patients (50 %) and extrahepatic cholestasis in another 4 patients (50 %). Dose-limiting toxicity was increasing hyperbilirubinemia and severe neutropenia in 2 patients each. Hepatic dysfunction was not associated with dose-limiting toxicity or severe hematological or non-hematological toxicity. However, hepatic dysfunction was associated with low clearance of both gemcitabine (p = 10(-3)) and capecitabine (p = 10(-5)), and low intracellular gemcitabine triphosphate concentrations (p = 10(-3)). CONCLUSIONS: Gemcitabine/capecitabine can be given at the standard dose in patients with severe hyperbilirubinemia, though the present data suggest that gemcitabine's activity may be limited due to poor intracellular activation. In patients with severe hyperbilirubinemia, initial monotherapy with capecitabine should be considered, followed by the addition of gemcitabine with improving hyperbilirubinemia.
Higher capecitabine AUC in elderly patients with advanced colorectal cancer (SWOGS0030).[Pubmed:24022189]
Br J Cancer. 2013 Oct 1;109(7):1744-9.
BACKGROUND: The aging process is accompanied by physiological changes including reduced glomerular filtration and hepatic function, as well as changes in gastric secretions. To investigate what effect would aging have on the disposition of capecitabine and its metabolites, the pharmacokinetics between patients >/=70 years and <60 years were compared in SWOG0030. METHODS: Twenty-nine unresectable colorectal cancer patients were stratified to either >/=70 or <60 years of age, where the disposition of capecitabine and its metabolites were compared. RESULTS: Notable increase in capecitabine area under the curve (AUC) was accompanied by reduction in capecitabine clearance in >/=70 years patients (P<0.05). No difference in 5'-deoxy-5-fluorocytidine, 5'-deoxy-5-fluorouridine (DFUR), and 5-fluorouracil (5FU) AUCs between the two age groups, suggesting that carboxylesterase and cytidine deaminase (CDA) activity was similar between the two age groups. These results suggest that metabolic enzymes involved in converting capecitabine metabolites are not altered by age. An elevation in capecitabine Cmax and reduction in clearance was seen in females, where capecitabine AUC was 40.3% higher in women. Elevation of DFUR Cmax (45%) and AUC (46%) (P<0.05) was also noted, suggesting that CDA activity may be higher in females. CONCLUSION: Increases in capecitabine Cmax and AUC was observed in patients >/=70 years when compared with younger patients who were >60 years.


