Sorafenib TosylateRaf kinases and tyrosine kinases inhibitor CAS# 475207-59-1 |
- B-Raf inhibitor 1
Catalog No.:BCC4182
CAS No.:1093100-40-3
- Dabrafenib (GSK2118436)
Catalog No.:BCC4393
CAS No.:1195765-45-7
- TAK-632
Catalog No.:BCC3639
CAS No.:1228591-30-7
- R18
Catalog No.:BCC2383
CAS No.:211364-78-2
- AZ 628
Catalog No.:BCC3730
CAS No.:878739-06-1
- GDC-0879
Catalog No.:BCC2482
CAS No.:905281-76-7
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 475207-59-1 | SDF | Download SDF |
| PubChem ID | 406563 | Appearance | Powder |
| Formula | C28H24ClF3N4O6S | M.Wt | 637.03 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | Bay 43-9006 | ||
| Solubility | DMSO : ≥ 31 mg/mL (48.66 mM) H2O : < 0.1 mg/mL (insoluble) *"≥" means soluble, but saturation unknown. | ||
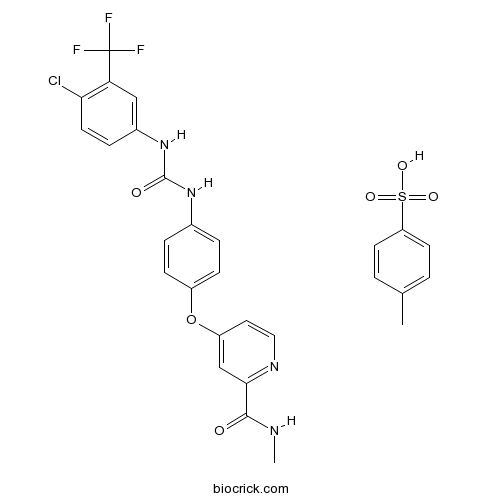
| Chemical Name | 4-[4-[[4-chloro-3-(trifluoromethyl)phenyl]carbamoylamino]phenoxy]-N-methylpyridine-2-carboxamide;4-methylbenzenesulfonic acid | ||
| SMILES | CC1=CC=C(C=C1)S(=O)(=O)O.CNC(=O)C1=NC=CC(=C1)OC2=CC=C(C=C2)NC(=O)NC3=CC(=C(C=C3)Cl)C(F)(F)F | ||
| Standard InChIKey | IVDHYUQIDRJSTI-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C21H16ClF3N4O3.C7H8O3S/c1-26-19(30)18-11-15(8-9-27-18)32-14-5-2-12(3-6-14)28-20(31)29-13-4-7-17(22)16(10-13)21(23,24)25;1-6-2-4-7(5-3-6)11(8,9)10/h2-11H,1H3,(H,26,30)(H2,28,29,31);2-5H,1H3,(H,8,9,10) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Sorafenib Tosylate (Bay 43-9006) is a multikinase inhibitor of Raf-1, B-Raf and VEGFR-2 with IC50 of 6 nM, 22 nM and 90 nM, respectively. | ||||||
| Targets | Raf-1 | B-Raf | VEGFR2 | PDGFRβ | |||
| IC50 | 6 nM | 22 nM | 90 nM | 57 nM | |||

Sorafenib Tosylate Dilution Calculator

Sorafenib Tosylate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.5698 mL | 7.8489 mL | 15.6978 mL | 31.3957 mL | 39.2446 mL |
| 5 mM | 0.314 mL | 1.5698 mL | 3.1396 mL | 6.2791 mL | 7.8489 mL |
| 10 mM | 0.157 mL | 0.7849 mL | 1.5698 mL | 3.1396 mL | 3.9245 mL |
| 50 mM | 0.0314 mL | 0.157 mL | 0.314 mL | 0.6279 mL | 0.7849 mL |
| 100 mM | 0.0157 mL | 0.0785 mL | 0.157 mL | 0.314 mL | 0.3924 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Sorafenib tosylate, also named nexavar, is a small-molecule anticancer compound [1]. It is also a novel oral Raf kinase and a vascular endothelial growth factor receptor (VEGFR) inhibitor. It inhibits tumor cell proliferation and tumor angiogenesis [2]. To HepG2 cells (1× 106), the IC50 of sorafenib tosylate is 2.09μg/ml [3].
Raf is a mitogen-stimulated protein kinase that functions as a component of the signaling cascade that leads to the stimulation of mitogen-activated protein kinase [4].
Vascular endothelial growth factor (VEGF) is a highly specific mitogen for vascular endothelial cells [5].
Treatment with nexavar potently inhibited the cell proliferation of MV4-11 cells (FLT3-ITD) in a dose-dependent manner with an IC50 of 0.88 nM. In MV4-11 cells, sorafenib tosylate of a concentration of 100 nM induced 43.6±5.2% of the cells to undergo apoptosis whereas in EOL-1 cells a concentration as low as 10 nM induced 89.29±1.8% of the cells to be apoptotic [6].
Nude rats at the age of 6 weeks injected with 105 MDA-MB-231 cells were involved. After monotherapy with sorafenib tosylate a significant reduction of the osteolytic lesion volume was observed on days 45 and 55 and of the soft tissue component volume on day 55 in comparison to untreated animals (p < 0.05). Compared to controls, treatment with sorafenib tosylate made bone metastases show significantly decreased values of Amplitude A and kep from day 35 to 55 (Amplitude A: p<0.01; kep p<0.01 on days 35 and 55; p<0.05 on day 45) [7].
References:
[1]. Chetan Lathia, John Lettieri, Frank Cihon, et al. Lack of effect of ketoconazole-mediated CYP3A inhibition on sorafenib clinical pharmacokinetics. Cancer Chemother Pharmacol, 2006, 57: 685-692.
[2]. M. J. Gnoth, S. Sandmann, K. Engel, et al. In Vitro to In Vivo Comparison of the Substrate Characteristics of Sorafenib Tosylate toward P-Glycoprotein. Drug Metabolism & Disposition, 2010, 38: 1341–1346.
[3]. Sayantan Dey, Subhadeep Roy, Nilanjana Deb, et al. Anti-carcinogenic Activity of Ruellia Tuberosa L. (Acanthaceae) Leaf Extract on Hepatoma Cell Line & Increased Superoxide Dismutase Activity on Macrophage Cell Lysate. Int J Pharm Pharm Sci, 2010, 5(Suppl 3): 854-861.
[4]. Markus Wartmann and Roger J. Davis. The Native Structure of the Activated Raf Protein Kinase Is a Membrane-bound Multi-subunit Complex. The Journal of Biological Chemistry, 1994, 269(9): 6695-6701.
[5]. Gera Neufeld, Tzafra Cohen, Stela Gengrinovitch, et al. Vascular endothelial growth factor (VEGF) and its receptors. The FASEB Journal, 1999, 13: 9-22.
[6]. D Auclair, D Miller, V Yatsula, et al. Antitumor activity of sorafenib in FLT3-driven leukemic cells. Leukemia, 2007, 21:439-445.
[7]. Maximilian Merz, Dorde Komljenovic, Stefan Zwick, et al. Sorafenib tosylate and paclitaxel induce anti-angiogenic, anti-tumour and anti-resorptive effects in experimental breast cancer bone metastases. European Journal of Cancer, 2011, 47:277-286.
- A-317491
Catalog No.:BCC1320
CAS No.:475205-49-3
- 2-Methylthioadenosine diphosphate trisodium salt
Catalog No.:BCC5794
CAS No.:475193-31-8
- BAN ORL 24
Catalog No.:BCC1398
CAS No.:475150-69-7
- Galnon
Catalog No.:BCC5871
CAS No.:475115-35-6
- ZSTK474
Catalog No.:BCC3657
CAS No.:475110-96-4
- Tivozanib (AV-951)
Catalog No.:BCC1179
CAS No.:475108-18-0
- NS 304
Catalog No.:BCC7661
CAS No.:475086-01-2
- Nuciferine
Catalog No.:BCN1223
CAS No.:475-83-2
- Glaucine
Catalog No.:BCN2550
CAS No.:475-81-0
- Aristolochic acid B
Catalog No.:BCN6263
CAS No.:475-80-9
- Liriodenine
Catalog No.:BCN5532
CAS No.:475-75-2
- (+)-Isocorynoline
Catalog No.:BCN2361
CAS No.:475-67-2
- Nogo-66 (1-40)
Catalog No.:BCC5862
CAS No.:475221-20-6
- CORM-3
Catalog No.:BCC5108
CAS No.:475473-26-8
- Aleglitazar
Catalog No.:BCC1337
CAS No.:475479-34-6
- NVP-ADW742
Catalog No.:BCC4553
CAS No.:475488-23-4
- NVP-AEW541
Catalog No.:BCC1180
CAS No.:475489-16-8
- MCL 0020
Catalog No.:BCC6025
CAS No.:475498-26-1
- Isotretinoin
Catalog No.:BCC2284
CAS No.:4759-48-2
- Xylotriose
Catalog No.:BCN8428
CAS No.:47592-59-6
- Lycorine
Catalog No.:BCN2409
CAS No.:476-28-8
- Chelidonine
Catalog No.:BCN2463
CAS No.:476-32-4
- Ellagic acid
Catalog No.:BCN5533
CAS No.:476-66-4
- Corydine
Catalog No.:BCN2669
CAS No.:476-69-7
Improving anti-tumor activity of sorafenib tosylate by lipid- and polymer-coated nanomatrix.[Pubmed:28165798]
Drug Deliv. 2017 Nov;24(1):270-277.
In the present study, we select the Sylysia 350 (Sylysia) as mesoporous material, distearoylphosphatidylethanolamine-poly(ethylene glycol)2000 (DSPE-PEG) as absorption enhancer and hydroxy propyl methyl cellulose (HPMC) as crystallization inhibitor to prepare Sorafenib Tosylate (SFN) nanomitrix (MSNM@SFN) for improving the anti-tumor activity of SFN. The MSNM@SFN was prepared by solvent evaporation method. The solubility, dissolution, and bioavailability of SFN in MSNM@SFN were also investigated. The anti-tumor activity of MSNM@SFN was evaluated in vitro and in vivo. Our results indicated that the solubility and dissolution of SFN in MSNM@SFN were significantly increased. The oral bioavailability of SFN in MSNM@SFN was greatly improved 7.7-fold compared with that in SFN suspension. The enhanced anti-tumor activity of MSNM@SFN was confirmed in vitro and in vivo experiments. This nanomatrix developed in this study could be a promising drug delivery platform for improving the therapeutic efficacy of poorly water-soluble drugs.
A large-scale prospective registration study of the safety and efficacy of sorafenib tosylate in unresectable or metastatic renal cell carcinoma in Japan: results of over 3200 consecutive cases in post-marketing all-patient surveillance.[Pubmed:26206897]
Jpn J Clin Oncol. 2015 Oct;45(10):953-62.
OBJECTIVE: Real-life safety and efficacy of sorafenib in advanced renal cell carcinoma in a nationwide patient population were evaluated by post-marketing all-patient surveillance. METHODS: All patients with unresectable or metastatic renal cell carcinoma in Japan who started sorafenib therapy from February 2008 to September 2009 were registered and followed for up to 12 months. Baseline characteristics, treatment status, tumor response, survival and safety data were recorded by the prescribing physicians. RESULTS: Safety and efficacy were evaluated in 3255 and 3171 patients, respectively. The initial daily dose was 800 mg in 78.2% of patients. Median duration of treatment was 6.7 months and the mean relative dose intensity was 68.4%. Overall, 2227 patients (68.4%) discontinued the treatment by 12 months, half of which (52.0% of discontinued patients) were due to adverse events. The most common adverse drug reactions were hand-foot skin reaction (59%), hypertension (36%), rash (25%) and increase in lipase/amylase (23%). The median progression-free survival was 7.3 months (95% confidence intervals: 6.7-8.1), and the overall survival rate at 1 year was 75.4% (73.5-77.1). Prognostic factors for overall survival were mostly consistent with those in previous clinical trials in the univariate analysis and largely similar to those for progression-free survival and duration of treatment in the multivariate analysis. CONCLUSIONS: Sorafenib for the treatment of advanced renal cell carcinoma under the labeled dose was feasible in daily medical practice, for its acceptable toxicity profile and favorable clinical benefit that were consistent with those in clinical trials.
Sorafenib and its tosylate salt: a multikinase inhibitor for treating cancer.[Pubmed:21206080]
Acta Crystallogr C. 2011 Jan;67(Pt 1):o29-32.
Sorafenib, a drug that targets malignant cancer cells and cuts off the blood supply feeding the tumour, has been crystallized as the free base, 4-(4-{3-[4-chloro-3-(trifluoromethyl)phenyl]ureido}phenoxy)-N-methylpyridine-2-ca rboxamide, C(21)H(16)ClF(3)N(4)O(3), (I), and as a tosylate salt, 4-(4-{3-[4-chloro-3-(trifluoromethyl)phenyl]ureido}phenoxy)-2-(N-methylcarbamoyl) pyridinium 4-methylbenzenesulfonate, C(21)H(17)ClF(3)N(4)O(3)(+).C(7)H(7)O(3)S(-), (II). In both structures, the sorafenib molecule is in an extended conformation. The pyridine-2-carboxamide group exhibits a syn conformation of the N atoms in (I), whereas an almost anti orientation is present in (II). In both crystal structures, the two terminal groups, viz. pyridine-2-carboxamide and the trifluorophenyl ring, are oriented differently to the conformations found in enzyme-bound sorafenib. The sorafenib molecules in (I) are linked into zigzag chains by N-H...O hydrogen bonds, whereas in (II) the presence of the additional tosylate anion results in the formation of chains of fused hydrogen-bonded rings. This study reveals the variations in the solid-state conformation of the sorafenib molecule in different crystalline environments.
Sorafenib tosylate as a radiosensitizer in malignant astrocytoma.[Pubmed:24139873]
J Clin Neurosci. 2014 Jan;21(1):131-6.
Progress in research on the molecular aspects of glioblastoma has yet to provide a medical therapy that significantly improves prognosis. Glioblastoma invariably progress through current treatment regimens with radiotherapy as a key component. Activation of several signaling pathways is thought to be associated with this resistance to radiotherapy. Ras activity is exceptionally high in glioblastoma and may regulate sensitivity to radiotherapy. Raf-1, a downstream effector of Ras, demonstrates a high amount of activity in glioblastoma. Therefore, Raf-1 inhibition should be considered as a mechanism to increase the effectiveness of radiotherapy in treatment regimen. In vitro analysis was performed with a novel Raf-1 kinase inhibitor (BAY 54-9085) in culture with the glioblastoma cell line U1242. The cell line was treated in serum-containing media and analyzed for the effect of the BAY 54-9085 alone and BAY 54-9085 combined with radiation on cell death. BAY 54-9085 displayed a cytocidal effect on glioblastoma cells following a 3 day incubation with the drug in serum-containing media. A dose of 2.5 muM displayed moderate cell death which significantly increased with a dose of 5.0 muM. In addition, glioblastoma cells treated with both the BAY 54-9085 and gamma radiation displayed a significant increase in cell death (85.5%) as compared to either BAY 54-9085 (73.1%) or radiation (34.4%) alone. Radiation therapy is a key component of treatment for glioblastoma. A novel Raf-1 inhibitor displayed in vitro evidence of synergistically increasing cell death of glioblastoma cells in combination with radiation.


