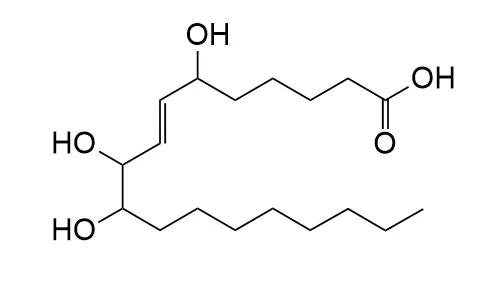
Sanleng acidCAS# 163860-24-0 |
Quality Control & MSDS
Package In Stock
Number of papers citing our products

| Cas No. | 163860-24-0 | SDF | File under preparation. |
| PubChem ID | N/A | Appearance | Powder |
| Formula | C18H34O5 | M.Wt | 330.5 |
| Type of Compound | Miscellaneous | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Sanleng acid Dilution Calculator

Sanleng acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.0257 mL | 15.1286 mL | 30.2572 mL | 60.5144 mL | 75.643 mL |
| 5 mM | 0.6051 mL | 3.0257 mL | 6.0514 mL | 12.1029 mL | 15.1286 mL |
| 10 mM | 0.3026 mL | 1.5129 mL | 3.0257 mL | 6.0514 mL | 7.5643 mL |
| 50 mM | 0.0605 mL | 0.3026 mL | 0.6051 mL | 1.2103 mL | 1.5129 mL |
| 100 mM | 0.0303 mL | 0.1513 mL | 0.3026 mL | 0.6051 mL | 0.7564 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Samioside
Catalog No.:BCX2154
CAS No.:360768-68-9
- rel-(7R,8'R,8S)-forsythialan C
Catalog No.:BCX2153
CAS No.:2089054-57-7
- (6-O-p-coumaroyl)-beta-glucopyranosyl-2-O-(4-hydroxybenzoyl)-4-O-beta-glucopyranosyl-6-hydroxyphenylacetate
Catalog No.:BCX2152
CAS No.:1987871-73-7
- Leonoside F
Catalog No.:BCX2151
CAS No.:1360075-79-1
- 3,4-Dihydroxy-5-methoxybenzoic acidmethylester
Catalog No.:BCX2150
CAS No.:3934-86-9
- Obtusichromoneside B
Catalog No.:BCX2149
CAS No.:2414481-39-1
- Ajugamarin A1
Catalog No.:BCX2148
CAS No.:78798-40-0
- Monoisovalerate
Catalog No.:BCX2147
CAS No.:95486-32-1
- Mangostanin
Catalog No.:BCX2146
CAS No.:463342-39-4
- 4-Hydroxy-2-methoxybenzoic acid
Catalog No.:BCX2145
CAS No.:90111-34-5
- 15-O-Methylgraciliflorin F
Catalog No.:BCX2144
CAS No.:1411994-51-8
- Hydrastinine
Catalog No.:BCX2143
CAS No.:5936-29-8
- Clerodendroside (Scutellarein 4'-methyl ether 7-glucuronide)
Catalog No.:BCX2156
CAS No.:64924-06-7
- Tetrandrine 2' alpha-oxide (Tetrandrine-2'-N-alpha-oxide)
Catalog No.:BCX2157
CAS No.:90365-80-3
- Acetyl-11 alpha-methoxy-beta-boswellic acid
Catalog No.:BCX2158
CAS No.:918548-43-3
- 1'-Hydroxyselinidin
Catalog No.:BCX2159
CAS No.:97274-82-3
- 5-Methoxytaxifolin (5-Methoxyl-3',4',7-trihydroxylflavanonol)
Catalog No.:BCX2160
CAS No.:139493-38-2
- 2,3,4,6-Tetragalloylglucose
Catalog No.:BCX2161
CAS No.:40410-95-5
- Obtusinaphthalenside B
Catalog No.:BCX2162
CAS No.:2584414-13-9
- Cassialactone 9-O-beta-D-glucopyranosyl-(1→6)-beta-D-glucopyranoside
Catalog No.:BCX2163
CAS No.:2101837-94-7
- Cistanoside H
Catalog No.:BCX2164
CAS No.:104806-92-0
- Styraxlignolide G
Catalog No.:BCX2165
CAS No.:41607-21-0
- Meliasendanin D 4'-O-beta-D-glucopyranoside
Catalog No.:BCX2166
CAS No.:1931100-79-6
- Steponine
Catalog No.:BCX2167
CAS No.:17132-69-3
Study of the active ingredients and mechanism of Sparganii rhizoma in gastric cancer based on HPLC-Q-TOF-MS/MS and network pharmacology.[Pubmed:33479376]
Sci Rep. 2021 Jan 21;11(1):1905.
Sparganii rhizoma (SL) has potential therapeutic effects on gastric cancer (GC), but its main active ingredients and possible anticancer mechanism are still unclear. In this study, we used HPLC-Q-TOF-MS/MS to comprehensively analyse the chemical components of the aqueous extract of SL. On this basis, a network pharmacology method incorporating target prediction, gene function annotation, and molecular docking was performed to analyse the identified compounds, thereby determining the main active ingredients and hub genes of SL in the treatment of GC. Finally, the mRNA and protein expression levels of the hub genes of GC patients were further analysed by the Oncomine, GEPIA, and HPA databases. A total of 41 compounds were identified from the aqueous extract of SL. Through network analysis, we identified seven main active ingredients and ten hub genes: acacetin, Sanleng acid, ferulic acid, methyl 3,6-dihydroxy-2-[(2-hydroxyphenyl) ethynyl]benzoate, caffeic acid, adenine nucleoside, azelaic acid and PIK3R1, PIK3CA, SRC, MAPK1, AKT1, HSP90AA1, HRAS, STAT3, FYN, and RHOA. The results indicated that SL might play a role in GC treatment by controlling the PI3K-Akt and other signalling pathways to regulate biological processes such as proliferation, apoptosis, migration, and angiogenesis in tumour cells. In conclusion, this study used HPLC-Q-TOF-MS/MS combined with a network pharmacology approach to provide an essential reference for identifying the chemical components of SL and its mechanism of action in the treatment of GC.


