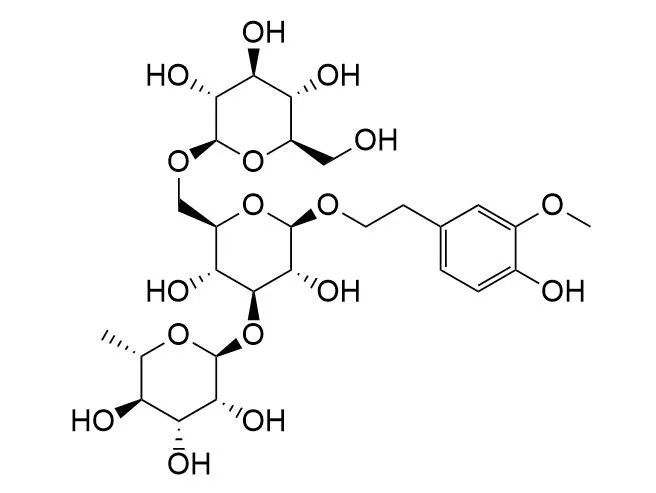
Leonoside FCAS# 1360075-79-1 |
Quality Control & MSDS
Package In Stock
Number of papers citing our products

| Cas No. | 1360075-79-1 | SDF | File under preparation. |
| PubChem ID | N/A | Appearance | Powder |
| Formula | C27H42O17 | M.Wt | 638.6 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Leonoside F Dilution Calculator

Leonoside F Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.5659 mL | 7.8296 mL | 15.6593 mL | 31.3185 mL | 39.1481 mL |
| 5 mM | 0.3132 mL | 1.5659 mL | 3.1319 mL | 6.2637 mL | 7.8296 mL |
| 10 mM | 0.1566 mL | 0.783 mL | 1.5659 mL | 3.1319 mL | 3.9148 mL |
| 50 mM | 0.0313 mL | 0.1566 mL | 0.3132 mL | 0.6264 mL | 0.783 mL |
| 100 mM | 0.0157 mL | 0.0783 mL | 0.1566 mL | 0.3132 mL | 0.3915 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 3,4-Dihydroxy-5-methoxybenzoic acidmethylester
Catalog No.:BCX2150
CAS No.:3934-86-9
- Obtusichromoneside B
Catalog No.:BCX2149
CAS No.:2414481-39-1
- Ajugamarin A1
Catalog No.:BCX2148
CAS No.:78798-40-0
- Monoisovalerate
Catalog No.:BCX2147
CAS No.:95486-32-1
- Mangostanin
Catalog No.:BCX2146
CAS No.:463342-39-4
- 4-Hydroxy-2-methoxybenzoic acid
Catalog No.:BCX2145
CAS No.:90111-34-5
- 15-O-Methylgraciliflorin F
Catalog No.:BCX2144
CAS No.:1411994-51-8
- Hydrastinine
Catalog No.:BCX2143
CAS No.:5936-29-8
- Notoginsenoside E
Catalog No.:BCX2142
CAS No.:193976-50-0
- 5-Allyl-1-methoxy-2,3-dihydroxybenzene
Catalog No.:BCX2141
CAS No.:4055-72-5
- Auranamide
Catalog No.:BCX2140
CAS No.:740813-53-0
- Terrestriamide
Catalog No.:BCX2139
CAS No.:157536-49-7
- (6-O-p-coumaroyl)-beta-glucopyranosyl-2-O-(4-hydroxybenzoyl)-4-O-beta-glucopyranosyl-6-hydroxyphenylacetate
Catalog No.:BCX2152
CAS No.:1987871-73-7
- rel-(7R,8'R,8S)-forsythialan C
Catalog No.:BCX2153
CAS No.:2089054-57-7
- Samioside
Catalog No.:BCX2154
CAS No.:360768-68-9
- Sanleng acid
Catalog No.:BCX2155
CAS No.:163860-24-0
- Clerodendroside (Scutellarein 4'-methyl ether 7-glucuronide)
Catalog No.:BCX2156
CAS No.:64924-06-7
- Tetrandrine 2' alpha-oxide (Tetrandrine-2'-N-alpha-oxide)
Catalog No.:BCX2157
CAS No.:90365-80-3
- Acetyl-11 alpha-methoxy-beta-boswellic acid
Catalog No.:BCX2158
CAS No.:918548-43-3
- 1'-Hydroxyselinidin
Catalog No.:BCX2159
CAS No.:97274-82-3
- 5-Methoxytaxifolin (5-Methoxyl-3',4',7-trihydroxylflavanonol)
Catalog No.:BCX2160
CAS No.:139493-38-2
- 2,3,4,6-Tetragalloylglucose
Catalog No.:BCX2161
CAS No.:40410-95-5
- Obtusinaphthalenside B
Catalog No.:BCX2162
CAS No.:2584414-13-9
- Cassialactone 9-O-beta-D-glucopyranosyl-(1→6)-beta-D-glucopyranoside
Catalog No.:BCX2163
CAS No.:2101837-94-7
Interrupted Pummerer Reaction in Latent-Active Glycosylation: Glycosyl Donors with a Recyclable and Regenerative Leaving Group.[Pubmed:26448185]
Angew Chem Int Ed Engl. 2015 Nov 23;54(48):14432-6.
Latent O-glycosides, 2-(2-propylthiol)benzyl (PTB) glycosides, were converted into the corresponding active glycosyl donors, 2-(2-propylsulfinyl)benzyl (PSB) glycosides, by a simple and efficient oxidation. Treatment of the PSB donor and various acceptors with triflic anhydride provided the desired glycosides in good to excellent yields. The leaving group, which was activated by an interrupted Pummerer reaction, can be recycled (PSB-OH) and regenerated as the precursor (PTB-OH). A natural hepatoprotective glycoside, Leonoside F, was efficiently synthesized in a convergent [3+1] manner with this newly developed method. The present total synthesis also led to a structural revision of this phenylethanoid glycoside.
Hepatoprotective glycosides from Leonurus japonicus Houtt.[Pubmed:22196928]
Carbohydr Res. 2012 Feb 1;348:42-6.
Two new phenylethanoid glycosides 1 and 2 named leonoside E and Leonoside F, and one new sesquiterpene glycoside (3) identified as 7alpha (H)-eudesmane-4,11 (12)-diene-3-one-2beta-hydroxy-13-beta-d-glucopyranoside, together with seven known glycosides (4-10), were isolated from the aerial part of Leonurus japonicus Houtt. Their structures were elucidated on the basis of spectroscopic data and chemical evidence. When tested in in vitro assays, compounds 1, 2, 4, and 6 exhibited potent hepatoprotective activity against d-galactosamine-induced toxicity in HL-7702 cells at concentration of 1x10(-5) M.


