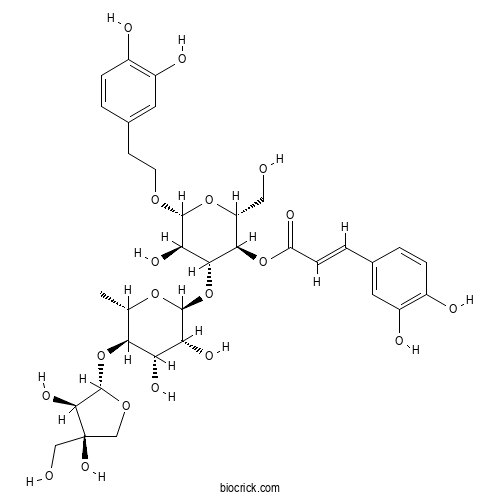
SamiosideCAS# 360768-68-9 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 360768-68-9 | SDF | Download SDF |
| PubChem ID | 10350153 | Appearance | Powder |
| Formula | C34H44O19 | M.Wt | 756.7 |
| Type of Compound | Phenylpropanoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | [(2R,3R,4R,5R,6R)-4-[(2S,3R,4S,5R,6S)-5-[(2S,3R,4R)-3,4-dihydroxy-4-(hydroxymethyl)oxolan-2-yl]oxy-3,4-dihydroxy-6-methyloxan-2-yl]oxy-6-[2-(3,4-dihydroxyphenyl)ethoxy]-5-hydroxy-2-(hydroxymethyl)oxan-3-yl] (E)-3-(3,4-dihydroxyphenyl)prop-2-enoate | ||
| SMILES | CC1C(C(C(C(O1)OC2C(C(OC(C2OC(=O)C=CC3=CC(=C(C=C3)O)O)CO)OCCC4=CC(=C(C=C4)O)O)O)O)O)OC5C(C(CO5)(CO)O)O | ||
| Standard InChIKey | MYTQIOQLRFFFEC-HQQSBTJXSA-N | ||
| Standard InChI | InChI=1S/C34H44O19/c1-15-27(52-33-30(45)34(46,13-36)14-48-33)24(42)25(43)32(49-15)53-29-26(44)31(47-9-8-17-3-6-19(38)21(40)11-17)50-22(12-35)28(29)51-23(41)7-4-16-2-5-18(37)20(39)10-16/h2-7,10-11,15,22,24-33,35-40,42-46H,8-9,12-14H2,1H3/b7-4+/t15-,22+,24-,25+,26+,27-,28+,29+,30-,31+,32-,33-,34+/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Samioside Dilution Calculator

Samioside Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.3215 mL | 6.6076 mL | 13.2153 mL | 26.4306 mL | 33.0382 mL |
| 5 mM | 0.2643 mL | 1.3215 mL | 2.6431 mL | 5.2861 mL | 6.6076 mL |
| 10 mM | 0.1322 mL | 0.6608 mL | 1.3215 mL | 2.6431 mL | 3.3038 mL |
| 50 mM | 0.0264 mL | 0.1322 mL | 0.2643 mL | 0.5286 mL | 0.6608 mL |
| 100 mM | 0.0132 mL | 0.0661 mL | 0.1322 mL | 0.2643 mL | 0.3304 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- rel-(7R,8'R,8S)-forsythialan C
Catalog No.:BCX2153
CAS No.:2089054-57-7
- (6-O-p-coumaroyl)-beta-glucopyranosyl-2-O-(4-hydroxybenzoyl)-4-O-beta-glucopyranosyl-6-hydroxyphenylacetate
Catalog No.:BCX2152
CAS No.:1987871-73-7
- Leonoside F
Catalog No.:BCX2151
CAS No.:1360075-79-1
- 3,4-Dihydroxy-5-methoxybenzoic acidmethylester
Catalog No.:BCX2150
CAS No.:3934-86-9
- Obtusichromoneside B
Catalog No.:BCX2149
CAS No.:2414481-39-1
- Ajugamarin A1
Catalog No.:BCX2148
CAS No.:78798-40-0
- Monoisovalerate
Catalog No.:BCX2147
CAS No.:95486-32-1
- Mangostanin
Catalog No.:BCX2146
CAS No.:463342-39-4
- 4-Hydroxy-2-methoxybenzoic acid
Catalog No.:BCX2145
CAS No.:90111-34-5
- 15-O-Methylgraciliflorin F
Catalog No.:BCX2144
CAS No.:1411994-51-8
- Hydrastinine
Catalog No.:BCX2143
CAS No.:5936-29-8
- Notoginsenoside E
Catalog No.:BCX2142
CAS No.:193976-50-0
- Sanleng acid
Catalog No.:BCX2155
CAS No.:163860-24-0
- Clerodendroside (Scutellarein 4'-methyl ether 7-glucuronide)
Catalog No.:BCX2156
CAS No.:64924-06-7
- Tetrandrine 2' alpha-oxide (Tetrandrine-2'-N-alpha-oxide)
Catalog No.:BCX2157
CAS No.:90365-80-3
- Acetyl-11 alpha-methoxy-beta-boswellic acid
Catalog No.:BCX2158
CAS No.:918548-43-3
- 1'-Hydroxyselinidin
Catalog No.:BCX2159
CAS No.:97274-82-3
- 5-Methoxytaxifolin (5-Methoxyl-3',4',7-trihydroxylflavanonol)
Catalog No.:BCX2160
CAS No.:139493-38-2
- 2,3,4,6-Tetragalloylglucose
Catalog No.:BCX2161
CAS No.:40410-95-5
- Obtusinaphthalenside B
Catalog No.:BCX2162
CAS No.:2584414-13-9
- Cassialactone 9-O-beta-D-glucopyranosyl-(1→6)-beta-D-glucopyranoside
Catalog No.:BCX2163
CAS No.:2101837-94-7
- Cistanoside H
Catalog No.:BCX2164
CAS No.:104806-92-0
- Styraxlignolide G
Catalog No.:BCX2165
CAS No.:41607-21-0
- Meliasendanin D 4'-O-beta-D-glucopyranoside
Catalog No.:BCX2166
CAS No.:1931100-79-6
Chemical Characterization, Antioxidant and Enzyme-Inhibitory Activities of Different Extracts from Three Phlomis Species.[Pubmed:40351016]
ChemistryOpen. 2025 Aug;14(8):e202500004.
Phlomis species (family Lamiaceae) are highly valued as food and herbal medicine. The present study is designed to investigate the chemical composition and antioxidant and enzyme inhibitory activities of extracts from P. fruticosa, P. herba-venti, and P. kurdica aerial parts. Different classes of metabolites, including phenolic acids, phenylethanoids, flavonoids, iridoids, organic acids, terpenes, and fatty acids, are identified in the three species, with methanol as the best solvent to recover bioactive compounds from the three species in addition to ethyl acetate for P. kurdica. Around 70% methanol extract of P. herba-venti exerts the best radical scavenging and ions-reducing properties, while its methanol extract exhibits the highest acetylcholinesterase inhibitory activity. The ethyl acetate extract of P. fruticosa displays the best chelating power, and its other polar extracts have the highest total antioxidant activity. Furthermore, molecular docking and molecular dynamics simulations have underscored the therapeutic potential of bioactive compounds, including isoverbascoside, Samioside, forsythoside B, and hattushoside. In conclusion, the study indicates that these three Phlomis species are a rich source of bioactive molecules with possible therapeutic applications, and the selection of appropriate extraction solvents is crucial for the targeted biological activity.
Anti-Alzheimer's Potency of Rich Phenylethanoid Glycosides Extract from Marrubium vulgare L.: In Vitro and In Silico Studies.[Pubmed:39458923]
Pharmaceuticals (Basel). 2024 Sep 27;17(10):1282.
BACKGROUND/OBJECTIVES: Marrubium vulgare L. (M. vulgare), the white horehound, is well known for treating inflammation-related diseases. METHODS: In this context, we investigated the efficacy of M. vulgare ingredients in treating Alzheimer's disease using various in vitro and in silico antioxidant, anti-inflammatory, anti-cholinesterase, and anti-tyrosinase mechanisms. RESULTS: In our results, sixty-one components were tentatively identified using gas and liquid chromatography (GC-MS and LC-MS(n)) and categorized as hydrocarbons, fatty acids, and polyphenolics. The extract inhibited linoleic oxidation with an IC(50) value of 114.72 microg/mL, captured iron (Fe(2+)) ions with an IC(50) value of 164.19 microg/mL, and displayed reducing power. In addition, the extract showed radical-scavenging ability towards DPPH(*), NO(*), ABTS(*+), and H(2)O(2) assays compared to L-ascorbic acid and butylated hydroxytoluene. The DPPH(*) was scavenged by 77.62% at 100 microg/mL, and NO(*), ABTS(*+), and H(2)O(2) were scavenged with IC(50) values of 531.66, 117.51, and 143.10 microg/mL, respectively. M. vulgare also exhibited discriminating anti-inflammatory potency against cyclooxygenase (COX-2) with IC(50) values of 619.15 microg/mL compared to celecoxib (p > 0.05). Notably, three Alzheimer's biomarkers, acetylcholinesterase (AChE), butyrylcholinesterase (BChE), and tyrosinase were significantly inhibited. The molecular docking study supposed that the phenylethanoid glycosides of Samioside and forsythoside B inhibited AChE and tyrosinase enzymes with low binding affinities of -9.969 and -8.804 kcal/mol, respectively. Marruboside was a proper inhibitor of COX and BChE enzymes with a binding score of -10.218 and -10.306 kcal/mol, respectively. CONCLUSIONS: M. vulgare extract showed significant inhibitory actions, which suggest that it could have a promising potential as an anti-Alzheimer agent.
Phytochemical Analysis and Trypanocidal Activity of Marrubium incanum Desr.[Pubmed:32660058]
Molecules. 2020 Jul 9;25(14):3140.
The rationale inspiring the discovery of lead compounds for the treatment of human parasitic protozoan diseases from natural sources is the well-established use of medicinal plants in various systems of traditional medicine. On this basis, we decided to select an overlooked medicinal plant growing in central Italy, Marrubium incanum Desr. (Lamiaceae), which has been used as a traditional remedy against protozoan diseases, and to investigate its potential against Human African trypanosomiasis (HAT). For this purpose, we assayed three extracts of different polarities obtained from the aerial parts of M. incanum-namely, water (MarrInc-H(2)O), ethanol (MarrInc-EtOH) and dichloromethane (MarrInc-CH(2)Cl(2))-against Trypanosoma brucei (TC221), with the aim to discover lead compounds for the development of antitrypanosomal drugs. Their selectivity index (SI) was determined on mammalian cells (BALB/3T3 mouse fibroblasts) as a counter-screen for toxicity. The preliminary screening selected the MarrInc-CH(2)Cl(2) extract as the most promising candidate against HAT, showing an IC(50) value of 28 mug/mL. On this basis, column chromatography coupled with the NMR spectroscopy of a MarrInc-CH(2)Cl(2) extract led to the isolation and identification of five compounds i.e. 1-alpha-linolenoyl-2-palmitoyl-3-stearoyl-sn- glycerol (1), 1-linoleoyl-2-palmitoyl-3-stearoyl-sn-glycerol (2), stigmasterol (3), palmitic acid (4), and salvigenin (5). Notably, compounds 3 and 5 were tested on T. brucei, with the latter being five-fold more active than the MarrInc-CH(2)Cl(2) extract (IC(50) = 5.41 +/- 0.85 and 28 +/- 1.4 mug/mL, respectively). Furthermore, the SI for salvigenin was >18.5, showing a preferential effect on target cells compared with the dichloromethane extract (>3.6). Conversely, stigmasterol was found to be inactive. To complete the work, also the more polar MarrInc-EtOH extract was analyzed, giving evidence for the presence of 2''-O-allopyranosyl-cosmosiin (6), verbascoside (7), and Samioside (8). Our findings shed light on the phytochemistry of this overlooked species and its antiprotozoal potential, providing evidence for the promising role of flavonoids such as salvigenin for the treatment of protozoal diseases.
Iridoid glycosides and polyphenolic compounds from Teucrium chamaedrys L.[Pubmed:29058476]
Nat Prod Res. 2018 Jul;32(13):1583-1589.
In this work, the phytochemical analysis of Teucrium chamaedrys L. collected in Italy was reported. Eight compounds were isolated and identified by means of classical column chromatography and spectroscopic techniques, such as NMR and MS. In detail, these compounds were: verbascoside (1), forsythoside b (2), Samioside (3), alyssonoside (4), harpagide (5), 8-O-acetyl-harpagide (6), cirsiliol (7) and beta-arbutin (8). The presence of these compounds, in particular iridoids and phenyl-ethanoid glycosides, has a chemotaxonomic relevance and results to be in perfect accordance with the current botanical classification of the species. In addition, it provides a phytochemical rationale for the use of this particular plant in the ethno-pharmacological field. Conversely, it is worth of mention the absence of potentially toxic components, unlike to what observed in other species of the genus which can no longer be used for ethno-medicinal purposes.
[Chemical constituents from Callicarpa nudiflora and their hemostatic activity].[Pubmed:21438394]
Zhongguo Zhong Yao Za Zhi. 2010 Dec;35(24):3297-301.
OBJECTIVE: To study the hemostatic effect of chemical constituents from Callicarpa nudiflora. METHOD: The chemical constituents were isolated and purified via silica gel and Sephadex LH-20 column chromatography. Their structures were determined on the basis of spectral analysis. prothrombin time (PT), activated partial thromboplastin time (APTT), thrombin time (TT) and fibrinogen (FIB) of the constituents rabbit blood samples were tested with rabbit blood in vitro. RESULT: Eleven compounds were isolated and identified as two diterpenens: 7alpha-hydroxy sandaracopimaric acid (1), 16, 17-dihydroxy-3-oxophyllocladane (2). Two phenoic glycosides: acteoside (3), Samioside(4). Three triterpenes: 2alpha, 3alpha, 24-trihydroxy-ursa-12-en-28-oic acid (5), 2alpha, 3alpha, 19alpha-trihydroxyursa-12-en-28-oic acid-28-0-beta-D-glucopyranosyl ester (6), and 2alpha, 3alpha, 19alpha, 23-tetrahydroxy-ursa-12-en-28-oic acid-28-0-beta-D-glucopyranosyl ester (7). Four flavones: rhamnazin (8), 5-Hydroxy-3, 7, 4'-trimethoxy-flavone (9) , 5-Hydroxy-3, 7, 3', 4'-tetramethoxyflavone (10), and luteoloside (11). All Compounds cannot significantly shorten the PT (P < 0.01), compounds 3, 4, 7, 10 can remarkedly increase APTT (P < 0.01), compound 5 can prolong the T( P < 0.01) obviously, and compound 8 can significantly increase the contents of FIB (P < 0.01). CONCLUSION: Compounds 2, 4 and 10 were isolated from this genus for the first time, and compounds 1, 3, 5, 6, 7 and 9 had been isolated from this plant for the first time. The hemostatic effect of C. nudiflora may be related to the activation of the intrinsic blood coagulation system.
Phenylethanoid and lignan glycosides from polar extracts of Lantana, a genus of verbenaceous plants widely used in traditional herbal therapies.[Pubmed:19485331]
J Nat Prod. 2009 Jul;72(7):1344-7.
Many references to the use of Lantana spp. can be found in the ethnopharmacological literature from locations around the globe. This study was focused on examining constituents from the polar extracts of Lantana radula Sw. and Lantana canescens Kunth, for which no prior chemical investigations had been reported. A new phenylethanoid glycoside, raduloside, and lignan glycoside, radulignan, were identified along with the known compounds alyssonoside, arenarioside, calceolarioside E, isonuomioside, Samioside, and verbascoside.
Identification by HPLC-PAD-MS and quantification by HPLC-PAD of phenylethanoid glycosides of five Phlomis species.[Pubmed:15688949]
Phytochem Anal. 2005 Jan-Feb;16(1):1-6.
The qualitative and quantitative determination of phenylethanoid glycosides in methanolic extracts of five species of the genus Phlomis (Lamiaceae) has been investigated using a new reversed-phase HPLC method combined with photodiode-array detection and electrospray/MS analysis. Forsythoside B, verbascoside, Samioside, alyssonoside, isoverbascoside, leucosceptosides A and B and martynoside were detected. The quantification of the above constituents was performed using echinacoside as internal standard. Knowledge of the content of phenylethanoid glycosides contributes to the chemotaxonomy of the genus.
Samioside, a new phenylethanoid glycoside with free-radical scavenging and antimicrobial activities from Phlomis samia.[Pubmed:11520237]
J Nat Prod. 2001 Aug;64(8):1095-7.
A new phenylethanoid glycoside, Samioside, was isolated from the aerial parts of Phlomis samia and identified as 1-O-3,4-(dihydroxyphenyl)ethyl beta-D-apiofuranosyl-(1-->4)-alpha-L-rhamnopyranosyl-(1-->3)-4-O-caffeoyl-beta-D-glucopyranoside (1). In addition, one known phenylethanoid glycoside and three known flavonoids were identified as acteoside (2), apigenin, chrysoeriol, and ermanin, respectively. The structure of 1 was elucidated on the basis of its spectroscopic data. Samioside (1) demonstrated scavenging properties toward the DPPH radical and antimicrobial activity against Gram-positive and -negative bacteria.


