PseudohypericinCAS# 55954-61-5 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 55954-61-5 | SDF | Download SDF |
| PubChem ID | 4978 | Appearance | Black-violet powder |
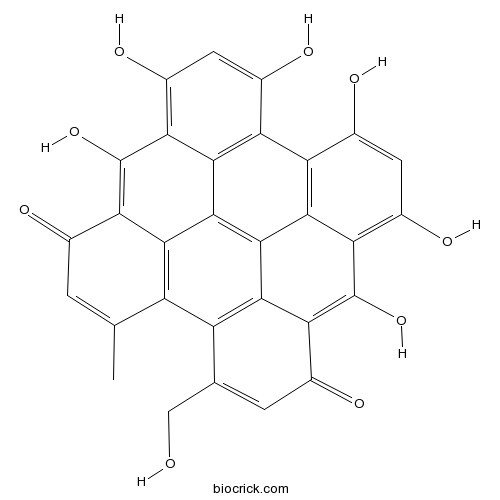
| Formula | C30H16O9 | M.Wt | 520.44 |
| Type of Compound | Anthraquinones | Storage | Desiccate at -20°C |
| Solubility | Soluble in pyridine; slightly soluble in aqueous alkaline solution | ||
| Chemical Name | 9,11,13,16,18,20-hexahydroxy-5-(hydroxymethyl)-24-methyloctacyclo[13.11.1.12,10.03,8.04,25.019,27.021,26.014,28]octacosa-1(26),2,4(25),5,8,10,12,14(28),15(27),16,18,20,23-tridecaene-7,22-dione | ||
| SMILES | CC1=CC(=O)C2=C(C3=C(C=C(C4=C3C5=C6C7=C(C1=C25)C(=CC(=O)C7=C(C8=C(C=C(C4=C86)O)O)O)CO)O)O)O | ||
| Standard InChIKey | NODGUBIGZKATOM-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C30H16O9/c1-7-2-9(32)19-23-15(7)16-8(6-31)3-10(33)20-24(16)28-26-18(12(35)5-14(37)22(26)30(20)39)17-11(34)4-13(36)21(29(19)38)25(17)27(23)28/h2-5,31,34-39H,6H2,1H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Pseudohypericin has photocytotoxic, and antiretroviral activities, it has potential therapeutic value in diseases such as AIDS. Pseudohypericin can inhibit LPS-stimulated PGE2 and NO in mouse macrophages by potentiating suppressor of cytokine signaling 3 (SOCS3) expression, it has anti-tumor potential. |
| Targets | PGE | NO | SOCS3 |
| In vitro | Photocytotoxic effect of pseudohypericin versus hypericin.[Reference: WebLink]J.Photoch.Photobiol. B , 1998, 45(2-3):87-94.Pseudohypericin and hypericin, the major photosensitizing constituents of Hypericum perforatum, are believed to cause hypericism. Studies of the mechanisms of action of the antiretroviral agents hypericin and pseudohypericin.[Reference: WebLink]P. Natl. Acad. Sci., 1989, 86(15):5963-7.
Download citationShare Request full-text Pseudohypericin in Hypericum perforatum inhibited LPS-stimulated PGE2 and NO in mouse macrophages by potentiating SOCS3 expression[Reference: WebLink]Cancer Prev. Res., 2010, 3(12 Supplement).Hypericum perforatum, also known as ‘St John’s wort’, is one of the most studied medicinal plants. Despite its primary use as an anti-depression agent, certain compounds such as hypericin and hyperforin in H. perforatum extract have been shown to inhibit tumor growth by inducing apoptosis/necrosis and inhibiting angiogenesis. Using hypericin in photodynamic cancer therapy is also being studied. Our previous studies have demonstrated that H. perforatum extract and a group of 4 compounds in it, namely Pseudohypericin, amentoflavone, quercetin, and chlorogenic acid, decreased lipopolysaccharide (LPS)-stimulated macrophage inflammatory response, and identified the activation of suppressor of cytokine signaling 3 (SOCS3) as a candidate mechanism for this activity. Given that both inflammation and SOCS3 suppression are associated with tumor progression, we studied the role of SOCS3 activation in the anti-inflammatory activity of H. perforatum extract and its components. |
| Structure Identification | J Sep Sci. 2015 May 12. doi: 10.1002/jssc.201500260.Combination of integrated expanded bed adsorption chromatography and countercurrent chromatography for the direct extraction and purification of pseudohypericin and hypericin from St. John's wort (Hypericum perforatum L.).[Pubmed: 25964189]St. John's wort has attracted particular attention because of its beneficial effects such as antidepressant, antiviral, and anticancer agent. |

Pseudohypericin Dilution Calculator

Pseudohypericin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.9215 mL | 9.6073 mL | 19.2145 mL | 38.429 mL | 48.0363 mL |
| 5 mM | 0.3843 mL | 1.9215 mL | 3.8429 mL | 7.6858 mL | 9.6073 mL |
| 10 mM | 0.1921 mL | 0.9607 mL | 1.9215 mL | 3.8429 mL | 4.8036 mL |
| 50 mM | 0.0384 mL | 0.1921 mL | 0.3843 mL | 0.7686 mL | 0.9607 mL |
| 100 mM | 0.0192 mL | 0.0961 mL | 0.1921 mL | 0.3843 mL | 0.4804 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Methylpheophorbide A
Catalog No.:BCN7998
CAS No.:5594-30-9
- Betamethasone 17,21-dipropionate
Catalog No.:BCC8875
CAS No.:5593-20-4
- Pennogenin 3-O-beta-chacotrioside
Catalog No.:BCN6707
CAS No.:55916-52-4
- Polyphyllin VI
Catalog No.:BCN1053
CAS No.:55916-51-3
- Cucurbitacin R
Catalog No.:BCN7877
CAS No.:55903-92-9
- Friedelin
Catalog No.:BCN5747
CAS No.:559-74-0
- beta-Amyrin
Catalog No.:BCN5746
CAS No.:559-70-6
- Morolic acid
Catalog No.:BCN7475
CAS No.:559-68-2
- 6-Hydroxysugiol
Catalog No.:BCN3154
CAS No.:55898-07-2
- Boc-D-Lys(Z)-OH
Catalog No.:BCC3423
CAS No.:55878-47-2
- 6-beta-Hydroxyhyoscyamine
Catalog No.:BCN1915
CAS No.:55869-99-3
- VER-49009
Catalog No.:BCC5297
CAS No.:558640-51-0
- ARC 239 dihydrochloride
Catalog No.:BCC6851
CAS No.:55974-42-0
- Nitazoxanide
Catalog No.:BCC3824
CAS No.:55981-09-4
- Methylthiouracil
Catalog No.:BCC4800
CAS No.:56-04-2
- 2,4-Diamino-6-hydroxypyrimidine
Catalog No.:BCC6658
CAS No.:56-06-4
- 4-Aminobutanoic acid
Catalog No.:BCN2187
CAS No.:56-12-2
- Cystamine dihydrochloride
Catalog No.:BCC6344
CAS No.:56-17-7
- Cantharidin
Catalog No.:BCN1280
CAS No.:56-25-7
- Tetraethylammonium chloride
Catalog No.:BCC7554
CAS No.:56-34-8
- H-Gly-OH
Catalog No.:BCC2946
CAS No.:56-40-6
- H-Ala-OH
Catalog No.:BCC3190
CAS No.:56-41-7
- H-Ser-OH
Catalog No.:BCC3028
CAS No.:56-45-1
- Deoxycorticosterone acetate
Catalog No.:BCC4655
CAS No.:56-47-3
Combination of integrated expanded bed adsorption chromatography and countercurrent chromatography for the direct extraction and purification of pseudohypericin and hypericin from St. John's wort (Hypericum perforatum L.).[Pubmed:25964189]
J Sep Sci. 2015 Aug;38(15):2588-96.
St. John's wort has attracted particular attention because of its beneficial effects as an antidepressant, antiviral, and anticancer agent. A method for the combination of integrated expanded bed adsorption chromatography and countercurrent chromatography for the simultaneous extraction and purification of Pseudohypericin and hypericin from the herb is presented in this paper. Firstly, the constituents were extracted and directly adsorbed by expanded bed adsorption chromatography under optimal conditions. The stepwise elution was then performed by expanded bed adsorption chromatography that enriched the targets with higher purities and recoveries compared to other methods. Secondly, the eluent fractions from expanded bed adsorption chromatography were further separated by two-step high-speed countercurrent chromatography. A two-step high-speed countercurrent chromatography method with a biphasic solvent system composed of n-hexane/ethyl acetate/methanol/water with a volume ratio of 1:2:1:2 was performed by stepwise changing the flow rate of the mobile phase. Consequently, 5.6 mg of Pseudohypericin and 2.2 mg of hypericin with purities of 95.5 and 95.0%, respectively, were successfully obtained from 40 mg of crude sample.


