PenduloneCAS# 69359-09-7 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 69359-09-7 | SDF | Download SDF |
| PubChem ID | 5320464 | Appearance | Powder |
| Formula | C17H16O6 | M.Wt | 316.31 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
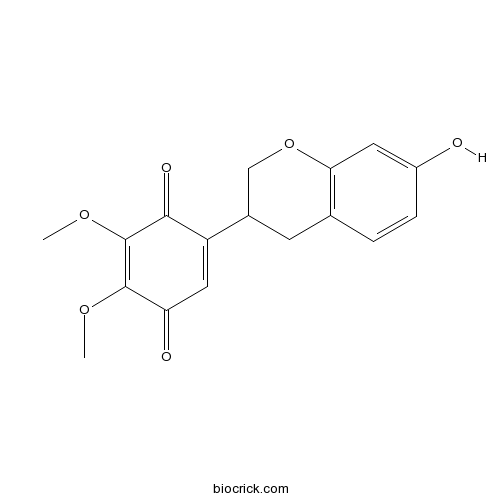
| Chemical Name | 5-(7-hydroxy-3,4-dihydro-2H-chromen-3-yl)-2,3-dimethoxycyclohexa-2,5-diene-1,4-dione | ||
| SMILES | COC1=C(C(=O)C(=CC1=O)C2CC3=C(C=C(C=C3)O)OC2)OC | ||
| Standard InChIKey | SHZOHJDZQPQBSW-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C17H16O6/c1-21-16-13(19)7-12(15(20)17(16)22-2)10-5-9-3-4-11(18)6-14(9)23-8-10/h3-4,6-7,10,18H,5,8H2,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Pendulone exhibits good anti-plasmodial activity with the IC50 value of 7.0 ± 0.8 uM. 2. Pendulone displays antibacterial activity against Staphylococcus aureus and methicillin-resistant S. aureus (each IC50 1.44 microg/mL). 3. Pendulone shows potent leishmanicidal activity. 4. Pendulone could be a valuable chemopreventive agent, it shows strong inhibition on the effect of the cell cycle induced by TPA and shows potent anti-tumor-promoting activity for an in vivo two-stage carcinogenesis test. |
| Targets | Antifection |

Pendulone Dilution Calculator

Pendulone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.1615 mL | 15.8073 mL | 31.6146 mL | 63.2291 mL | 79.0364 mL |
| 5 mM | 0.6323 mL | 3.1615 mL | 6.3229 mL | 12.6458 mL | 15.8073 mL |
| 10 mM | 0.3161 mL | 1.5807 mL | 3.1615 mL | 6.3229 mL | 7.9036 mL |
| 50 mM | 0.0632 mL | 0.3161 mL | 0.6323 mL | 1.2646 mL | 1.5807 mL |
| 100 mM | 0.0316 mL | 0.1581 mL | 0.3161 mL | 0.6323 mL | 0.7904 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Boc-D-Thr(Bzl)-OH
Catalog No.:BCC3454
CAS No.:69355-99-3
- Galanthamine hydrochloride
Catalog No.:BCC8277
CAS No.:5072-47-9
- Obtucarbamate A
Catalog No.:BCN3936
CAS No.:6935-99-5
- 2-Benzylaminopyridine
Catalog No.:BCC8565
CAS No.:6935-27-9
- CPPHA
Catalog No.:BCC1501
CAS No.:693288-97-0
- CYC116
Catalog No.:BCC2181
CAS No.:693228-63-6
- H-Ile-OtBu.HCl
Catalog No.:BCC2965
CAS No.:69320-89-4
- (R)-baclofen
Catalog No.:BCC4503
CAS No.:69308-37-8
- 12-Aminolauric acid
Catalog No.:BCC8436
CAS No.:693-57-2
- 3-beta-O-(cis-p-Coumaroyl)maslinic acid
Catalog No.:BCN1375
CAS No.:69297-40-1
- 8-O-Acetylharpagide
Catalog No.:BCN4256
CAS No.:6926-14-3
- Harpagide
Catalog No.:BCN4996
CAS No.:6926-08-5
- Schisanhenol
Catalog No.:BCN2508
CAS No.:69363-14-0
- Sulbactam sodium
Catalog No.:BCC4852
CAS No.:69388-84-7
- Lappaol H
Catalog No.:BCN8415
CAS No.:69394-18-9
- Amonafide
Catalog No.:BCC1249
CAS No.:69408-81-7
- SB505124
Catalog No.:BCC5087
CAS No.:694433-59-5
- 17-PA
Catalog No.:BCC7452
CAS No.:694438-95-4
- Parvifuran
Catalog No.:BCN7780
CAS No.:69470-93-5
- 2-Methyl-4-isobutyrylphloroglucinol
Catalog No.:BCN7176
CAS No.:69480-03-1
- Boc-D-His(Tos)-OH
Catalog No.:BCC3405
CAS No.:69541-68-0
- Thymopentin
Catalog No.:BCN8347
CAS No.:69558-55-0
- Octadecyl caffeate
Catalog No.:BCN6609
CAS No.:69573-60-0
- Tupichilignan A
Catalog No.:BCN4257
CAS No.:69586-96-5
In vitro leishmanicidal constituents of Millettia pendula.[Pubmed:16755071]
Chem Pharm Bull (Tokyo). 2006 Jun;54(6):915-7.
The in vitro leishmanicidal constituents of Millettia pendula were examined. Two new compounds, 1 (millettilone A) and 2 (millettilone B), were isolated from the methanol extract of M. pendula, together with six known compounds: 3R-claussequinone (3), Pendulone (4), secundiflorol I (5), 3,8-dihydroxy-9-methoxypterocarpan (6), 3,10-dihydroxy-7,9-dimethoxypterocarpan (7), and formononetin (8). Among these, Pendulone showed the most potent leishmanicidal activity. Compound 2 was found to be a purple pigment in this heartwood. Their chemical structures were elucidated using spectral methods.
Anti-tumor promoting activities of isoflavonoids from Wistaria brachybotrys.[Pubmed:9300132]
Biol Pharm Bull. 1997 Aug;20(8):865-8.
Eight minor isoflavonoids (3-10) isolated from the knot of Wistaria brachybotrys were tested for their inhibitory effects on Epstein-Barr virus (EBV) activation induced by the tumor promoter, 12-O-tetradecanoyl-phorbol-13-acetate (TPA), in Raji cells as a primary screening test for anti-tumor-promoters (cancer chemopreventive agents), and all the tested compounds showed inhibitory activity. Of these compounds, Pendulone (3) was further examined on the cell cycle of Raji cells, and indicated strong inhibition on the effect of the cell cycle induced by TPA. In addition, the compound showed potent anti-tumor-promoting activity for an in vivo two-stage carcinogenesis test of mouse skin using 7,12-dimethylbenz[a]-anthracene and TPA. Consequently, it suggests that 3 could be a valuable chemopreventive agent in chemical carcinogenesis.
Antiparasitic and antimicrobial isoflavanquinones from Abrus schimperi.[Pubmed:22224279]
Nat Prod Commun. 2011 Nov;6(11):1645-50.
The EtOH extract of Abrus schimperi (Fabaceae), collected in Kenya, demonstrated significant activity against Leishmania donovani promastigotes with IC50 value of 3.6 microg/mL. Bioassay-guided fractionation of CHCl3 fraction using Centrifugal Preparative TLC afforded two antiparasitic isoflavanquinones, namely amorphaquinone (1) and Pendulone (2). They displayed IC50 values of 0.63 microg/mL and 0.43 microg/mL, respectively, against L. donovani promastigotes. Both the compounds were also evaluated against L. donovani axenic amastigotes and amastigotes in THPI macrophage cultures. In addition, compounds 1 and 2 showed antiplasmodial activity against Plasmodium falciparum D6 and W2 strains, while 2 displayed antibacterial activity against Staphylococcus aureus and methicillin-resistant S. aureus (each IC50 1.44 microg/mL). The 1H and 13C data of 1, not fully assigned previously, were unambiguously assigned using 1D and 2D NMR HMBC and HMQC experiments. In addition, the absolute stereochemistry of the isolated compounds 1 and 2 was revised as C-(3S) based on Circular Dichroism experiments. This appears to be the first report of amorphaquinone (1) and Pendulone (2) from the genus Abrus.
Antiplasmodial Isoflavanes and Pterocarpans from Apoplanesia paniculata.[Pubmed:26018916]
Planta Med. 2015 Aug;81(12-13):1128-32.
Bioassay-guided fractionation of an EtOH extract of the roots of the plant Apoplanesia paniculata (Fabaceae) led to the isolation of the three known compounds amorphaquinone (1), Pendulone (2), and melilotocarpan C (3), and the two new pterocarpans 4 and 5. Compounds 1 and 2 exhibited good antiplasmodial activity with IC50 values of 5.7 +/- 1.5 and 7.0 +/- 0.8 microM, respectively. Compound 3 exhibited weak antiplasmodial activity (41.8 +/- 5.2 microM), while compounds 4 and 5 were inactive. Compound 6 was synthesized to confirm the structure of 5, and it showed enhanced antiplasmodial activity (15.8 +/- 1.4 microM) compared to its analogues 3-5.


