Malonic acidCAS# 141-82-2 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 141-82-2 | SDF | Download SDF |
| PubChem ID | 867 | Appearance | Powder |
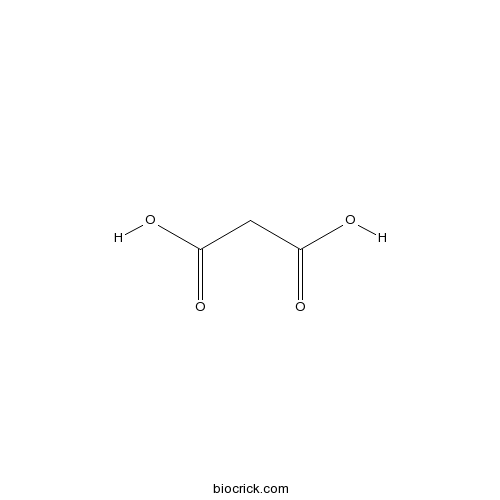
| Formula | C3H4O4 | M.Wt | 104.06 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | propanedioic acid | ||
| SMILES | C(C(=O)O)C(=O)O | ||
| Standard InChIKey | OFOBLEOULBTSOW-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C3H4O4/c4-2(5)1-3(6)7/h1H2,(H,4,5)(H,6,7) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Malonic acid Dilution Calculator

Malonic acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 9.6098 mL | 48.0492 mL | 96.0984 mL | 192.1968 mL | 240.246 mL |
| 5 mM | 1.922 mL | 9.6098 mL | 19.2197 mL | 38.4394 mL | 48.0492 mL |
| 10 mM | 0.961 mL | 4.8049 mL | 9.6098 mL | 19.2197 mL | 24.0246 mL |
| 50 mM | 0.1922 mL | 0.961 mL | 1.922 mL | 3.8439 mL | 4.8049 mL |
| 100 mM | 0.0961 mL | 0.4805 mL | 0.961 mL | 1.922 mL | 2.4025 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 2-Aminoethanol
Catalog No.:BCN1756
CAS No.:141-43-5
- Ricinoleic acid
Catalog No.:BCC8248
CAS No.:141-22-0
- Nerylacetate
Catalog No.:BCN3802
CAS No.:141-12-8
- Benzo-15-crown 5-ether
Catalog No.:BCC8849
CAS No.:14098-44-3
- Benzo-18-crown-6 ether
Catalog No.:BCC8850
CAS No.:14098-24-9
- (2R,3S)-Chlorpheg
Catalog No.:BCC6805
CAS No.:140924-23-8
- 6-Hydroxy-5,6-dehydrosugiol
Catalog No.:BCN3127
CAS No.:140923-35-9
- 4-Chloro-D-phenylalanine
Catalog No.:BCC2637
CAS No.:14091-08-8
- Aminopotentidine
Catalog No.:BCC6761
CAS No.:140873-26-3
- ICI 215,001 hydrochloride
Catalog No.:BCC5688
CAS No.:140850-02-8
- 1-Hydroxymethyl-beta-carboline glucoside
Catalog No.:BCN7026
CAS No.:1408311-12-5
- JNK-IN-7
Catalog No.:BCC1672
CAS No.:1408064-71-0
- 2-Thiouracil
Catalog No.:BCC4752
CAS No.:141-90-2
- Frangulin B
Catalog No.:BCC8175
CAS No.:14101-04-3
- Gama-Tocotrienol
Catalog No.:BCN3720
CAS No.:14101-61-2
- PACOCF3
Catalog No.:BCC7074
CAS No.:141022-99-3
- 1-Hydroxy-2,3,4,7-tetramethoxyxanthone
Catalog No.:BCN6506
CAS No.:14103-09-4
- Xanomeline oxalate
Catalog No.:BCC4146
CAS No.:141064-23-5
- JNK-IN-8
Catalog No.:BCC1673
CAS No.:1410880-22-6
- 12-Hydroxydodecanoic Acid
Catalog No.:BCN8405
CAS No.:505-95-3
- TRAP-6
Catalog No.:BCC3957
CAS No.:141136-83-6
- Epiguajadial B
Catalog No.:BCN4075
CAS No.:1411629-26-9
- 2-(Dimethylaminomethyl)-2-propanol
Catalog No.:BCN1774
CAS No.:14123-48-9
- Btk inhibitor 1
Catalog No.:BCC4238
CAS No.:1412418-47-3
Synthesis and Properties of Energy-Rich Methanofullerenes Containing Norbornadiene and Quadricyclane Moieties.[Pubmed:29533625]
J Org Chem. 2018 Apr 6;83(7):4160-4166.
The energy-rich methanofullerenes were synthesized for the first time by the reaction of fullerene C60 with mono- and bisquadricyclane esters of Malonic acid. The C-C bond cleavage in the quadricyclane moieties of new hybrid molecules takes place in the presence of catalytic amounts of Cu, Pd, and Pt salts or complexes or SiO2 and is accompanied by heat evolution.
Pharmacokinetics and Toxicology of the Neuroprotective e,e,e-Methanofullerene(60)-63-tris Malonic Acid [C3] in Mice and Primates.[Pubmed:29520718]
Eur J Drug Metab Pharmacokinet. 2018 Oct;43(5):543-554.
BACKGROUND AND OBJECTIVES: Fullerene-based compounds are a novel class of molecules being developed for a variety of biomedical applications, with nearly 1000 publications in this area in the last 4 years alone. One such compound, the e,e,e-methanofullerene(60)-63-tris Malonic acid (designated C3), is a potent catalytic superoxide dismutase mimetic which has shown neuroprotective efficacy in a number of animal models of neurologic disease, including Parkinsonian Macaca fascicularis monkeys. The aim of this study was to characterize its toxicity and pharmacokinetics in mice and monkeys. METHODS: To assess pharmacokinetics in mice, we synthesized and administered (14)C-C3 to mice using various routes of delivery, including orally. To assess potential toxicity in primates, serial blood studies and electrocardiograms (ECGs) were obtained from monkeys treated with C3 (3 or 7 mg/kg/day) for 2 months. RESULTS AND CONCLUSIONS: The plasma half-life of C3 was 8.2 +/- 0.2 h, and there was wide tissue distribution, including uptake into brain. The compound was cleared by both hepatic and renal excretion. C3 was quite stable, with minimal metabolism of the compound even after 7 days of treatment. The LD50 in mice was 80 mg/kg for a single intraperitoneal injection, and was > 30 mg/kg/day for sustained administration; therapeutic doses are 1-5 mg/kg/day. For primates, no evidence of renal, hepatic, electrolyte, or hematologic abnormalities were noted, and serial ECGs demonstrated no alteration in cardiac electrical activity. Thus, doses of C3 that have therapeutic efficacy appear to be well tolerated after 2 years (mice) or 2 months (non-human primates) of treatment.
Metabolic Profile of Oral Squamous Carcinoma Cell Lines Relies on a Higher Demand of Lipid Metabolism in Metastatic Cells.[Pubmed:29456966]
Front Oncol. 2018 Feb 2;8:13.
Tumor cells are subjected to a broad range of selective pressures. As a result of the imposed stress, subpopulations of surviving cells exhibit individual biochemical phenotypes that reflect metabolic reprograming. The present work aimed at investigating metabolic parameters of cells displaying increasing degrees of metastatic potential. The metabolites present in cell extracts fraction of tongue fibroblasts and of cell lines derived from human tongue squamous cell carcinoma lineages displaying increasing metastatic potential (SCC9 ZsG, LN1 and LN2) were analyzed by (1)H NMR (nuclear magnetic resonance) spectroscopy. Living, intact cells were also examined by the non-invasive method of fluorescence lifetime imaging microscopy (FLIM) based on the auto fluorescence of endogenous NADH. The cell lines reproducibly exhibited distinct metabolic profiles confirmed by Partial Least-Square Discriminant Analysis (PLS-DA) of the spectra. Measurement of endogenous free and bound NAD(P)H relative concentrations in the intact cell lines showed that ZsG and LN1 cells displayed high heterogeneity in the energy metabolism, indicating that the cells would oscillate between glycolysis and oxidative metabolism depending on the microenvironment's composition. However, LN2 cells appeared to have more contributions to the oxidative status, displaying a lower NAD(P)H free/bound ratio. Functional experiments of energy metabolism, mitochondrial physiology, and proliferation assays revealed that all lineages exhibited similar energy features, although resorting to different bioenergetics strategies to face metabolic demands. These differentiated functions may also promote metastasis. We propose that lipid metabolism is related to the increased invasiveness as a result of the accumulation of malonate, methyl Malonic acid, n-acetyl and unsaturated fatty acids (CH2)n in parallel with the metastatic potential progression, thus suggesting that the NAD(P)H reflected the lipid catabolic/anabolic pathways.
A review on cationic lipids with different linkers for gene delivery.[Pubmed:29454463]
Adv Colloid Interface Sci. 2018 Mar;253:117-140.
Cationic lipids have become known as one of the most versatile tools for the delivery of DNA, RNA and many other therapeutic molecules, and are especially attractive because they can be easily designed, synthesized and characterized. Most of cationic lipids share the common structure of cationic head groups and hydrophobic portions with linker bonds between both domains. The linker bond is an important determinant of the chemical stability and biodegradability of cationic lipid, and further governs its transfection efficiency and cytotoxicity. Based on the structures of linker bonds, they can be grouped into many types, such as ether, ester, amide, carbamate, disulfide, urea, acylhydrazone, phosphate, and other unusual types (carnitine, vinyl ether, ketal, glutamic acid, aspartic acid, Malonic acid diamide and dihydroxybenzene). This review summarizes some research results concerning the nature (such as the structure and orientation of linker groups) and density (such as the spacing and the number of linker groups) of linker bond for improving the chemical stability, biodegradability, transfection efficiency and cytotoxicity of cationic lipid to overcome the critical barriers of in vitro and in vivo transfection.
Iterative Polyketide Synthesis via a Consecutive Carbonyl-Protecting Strategy.[Pubmed:29509410]
J Org Chem. 2018 Apr 6;83(7):4279-4285.
To address the difficulty in protecting a beta-polycarbonyl compound, a method for the sequential protection of elongating carbonyl groups was demonstrated. The iterative chain elongation of a carboxylic acid with Malonic acid half thioester followed by the protection of the resulting beta-ketothioester was performed via the stepwise formation of an isoxazole ring using an O-protected oxime functionality. Yangonin and isosakuranetin were synthesized according to this procedure.
Iron Catalyzed Hydroformylation of Alkenes under Mild Conditions: Evidence of an Fe(II) Catalyzed Process.[Pubmed:29526081]
J Am Chem Soc. 2018 Mar 28;140(12):4430-4439.
Earth abundant, first row transition metals offer a cheap and sustainable alternative to the rare and precious metals. However, utilization of first row metals in catalysis requires harsh reaction conditions, suffers from limited activity, and fails to tolerate functional groups. Reported here is a highly efficient iron catalyzed hydroformylation of alkenes under mild conditions. This protocol operates at 10-30 bar syngas pressure below 100 degrees C, utilizes readily available ligands, and applies to an array of olefins. Thus, the iron precursor [HFe(CO)4](-)[Ph3PNPPh3](+) (1) in the presence of triphenyl phosphine catalyzes the hydroformylation of 1-hexene (S2), 1-octene (S1), 1-decene (S3), 1-dodecene (S4), 1-octadecene (S5), trimethoxy(vinyl)silane (S6), trimethyl(vinyl)silane (S7), cardanol (S8), 2,3-dihydrofuran (S9), allyl Malonic acid (S10), styrene (S11), 4-methylstyrene (S12), 4- iBu-styrene (S13), 4- tBu-styrene (S14), 4-methoxy styrene (S15), 4-acetoxy styrene (S16), 4-bromo styrene (S17), 4-chloro styrene (S18), 4-vinylbenzonitrile (S19), 4-vinylbenzoic acid (S20), and allyl benzene (S21) to corresponding aldehydes in good to excellent yields. Both electron donating and electron withdrawing substituents could be tolerated and excellent conversions were obtained for S11-S20. Remarkably, the addition of 1 mol % acetic acid promotes the reaction to completion within 16-24 h. Detailed mechanistic investigations revealed in situ formation of an iron-dihydride complex [H2Fe(CO)2(PPh3)2] (A) as an active catalytic species. This finding was further supported by cyclic voltammetry investigations and intermediacy of an Fe(0)-Fe(II) species was established. Combined experimental and computational investigations support the existence of an iron-dihydride as the catalyst resting state, which then follows a Fe(II) based catalytic cycle to produce aldehyde.
One-step synthesis of NaLu80-xGdxF4:Yb18(3+)/Er2(3+)(Tm(3+)) upconversion nanoparticles for in vitro cell imaging.[Pubmed:29525097]
Mater Sci Eng C Mater Biol Appl. 2018 May 1;86:56-61.
Upconversion nanoparticles (UCNPs) possess a unique type of photoluminescence (PL) in which lower-energy excitation is converted into higher-energy emission via multi-photon absorption processes. In this work, we have used a facile one-step hydrothermal method promoted water solubility to synthesis NaLuGdF4:Yb(3+)/Er(3+)(Tm(3+)) UCNPs coated with Malonic acid (MA). Scanning electron microscopy images and X-ray diffraction patterns reveal sphere-shaped UCNPs with an average size of ~80nm crystallized in the cubic NaLuF4 structure. The characteristic vibrations of cubic UCNPs have been taken into account by using Fourier-transform infrared spectroscopy. Based on PL studies, we have determined an optimal concentration of Gd(3+) doping. The dependence of upconversion PL intensity on Gd(3+) concentration is discussed via the results of magnetization measurements, which is related to the coupling/uncoupling of Gd(3+) ions. Particularly, our study reveals that carboxyl-functionalized NaLuGdF4:Yb(3+)/Er(3+)(Tm(3+)) UCNPs have a relatively high cell viability with HeLa cells.


