IsobavachinCAS# 31524-62-6 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 31524-62-6 | SDF | Download SDF |
| PubChem ID | 193679 | Appearance | Powder |
| Formula | C20H20O4 | M.Wt | 324.4 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
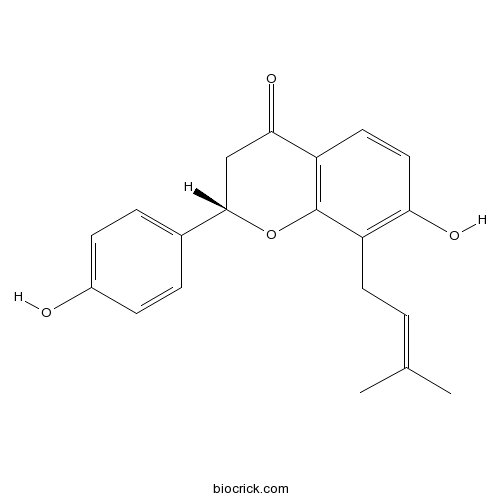
| Chemical Name | (2S)-7-hydroxy-2-(4-hydroxyphenyl)-8-(3-methylbut-2-enyl)-2,3-dihydrochromen-4-one | ||
| SMILES | CC(=CCC1=C(C=CC2=C1OC(CC2=O)C3=CC=C(C=C3)O)O)C | ||
| Standard InChIKey | KYFBXCHUXFKMGQ-IBGZPJMESA-N | ||
| Standard InChI | InChI=1S/C20H20O4/c1-12(2)3-8-15-17(22)10-9-16-18(23)11-19(24-20(15)16)13-4-6-14(21)7-5-13/h3-7,9-10,19,21-22H,8,11H2,1-2H3/t19-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Isobavachin can stimulate osteoblasts proliferation and differentiation; it also can facilitate mouse embryonic stem cells differentiating into neuronal cells, the mechanism involved protein prenylation and, subsequently, phos-ERK activation and the phos-p38 off pathway. Isobavachin possesses estrogen-like activity in MCF-7/BOS cells, it can significantly stimulate the proliferation of MCF-7/BOS cells in a dose-dependent manner. Isobavachin has cytotoxic effects on H4IIE hepatoma and metabolically poorly active C6 glioma cells. |
| Targets | ERK | p38MAPK | Caspase |
| In vitro | Promoting effects of isobavachin on neurogenesis of mouse embryonic stem cells were associated with protein prenylation.[Pubmed: 21441946]Acta Pharmacol Sin. 2011 Apr;32(4):425-32.Some small molecules can induce mouse embryonic stem (ES) cells to differentiate into neuronal cells. Here, we explored the effect of Isobavachin (IBA), a compound with a prenyl group at position 8 of ring A, on promoting neuronal differentiation and the potential role of its protein prenylation. Prenylation enhances cytotoxicity of apigenin and liquiritigenin in rat H4IIE hepatoma and C6 glioma cells.[Pubmed: 17045382 ]Food Chem Toxicol. 2007 Jan;45(1):119-24.Antioxidative as well as cytotoxic effects of the prenylated flavonoids licoflavone C (8-prenylapigenin) and Isobavachin (8-prenylliquiritigenin) were investigated in comparison to the corresponding non-prenylated flavonoids (apigenin, liquiritigenin) and vitexin (apigenin-C8-glucoside) using metabolically active H4IIE hepatoma and metabolically poorly active C6 glioma cells.
|
| Cell Research | Synthesis of four natural prenylflavonoids and their estrogen-like activities.[Pubmed: 17610303 ]Arch Pharm (Weinheim). 2007 Jul;340(7):372-6.
|
| Structure Identification | Phytomedicine. 2014 Mar 15;21(4):400-5.Osteoblasts proliferation and differentiation stimulating activities of the main components of Fructus Psoraleae corylifoliae.[Pubmed: 24220018]Osteoporosis is a disease of bones that leads to an increased risk of fracture. Fructus of Psoralea corylifolia L. (scurfpea fruit) is commonly utilized for treating bone fractures and joint diseases for thousands of years in China. This study was aimed to screen active principles, which might have the potency to stimulate osteoblasts proliferation and differentiation from scurfpea fruit.
|

Isobavachin Dilution Calculator

Isobavachin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.0826 mL | 15.4131 mL | 30.8261 mL | 61.6523 mL | 77.0654 mL |
| 5 mM | 0.6165 mL | 3.0826 mL | 6.1652 mL | 12.3305 mL | 15.4131 mL |
| 10 mM | 0.3083 mL | 1.5413 mL | 3.0826 mL | 6.1652 mL | 7.7065 mL |
| 50 mM | 0.0617 mL | 0.3083 mL | 0.6165 mL | 1.233 mL | 1.5413 mL |
| 100 mM | 0.0308 mL | 0.1541 mL | 0.3083 mL | 0.6165 mL | 0.7707 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Sutherlandin trans-p-coumarate
Catalog No.:BCN5231
CAS No.:315236-68-1
- Ifflaiamine
Catalog No.:BCN7061
CAS No.:31520-95-3
- PAC-1
Catalog No.:BCC3600
CAS No.:315183-21-2
- Acetylheliosupine
Catalog No.:BCN1981
CAS No.:31514-30-4
- Testosterone enanthate
Catalog No.:BCC9169
CAS No.:315-37-7
- Allopurinol
Catalog No.:BCC3720
CAS No.:315-30-0
- Crotaline
Catalog No.:BCN4983
CAS No.:315-22-0
- Sunifiram
Catalog No.:BCC4167
CAS No.:314728-85-3
- 6-Methoxysalicylic Acid
Catalog No.:BCC8288
CAS No.:3147-64-6
- Mebendazole
Catalog No.:BCC9016
CAS No.:31431-39-7
- 4-Amino-3-nitrobenzophenone
Catalog No.:BCC8682
CAS No.:31431-19-3
- Nocodazole
Catalog No.:BCC3826
CAS No.:31430-18-9
- 5-Hydroxyseselin
Catalog No.:BCN3428
CAS No.:31525-75-4
- O-Nornuciferine
Catalog No.:BCN7074
CAS No.:3153-55-7
- Matsukaze-lactone
Catalog No.:BCN7580
CAS No.:3153-73-9
- 1,18-Octadecanediol
Catalog No.:BCN5233
CAS No.:3155-43-9
- Kavain
Catalog No.:BCN8295
CAS No.:3155-48-4
- TC-DAPK 6
Catalog No.:BCC1989
CAS No.:315694-89-4
- STF-62247
Catalog No.:BCC4960
CAS No.:315702-99-9
- JK 184
Catalog No.:BCC3936
CAS No.:315703-52-7
- PTC-209
Catalog No.:BCC5111
CAS No.:315704-66-6
- 4EGI-1
Catalog No.:BCC5337
CAS No.:315706-13-9
- Heraclenol
Catalog No.:BCN5234
CAS No.:31575-93-6
- Berberine Sulphate
Catalog No.:BCC8131
CAS No.:316-41-6
Synthesis of four natural prenylflavonoids and their estrogen-like activities.[Pubmed:17610303]
Arch Pharm (Weinheim). 2007 Jul;340(7):372-6.
Four prenylflavonoids, bavachin 1, Isobavachin 2, 7,4'-dihydroxy-8-prenylflavone 3, and 8-prenylapigenin 4 were synthesized and recognized for possessing estrogen-like activity in MCF-7/BOS cells, as evaluated by an estrogen-screening assay. All compounds significantly stimulated the proliferation of MCF-7/BOS cells in a dose-dependent manner. Isobavachin 2 showed the most potent activity, while bavachin 1 was the weakest. The estrogenic potency of these compounds is ranked as follows: 2 > 4 > 3 > 1.
Promoting effects of isobavachin on neurogenesis of mouse embryonic stem cells were associated with protein prenylation.[Pubmed:21441946]
Acta Pharmacol Sin. 2011 Apr;32(4):425-32.
AIM: Some small molecules can induce mouse embryonic stem (ES) cells to differentiate into neuronal cells. Here, we explored the effect of Isobavachin (IBA), a compound with a prenyl group at position 8 of ring A, on promoting neuronal differentiation and the potential role of its protein prenylation. METHODS: The hanging drop method was employed for embryonic body (EB) formation to mimic embryo development in vivo. The EBs were treated with IBA at a final concentration of 10(-7) mol/L from EB stage (d 4) to d 8+10. Geranylgeranyltransferase I inhibitor GGTI-298 was subsequently used to disrupt protein prenylation. Neuronal subtypes, including neurons and astrocytes, were observed by fluorescence microscopy. Gene and protein expression levels were detected using RT-PCR and Western blot analysis, respectively. RESULTS: With IBA treatment, nestin was highly expressed in the neural progenitors generated from EBs (d 4, d 8+0). EBs then further differentiated into neurons (marked by beta-tubulin III) and astrocytes (marked by GFAP), which were both up-regulated in a time-dependent manner on d 8+5 and d 8+10. Co-treatment with GGTI-298 selectively abolished the IBA-induced neuronal differentiation. Moreover, in the MAPK pathway, p38 and JNK phosphorylation were down-regulated, while ERK phosphorylation was up-regulated after IBA treatment at different neuronal differentiation passages. CONCLUSION: IBA can facilitate mouse ES cells differentiating into neuronal cells. The mechanism involved protein prenylation and, subsequently, phos-ERK activation and the phos-p38 off pathway.
Prenylation enhances cytotoxicity of apigenin and liquiritigenin in rat H4IIE hepatoma and C6 glioma cells.[Pubmed:17045382]
Food Chem Toxicol. 2007 Jan;45(1):119-24.
Antioxidative as well as cytotoxic effects of the prenylated flavonoids licoflavone C (8-prenylapigenin) and Isobavachin (8-prenylliquiritigenin) were investigated in comparison to the corresponding non-prenylated flavonoids (apigenin, liquiritigenin) and vitexin (apigenin-C8-glucoside) using metabolically active H4IIE hepatoma and metabolically poorly active C6 glioma cells. None of the substances showed radical scavenging activities in the 2,2-diphenyl-1-picrylhydrazyl (DPPH)-assay nor were they effective in protection against H2O2-induced intracellular 2',7'-dichlorodihydrofluorescein (H2DCF) oxidation (fluorescent probe for oxidative stress) in H4IIE and C6 cells. When the intrinsic effects of the substances were investigated, licoflavone C and Isobavachin exerted a pronounced toxicity in both H4IIE (IC50 values of 42+/-5 and 96+/-19 micromol/L) and C6 cells (IC50 values of 37+/-6 and 69+/-3 micromol/L) while the non-prenylated analogues as well as the glycosylated derivate vitexin showed almost no cytotoxic effect up to 250 micromol/L. In H4IIE cells the induction of apoptotic cell death by licoflavone C and icobavachin was detected as an activation of caspase 3/7 (6- and 3.3-fold, respectively). Based on these experiments we suggest that C8-prenylation of a flavonoid enhances the cytotoxicity inducing an apoptotic cell death in H4IIE cells without affecting antioxidative properties.
Osteoblasts proliferation and differentiation stimulating activities of the main components of Fructus Psoraleae corylifoliae.[Pubmed:24220018]
Phytomedicine. 2014 Mar 15;21(4):400-5.
Osteoporosis is a disease of bones that leads to an increased risk of fracture. Fructus of Psoralea corylifolia L. (scurfpea fruit) is commonly utilized for treating bone fractures and joint diseases for thousands of years in China. This study was aimed to screen active principles, which might have the potency to stimulate osteoblasts proliferation and differentiation from scurfpea fruit. A HPLC method was established to analyze the main components in scurfpea fruit. Totally 11 compounds have been identified by comparing their retention time with correspondent standard substances. The MTT and ALP methods were utilized for the assay of osteoblasts proliferation and differentiation activity. Icariin, a prenylated flavonoid glycoside was treated as the positive control. Bavachin and Isobavachin significantly stimulated cell proliferation, while bakuchiol exhibited stronger effect to enhance osteoblasts differentiation. All these compounds were found with a characterized structure that in each of their molecule backbones, a prenylated side chain was attached. These results lead to a hypothesis that prenyl group might be crucial to exhibit the activity. The structure-effect relationship of these compounds with prenyl group in mouse primary calvarial osteoblasts needs to be explored in further research.


