CannabinolCAS# 521-35-7 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 521-35-7 | SDF | Download SDF |
| PubChem ID | 2543 | Appearance | Powder |
| Formula | C21H26O2 | M.Wt | 310.43 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
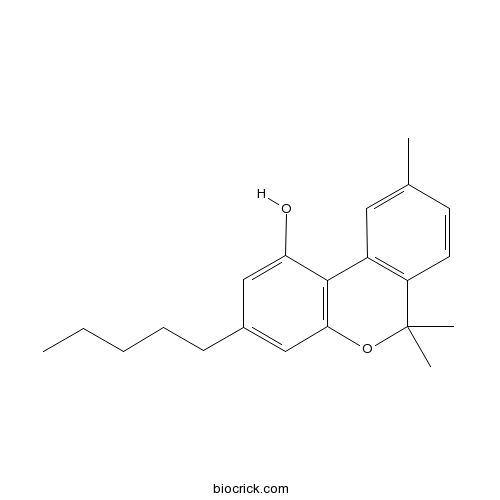
| Chemical Name | 6,6,9-trimethyl-3-pentylbenzo[c]chromen-1-ol | ||
| SMILES | CCCCCC1=CC2=C(C(=C1)O)C3=C(C=CC(=C3)C)C(O2)(C)C | ||
| Standard InChIKey | VBGLYOIFKLUMQG-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C21H26O2/c1-5-6-7-8-15-12-18(22)20-16-11-14(2)9-10-17(16)21(3,4)23-19(20)13-15/h9-13,22H,5-8H2,1-4H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Cannabinol activates capsaicin-sensitive sensory nerves via a CB1 and CB2 cannabinoid receptor-independent mechanism. |
| Targets | Calcium Channel |

Cannabinol Dilution Calculator

Cannabinol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.2213 mL | 16.1067 mL | 32.2134 mL | 64.4268 mL | 80.5335 mL |
| 5 mM | 0.6443 mL | 3.2213 mL | 6.4427 mL | 12.8854 mL | 16.1067 mL |
| 10 mM | 0.3221 mL | 1.6107 mL | 3.2213 mL | 6.4427 mL | 8.0533 mL |
| 50 mM | 0.0644 mL | 0.3221 mL | 0.6443 mL | 1.2885 mL | 1.6107 mL |
| 100 mM | 0.0322 mL | 0.1611 mL | 0.3221 mL | 0.6443 mL | 0.8053 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Sciadopitysin
Catalog No.:BCN5662
CAS No.:521-34-6
- Bilobetin
Catalog No.:BCN5661
CAS No.:521-32-4
- Stanolone
Catalog No.:BCC9153
CAS No.:521-18-6
- Androstenediol
Catalog No.:BCC8828
CAS No.:521-17-5
- Dromostanolone propionate
Catalog No.:BCC8954
CAS No.:521-12-0
- Mestanolone
Catalog No.:BCC9022
CAS No.:521-11-9
- 2-Hydroxy-7-O-methylscillascillin
Catalog No.:BCN5659
CAS No.:52096-50-1
- H-Val-pNA
Catalog No.:BCC3139
CAS No.:52084-13-6
- NCS-382
Catalog No.:BCC6794
CAS No.:520505-01-5
- Isoschaftoside
Catalog No.:BCN3011
CAS No.:52012-29-0
- Medroxyprogesterone
Catalog No.:BCC5231
CAS No.:520-85-4
- Echimidine
Catalog No.:BCN1967
CAS No.:520-68-3
- Pedicin
Catalog No.:BCN4845
CAS No.:521-51-7
- Vulpic acid
Catalog No.:BCN6546
CAS No.:521-52-8
- Physcion
Catalog No.:BCN5663
CAS No.:521-61-9
- Frangulin A
Catalog No.:BCC8174
CAS No.:521-62-0
- Cinnamoylcocaine
Catalog No.:BCN1429
CAS No.:521-67-5
- Broxyquinoline
Catalog No.:BCC4642
CAS No.:521-74-4
- Karanjin
Catalog No.:BCN8370
CAS No.:521-88-0
- 2,4-Dihydroxy-6-methoxy-3-formylacetophenone
Catalog No.:BCN1430
CAS No.:52117-67-6
- 3-O-Acetylpinobanksin
Catalog No.:BCN5660
CAS No.:52117-69-8
- H-Tyr(Bzl)-OBzl.HCl
Catalog No.:BCC3131
CAS No.:52142-01-5
- Piperitol
Catalog No.:BCN3968
CAS No.:52151-92-5
- 7-Hydroxy-2,3,4,5-tetrahydro-1H-benzofuro[2,3-c]azepin-1-one
Catalog No.:BCC3960
CAS No.:521937-07-5
Delta 9-tetrahydrocannabinol and cannabinol activate capsaicin-sensitive sensory nerves via a CB1 and CB2 cannabinoid receptor-independent mechanism.[Pubmed:12040079]
J Neurosci. 2002 Jun 1;22(11):4720-7.
Although Delta(9)-tetrahydroCannabinol (THC) produces analgesia, its effects on nociceptive primary afferents are unknown. These neurons participate not only in pain signaling but also in the local response to tissue injury. Here, we show that THC and Cannabinol induce a CB(1)/CB(2) cannabinoid receptor-independent release of calcitonin gene-related peptide from capsaicin-sensitive perivascular sensory nerves. Other psychotropic cannabinoids cannot mimic this action. The vanilloid receptor antagonist ruthenium red abolishes the responses to THC and Cannabinol. However, the effect of THC on sensory nerves is intact in vanilloid receptor subtype 1 gene knock-out mice. The THC response depends on extracellular calcium but does not involve known voltage-operated calcium channels, glutamate receptors, or protein kinases A and C. These results may indicate the presence of a novel cannabinoid receptor/ion channel in the pain pathway.
Inhibition of the cyclic AMP signaling cascade and nuclear factor binding to CRE and kappaB elements by cannabinol, a minimally CNS-active cannabinoid.[Pubmed:9605425]
Biochem Pharmacol. 1998 Apr 1;55(7):1013-23.
Immune suppression by cannabinoids has been widely demonstrated in a variety of experimental models. The identification of two major types of G-protein-coupled cannabinoid receptors expressed on leukocytes, CB1 and CB2, has provided a putative mechanism of action for immune modulation by cannabinoid compounds. Ligand binding to both receptors negatively regulates adenylate cyclase, thereby lowering intracellular cyclic AMP (cAMP) levels. In the present studies, we demonstrated that Cannabinol (CBN), a ligand that exhibits higher binding affinity for CB2, modulates immune responses and cAMP-mediated signal transduction in mouse lymphoid cells. Direct addition of CBN to naive cultured splenocytes produced a concentration-dependent inhibition of lymphoproliferative responses to anti-CD3, lipopolysaccharide, and phorbol-12-myristate-13-acetate/ionomycin stimulation. Similarly, a concentration-related inhibition of the in vitro anti-sheep red blood cell IgM antibody forming cell response was also observed by CBN. Evaluation of cAMP signaling in the presence of CBN showed a rapid and concentration-related inhibition of adenylate cyclase activity in both splenocytes and thymocytes. This decrease in intracellular cAMP levels produced by CBN resulted in a reduction of protein kinase A activity, consequently leading to an inhibition of transcription factor binding to the cAMP response element and kappaB motifs in both cell preparations. Collectively, these results demonstrate that CBN, a cannabinoid with minimal CNS activity, inhibited both cAMP signal transduction and immune function, further supporting the involvement of CB2 receptors in immune modulation by cannabimimetic agents.
Cannabinol derivatives: binding to cannabinoid receptors and inhibition of adenylylcyclase.[Pubmed:9379442]
J Med Chem. 1997 Sep 26;40(20):3228-33.
Several derivatives of Cannabinol and the 1,1-dimethylheptyl homolog (DMH) of Cannabinol were prepared and assayed for binding to the brain and the peripheral cannabinoid receptors (CB1 and CB2), as well as for activation of CB1- and CB2-mediated inhibition of adenylylcyclase. The DMH derivatives were much more potent than the pentyl (i.e., Cannabinol) derivatives. 11-HydroxyCannabinol (4a) was found to bind potently to both CB1 and CB2 (Ki values of 38.0 +/- 7.2 and 26.6 +/- 5.5 nM, respectively) and to inhibit CB1-mediated adenylylcyclase with an EC50 of 58.1 +/- 6.2 nM but to cause only 20% inhibition of CB2-mediated adenylylcyclase at 10 microM. It behaves as a specific, though not potent, CB2 antagonist. 11-HydroxyCannabinol-DMH (4b) is a very potent agonist for both CB1 and CB2 (Ki values of 100 +/- 50 and 200 +/- 40 pM; EC50 of adenylylcyclase inhibition 56.2 +/- 4.2 and 207.5 +/- 27.8 pM, respectively).


