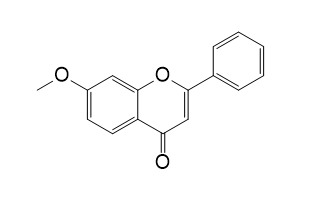
7-MethoxyflavoneCAS# 22395-22-8 |
Quality Control & MSDS
Package In Stock
Number of papers citing our products

| Cas No. | 22395-22-8 | SDF | Download SDF |
| PubChem ID | N/A | Appearance | Powder |
| Formula | C16H12O3 | M.Wt | 252.2 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 7-Methoxyflavone has peripheral antinociceptive activity. | |||||

7-Methoxyflavone Dilution Calculator

7-Methoxyflavone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.9651 mL | 19.8255 mL | 39.6511 mL | 79.3021 mL | 99.1277 mL |
| 5 mM | 0.793 mL | 3.9651 mL | 7.9302 mL | 15.8604 mL | 19.8255 mL |
| 10 mM | 0.3965 mL | 1.9826 mL | 3.9651 mL | 7.9302 mL | 9.9128 mL |
| 50 mM | 0.0793 mL | 0.3965 mL | 0.793 mL | 1.586 mL | 1.9826 mL |
| 100 mM | 0.0397 mL | 0.1983 mL | 0.3965 mL | 0.793 mL | 0.9913 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Oxyacanthine hydrochloride
Catalog No.:BCN9997
CAS No.:15352-74-6
- 3,4-Dimethoxychalcone
Catalog No.:BCN9996
CAS No.:5416-71-7
- 2',3,5,7-Tetrahydroxyflavone
Catalog No.:BCN9995
CAS No.:480-15-9
- 4-Hydroxyquinoline
Catalog No.:BCN9994
CAS No.:611-36-9
- Cytochalasin C
Catalog No.:BCN9993
CAS No.:22144-76-9
- Geranylacetate
Catalog No.:BCN9992
CAS No.:105-87-3
- Daclatasvir
Catalog No.:BCN9991
CAS No.:1009119-64-5
- Phloroglucinol aldehyde triethylether
Catalog No.:BCN9990
CAS No.:59652-88-9
- 3-Octyl alcohol
Catalog No.:BCN9989
CAS No.:589-98-0
- trans-Aconitic acid
Catalog No.:BCN9988
CAS No.:4023-65-8
- Quercetin-3'-glucoside
Catalog No.:BCN9987
CAS No.:19254-30-9
- 1,3-Diphenyl-2-propen-1-one
Catalog No.:BCN9986
CAS No.:94-41-7
- trans-5-Hydroxyferulic acid
Catalog No.:BCN9999
CAS No.:110642-42-7
- 8-p-Coumaroylharpagide
Catalog No.:BCN0152
CAS No.:87686-74-6
- 2-(4-Methoxybenzal)acetophenone
Catalog No.:BCN0001
CAS No.:959-33-1
- N-Methylcolchicine
Catalog No.:BCN0002
CAS No.:7336-40-5
- Hydrocotarnine hydrobromide
Catalog No.:BCN0003
CAS No.:5985-00-2
- 8-Acetyl-7-methoxycoumarin
Catalog No.:BCN0004
CAS No.:89019-07-8
- Shatavarin IV
Catalog No.:BCN0005
CAS No.:84633-34-1
- N-trans-p-Coumaroyltyrosine
Catalog No.:BCN0006
CAS No.:77201-66-2
- Chamigrenol
Catalog No.:BCN0007
CAS No.:19822-80-1
- Harmol hydrochloride monohydrate
Catalog No.:BCN0008
CAS No.:40580-83-4
- Daidzein 7-O-glucuronide
Catalog No.:BCN0009
CAS No.:38482-80-3
- Chamaemeloside
Catalog No.:BCN0010
CAS No.:173356-77-9
Flavonoid glycosides from the rhizomes of Pronephrium penangianum.[Pubmed:32862046]
Phytochemistry. 2020 Nov;179:112500.
Six flavonoid glycosides jixueqisus A-F, together with nine known flavonoids, were isolated from the rhizomes of the fern Pronephrium penangianum. Among them, two red pigments jixueqisus A and B possess the same rare 6,8-dimethyl-2-phenyl-7H-1-benzopyran-7-one skeleton (a long conjugated system). Jixueqisu Cwas a dihydrochalcone glycoside, jixueqisu D was a chalcone glycoside, jixueqisu E was an aurone glycoside, and jixueqisu F was a flavonone glycoside. Interestingly, jixueqisus D-F, (2S)-5,2',5'-trihydroxy-7-methoxyflavanone and 5,2',5'-trihydroxy-7-Methoxyflavone possessed a 2,5-dihydroxy substituted benzene ring (B-ring). Their structures were elucidated by various spectroscopic and chemical methods. Furthermore, the plausible biosynthetic pathways of jixueqisus A-F were discussed, respectively. All isolated compounds were evaluated for their activities against the proliferation of MCF-7, HepG-2, HCT-116 and BGC-823 tumor cell lines, four known flavane-4-ol glycosides, abacopterins A and C, eruberin B and triphyllin A, exhibited moderate activities to various cell lines.
Crystal structures of the flavonoid Oroxylin A and the regioisomers Negletein and Wogonin.[Pubmed:32367831]
Acta Crystallogr C Struct Chem. 2020 May 1;76(Pt 5):490-499.
The flavonoid Oroxylin A (6-methoxychrysin or 5,7-dihydroxy-6-methoxy-2-phenyl-4H-chromen-4-one, C16H12O5) and its regioisomers are of increasing interest for a variety of bioactive functions and their pharmaceutical formulation is of importance. Previous difficulties in the separation and misidentification of Oroxylin A from its regioisomers Wogonin (8-methoxychrysin or 5,7-dihydroxy-8-methoxy-2-phenyl-4H-chromen-4-one) and Negletein (5,6-dihydroxy-7-Methoxyflavone or 5,6-dihydroxy-7-methoxy-2-phenyl-4H-chromen-4-one) render its full structural and powder X-ray characterization highly desirable. The low-temperature (100 K) crystal structures of Oroxylin A, Negletein and Wogonin sesquihydrate are reported for the first time. Wogonin crystallizes in two related but distinct hydrated forms. These have very similar powder diffractograms, indicating that such issues need to be addressed for its pharmaceutical formulation.
A systematic analysis of natural alpha-glucosidase inhibitors from flavonoids of Radix scutellariae using ultrafiltration UPLC-TripleTOF-MS/MS and network pharmacology.[Pubmed:32143602]
BMC Complement Med Ther. 2020 Mar 6;20(1):72.
BACKGROUND: Flavonoids from plant medicines are supposed to be viable alternatives for the treatment of type 2 diabetes (T2D) as less toxicity and side effects. Radix scutellariae (RS) is a widely used traditional medicine in Asia. It has shown great potential in the research of T2D. However, the pharmacological actions remain obscured due to the complex chemical nature of plant medicines. METHODS: In the present study, a systematic method combining ultrafiltration UPLC-TripleTOF-MS/MS and network pharmacology was developed to screen alpha-glucosidase inhibitors from flavonoids of RS, and explore the underlying mechanism for the treatment of T2D. RESULTS: The n-butanol part of ethanol extract from RS showed a strong alpha-glucosidase inhibition activity (90.55%, IC50 0.551 mg/mL) against positive control acarbose (90.59%, IC50 1.079 mg/mL). A total of 32 kinds of flavonoids were identified from the extract, and their ESI-MS/MS behaviors were elucidated. Thirteen compounds were screened as alpha-glucosidase inhibitors, including viscidulin III, 2',3,5,6',7-pentahydroxyflavanone, and so on. A compound-target-pathway (CTP) network was constructed by integrating these alpha-glucosidase inhibitors, target proteins, and related pathways. This network exhibited an uneven distribution and approximate scale-free property. Chrysin (k = 87), 5,8,2'-trihydroxy-7-Methoxyflavone (k = 21) and wogonin (k = 20) were selected as the main active constituents with much higher degree values. A protein-protein interaction (PPI) weighted network was built for target proteins of these alpha-glucosidase inhibitors and drug targets of T2D. PPARG (Cd = 0.165, Cb = 0.232, Cc = 0.401), ACACB (Cd = 0.155, Cb = 0.184, Cc = 0.318), NFKB1 (Cd = 0.233, Cb = 0.161, Cc = 0.431), and PGH2 (Cd = 0.194, Cb = 0.157, Cc = 0.427) exhibited as key targets with the highest scores of centrality indices. Furthermore, a core subnetwork was extracted from the CTP and PPI weighted network. Type II diabetes mellitus (hsa04930) and PPAR signaling pathway (hsa03320) were confirmed as the critical pathways. CONCLUSIONS: These results improved current understanding of natural flavonoids on the treatment of T2D. The combination of ultrafiltration UPLC-TripleTOF-MS/MS and network pharmacology provides a novel strategy for the research of plant medicines and complex diseases.
Peptoid Helix Displaying Flavone and Porphyrin: Synthesis and Intramolecular Energy Transfer.[Pubmed:31657570]
J Org Chem. 2020 Feb 7;85(3):1392-1400.
Natural light-harvesting complexes (LHCs) absorb a broad spectrum of sunlight using a collection of photosynthetic pigments whose spatial arrangement is controlled by a protein matrix and exhibit efficient energy transfer. We constructed a novel light-harvesting protein mimic, which absorbs light in the UV to visible region (280-700 nm) by displaying flavone and porphyrin on a peptoid helix. First, an efficient synthesis of 4'-derivatized 7-Methoxyflavone (7-MF, 3 and 4) was developed. The flavone-porphyrin-peptoid conjugate (FPPC) was then prepared via Miyaura borylation on a resin-bound peptoid followed by Suzuki coupling between the peptoid and pigment. Circular dichroism spectroscopy indicated that the FPPC underwent helix-to-loop conversion of the peptoid scaffold upon changing the solvent conditions. A distinct intramolecular energy transfer was observed from 7-MF to porphyrin with greater efficiency in the helix than that in the loop conformation of the peptoid, whereas no clear evidence of energy transfer was obtained for unstructured FPPC. We thus demonstrate the value of the helical peptoid, which provided a controlled orientation for 7-MF and porphyrin and modulated the energy transfer efficiency via conformational switching. Our work provides a way to construct a sophisticated LHC mimic with enhanced coverage of the solar spectrum and controllable energy transfer efficiency.
5-Hydroxy-7-methoxyflavone derivatives from Kaempferia parviflora induce skeletal muscle hypertrophy.[Pubmed:30680186]
Food Sci Nutr. 2018 Nov 20;7(1):312-321.
Skeletal muscle plays a critical role in locomotion and energy metabolism. Maintenance or enhancement of skeletal muscle mass contributes to the improvement of mobility and prevents the development of metabolic diseases. The extracts from Kaempferia parviflora rhizomes contain at least ten methoxyflavone derivatives that exhibit enhancing effects on ATP production and glucose uptake in skeletal muscle cells. In the present study, we investigated the effects of ten K. parviflora-derived methoxyflavone derivatives (six 5,7-dimethoxyflavone (DMF) derivatives and four 5-hydroxy-7-Methoxyflavone (HMF) derivatives) on skeletal muscle hypertrophy. Murine C2C12 myotubes and senescence-accelerated mouse-prone 1 (SAMP1) mice treated with methoxyflavones were used as experimental models to determine the effects of HMF derivatives on myotube diameter and size and muscle mass. The four HMF derivatives, but not the six DMF derivatives, increased myotube diameter. The 5-hydroxyflavone, 7-Methoxyflavone, and 5,7-dihydroxyflavone had no influence on myotube size, a result that differed from HMF. Dietary administration of the mixture composed of the four HMF derivatives resulted in increase in the soleus muscle size and mass in SAMP1 mice. HMF derivatives also promoted protein synthesis in myotubes, and treatment with the intracellular Ca(2+) chelator BAPTA-AM, which depletes intracellular Ca(2+) levels, inhibited this promotion. Furthermore, BAPTA-AM inhibited HMF-promoted protein synthesis even when myotubes were incubated in Ca(2+)-free medium. These results indicate that HMF derivatives induce myotube hypertrophy and that both the 5-hydroxyl group and the 7-methoxy group in the flavones are necessary for myotube hypertrophy. Furthermore, these results suggest that HMF-induced protein synthesis requires intracellular Ca(2+), but not extracellular Ca(2+).
Methylation and Acetylation Enhanced the Antidiabetic Activity of Some Selected Flavonoids: In Vitro, Molecular Modelling and Structure Activity Relationship-Based Study.[Pubmed:30445784]
Biomolecules. 2018 Nov 15;8(4). pii: biom8040149.
Flavonoids have been reported to exert antihyperglycemic effects and have potential to enhance the current therapy options against type 2 diabetes mellitus. However, the structure activity relationships (SAR) studies of flavonoids against this disease have not been thoroughly comprehended. Hence, in the present study, 14 structurally related flavonoids viz. wogonin, techtochrysin, norwogonin, isoscutellarein, hypolaetin, kaempferol, quercetin, methyl ether of wogonin, acetate of wogonin, acetate of norwogonin, 8-hydroxy-7-Methoxyflavone, chrysin, (+)-catechin and (-)-epicatechin were taken into account for in vitro antidiabetic evaluation. Cell viability of RIN-5F pancreatic cells and 3T3-L1 pre-adipocyte cells was initially tested, then an insulin secretion assay of RIN-5F as well as adipogenesis and glucose uptake measurements of adipocyte were investigated. Subsequently, protein expressions study through adipokines measurement (leptin, adiponectin, TNF-alpha, RBP-4) via enzyme-linked immunosorbent assay (ELISA) kit, Western blotting analysis against GLUT4 and C/EBP-alpha as well as molecular docking against GLUT1 were analyzed. The results from cell culture antidiabetic assays (insulin secretion, adipogenesis, and glucose uptake), protein expressions and molecular docking pointed that the methoxy group at position C-8 is responsible for antidiabetic property of selected flavonoids via glucose uptake mechanism indicated by up regulation of GLUT4 and C/EBP-alpha expressions. The mechanism could be enhanced by the addition of an acetate group at C-5 and C-7 of the flavone skeleton.
Isoflavones from Camphorosma lessingii Inhibit the Organic Anion Transporters OAT1 and OAT3.[Pubmed:30248705]
Planta Med. 2019 Feb;85(3):225-230.
Phytochemical investigation of Camphorosma lessingii has resulted in the isolation of four previously unreported isoflavones (1: -4: ) and eight known compounds (5: -12: ). Nine of these compounds (1: -6, 8: -10: ) are reported for the first time from members of the family Amaranthaceae. The structures of all isolated compounds were determined by spectroscopic methods, primarily one-dimensional and two-dimensional nuclear magnetic resonance and mass spectrometry. The absolute configuration of 6: was confirmed by circular dichroism. Inhibition of the organic anion transporters, OAT1 and OAT3, by the isolated compounds was evaluated. Among them, 7, 2'-dihydroxy- 6,8-dimethoxyisoflavone (1: ), 2'-hydroxy-6,7,8-trimethoxyisoflavone (2: ), 6,2'-dihydroxy-7,8-dimethoxyisoflavone (3: ), and 7-Methoxyflavone (5: ) showed a significant inhibitory effect on 6-carboxyfluorescein uptake mediated by OAT1 and OAT3.
Phytochemical Study and Evaluation of the Cytotoxic Properties of Methanolic Extract from Baccharis obtusifolia.[Pubmed:30155301]
Int J Med Chem. 2018 Aug 1;2018:8908435.
Some species of the Baccharis genus have been shown to possess important biomedical properties, including cytotoxic activity. In this study, we examined the cytotoxic effect of methanol extract from Baccharis obtusifolia (Asteraceae) in cancer cell lines of prostate (PC-3), colon (RKO), astrocytoma (D-384), and breast (MCF-7). The methanolic extract displayed the largest substantial cytotoxic effect in lines of colon cancer (RKO) and cerebral astrocytoma (D-384). Chromatographic purification of the B. obtusifolia methanolic extract led to the isolation and identification of 5,4'-dihydroxy-7-Methoxyflavone (1) and 5-hydroxy-7,4'-dimethoxyflavone (2) compounds of the flavonoid type.
Demethylation of Polymethoxyflavones by Human Gut Bacterium, Blautia sp. MRG-PMF1.[Pubmed:28211698]
J Agric Food Chem. 2017 Mar 1;65(8):1620-1629.
Polymethoxyflavones (PMFs) were biotransformed to various demethylated metabolites in the human intestine by the PMF-metabolizing bacterium, Blautia sp. MRG-PMF1. Because the newly formed metabolites can have different biological activities, the pathways and regioselectivity of PMF bioconversion were investigated. Using an anaerobic in vitro study, 12 PMFs, 5,7-dimethoxyflavone (5,7-DMF), 5-hydroxy-7-Methoxyflavone (5-OH-7-MF), 3,5,7-trimethoxyflavone (3,5,7-TMF), 5-hydroxy-3,7-dimethoxyflavone (5-OH-3,7-DMF), 5,7,4'-trimethoxyflavone (5,7,4'-TMF), 5-hydroxy-7,4'-dimethoxyflavone (5-OH-7,4'-DMF), 3,5,7,4'-tetramethoxyflavone (3,5,7,4'-TMF), 5-hydroxy-3,7,4'-trimethoxyflavone (5-OH-3,7,4'-TMF), 5,7,3',4'-tetramethoxyflavone (5,7,3',4'-TMF), 3,5,7,3',4'-pentamethoxyflavone (3,5,7,3',4'-PMF), 5-hydroxy-3,7,3',4'-tetramethoxyflavone (5-OH-3,7,3',4'-TMF), and 5,3'-dihydroxy-3,7,4'-trimethoxyflavone (5,3'-diOH-3,7,4'-TMF), were converted to chrysin, apigenin, galangin, kaempferol, luteolin, and quercetin after complete demethylation. The time-course monitoring of PMF biotransformations elucidated bioconversion pathways, including the identification of metabolic intermediates. As a robust flavonoid demethylase, regioselectivity of PMF demethylation generally followed the order C-7 > C-4' approximately C-3' > C-5 > C-3. PMF demethylase in the MRG-PMF1 strain was suggested as a Co-corrinoid methyltransferase system, and this was supported by the experiments utilizing other methyl aryl ether substrates and inhibitors.
[Studies on flavonoids from Cirsium setosum].[Pubmed:28875641]
Zhongguo Zhong Yao Za Zhi. 2016 Mar;41(5):868-873.
To investigate the chemical constituents of ethyl acetate from Cirsium setosum, fifteen flavonoids were obtained by column chromatography on silica gel, MCI, Sephadex LH-20, and preparative HPLC. Their structures were identified as 4',5,6-trihydroxy-7-Methoxyflavone(1), 4',5-dihydroxy-7,8-dimethoxyflavone(2), sorbifolin-6-O-beta-glucopyranoside(3), kaempferol-7-O-alpha-L-rhamnoside(4), kaempferol(5), quercetin-3-O-beta-D-glucosyl-7-O-alpha-L-rhamnoside(6), myricetin(7), myricetin-3-O-beta-D-glucoside(8), 5,7- dihydroxy -3',4'- dimethoxyflavone(9), 3',4',5- trihydroxy-3,7-dimethoxyflavone(10), 3',3,4',5-tetrahydroxy-7-Methoxyflavone(11), 3'-hydroxy-4',5,7-trimethoxyflavone(12), 7-hydroxy-3',4',5-trimethoxyflavone(13), 4',5-dihydroxy-2',3',7,8-tetramethoxylflavone(14), and 5-hydroxy-2',3',7,8-tetramethoxylflavone(15) by spectroscopic data analysis. All compounds were isolated from this plant for the first time.Compounds(1-15) were evaluated for their hypoglycemic activities by PTP1B enzyme model. Among them, compounds 2, 12, and 14 showed significant PTP1B inhibitory activities with IC(5)(0) values of 2.54, 1.85, 2.11 mumol*L(-)(1), respectively.
Isolation of Flavonoids from Deguelia duckeana and Their Effect on Cellular Viability, AMPK, eEF2, eIF2 and eIF4E.[Pubmed:26861281]
Molecules. 2016 Feb 6;21(2). pii: molecules21020192.
Preparations of Deguelia duckeana, known in Brazil as timbo, are used by indigenous people to kill fish. Reinvestigation of its extracts resulted in the isolation and identification of 11 known flavonoids identified as 3,5,4'-trimethoxy-4-prenylstilbene (1), 4-methoxyderricidine (2), lonchocarpine (3), 4-hydroxylonchocarpine (4), 4-methoxylonchocarpine (5), 5-hydroxy-4',7-dimethoxy-6-prenylflavanone (6), 4'-hydroxyisolonchocarpine (7), 4'-methoxyisolonchocarpine (8), 3',4',7-trimethoxyflavone (9), 3',4'-methylenedioxy-7-Methoxyflavone (10), and 2,2-dimethyl-chromone-5,4'-hydroxy-5'-methoxyflavone (11). Except for 1, 3, and 4 all of these flavonoids have been described for the first time in D. duckeana and the flavanone 6 for the first time in nature. Compounds 2, 3, 4, 7, 9, and 10 were studied for their potential to induce cell death in neuronal SK-N-SH cells. Only the chalcone 4 and the flavanone 7 significantly induced lactate dehydrogenase (LDH) release, which was accompanied by activation of caspase-3 and impairment of energy homeostasis in the MTT assay and may explain the killing effect on fish. Interestingly, the flavone 10 reduced cell metabolism in the MTT assay without inducing cytotoxicity in the LDH assay. Furthermore, the flavonoids 2, 3, 4, 7, and 10 induced phosphorylation of the AMP-activated protein kinase (AMPK) and the eukaryotic elongation factor 2 (eEF2). The initiation factor eIF4E was dephosphorylated in the presence of these compounds. The initiation factor eIF2alpha was not affected. Further studies are needed to elucidate the importance of the observed effects on protein synthesis and potential therapeutic perspectives.
Antioxidant C-glycosylflavones of Drymaria cordata (Linn.) Willd.[Pubmed:26642770]
Arch Pharm Res. 2016 Jan;39(1):43-50.
A new C-glycosylflavone, drymaritin E (6-C-(3-keto-beta-digitoxopyranosyl)-4'-O-(beta-D-glucopyranosyl)-7-methoxyl-5,4' -dihydroxylflavone) 1 was isolated from the oily upper phase (SU) of the MeOH extract from aerial parts of Drymaria cordata together with two known compounds (cassiaoccidentalin A 2 and anemonin 3) and an inseparable mixture of two known C-glycosylflavones 5,4'-dihydroxy-7-Methoxyflavone-6-C-(2''-O-alpha-L-rhamnopyranosyl)-beta-D-glucop yranoside 4a and 5,7,3',4'-tetrahydroxyflavone-6-C-(2''-O-alpha-L-rhamnopyranosyl)-beta-D-glucopyr anoside 4b. The alkaline hydrolysis of 3 led to a new hemisynthetic derivative, sodium anemonate (sodium 2-((1'E) 2'-sodium-carboxylate-vinyl)-5-oxo-cyclohex-1-ene carboxylate) 3a. The chemical structures were determined by spectroscopic methods ((1)H NMR, (13)C NMR, (1)H-(1)H COSY, HMBC, HSQC, and NOESY) and mass spectrometry (ESI-MS). C-glycosylflavones had significant free radical-scavenging activities on the radical 2,2-diphenyl-1-picrylhydrazyl (DPPH). However, SU and compounds 3 and 3a exhibited no activity. In particular, compound 1 exhibited a concentration-dependent radical scavenging activity on DPPH with EC50 of 31.43 microg/mL.
Enhancement of energy production by black ginger extract containing polymethoxy flavonoids in myocytes through improving glucose, lactic acid and lipid metabolism.[Pubmed:26581843]
J Nat Med. 2016 Apr;70(2):163-72.
Enhancement of muscular energy production is thought to improve locomotive functions and prevent metabolic syndromes including diabetes and lipidemia. Black ginger (Kaempferia parviflora) has been cultivated for traditional medicine in Thailand. Recent studies have shown that black ginger extract (KPE) activated brown adipocytes and lipolysis in white adipose tissue, which may cure obesity-related dysfunction of lipid metabolism. However, the effect of KPE on glucose and lipid utilization in muscle cells has not been examined yet. Hence, we evaluated the effect of KPE and its constituents on energy metabolism in pre-differentiated (p) and differentiated (d) C2C12 myoblasts. KPE (0.1-10 mug/ml) was added to pC2C12 cells in the differentiation process for a week or used to treat dC2C12 cells for 24 h. After culturing, parameters of glucose and lipid metabolism and mitochondrial biogenesis were assessed. In terms of the results, KPE enhanced the uptake of 2-deoxyglucose and lactic acid as well as the mRNA expression of glucose transporter (GLUT) 4 and monocarboxylate transporter (MCT) 1 in both types of cells. The expression of peroxisome proliferator-activated receptor gamma coactivator (PGC)-1alpha was enhanced in pC2C12 cells. In addition, KPE enhanced the production of ATP and mitochondrial biogenesis. Polymethoxy flavonoids in KPE including 5-hydroxy-7-Methoxyflavone, 5-hydroxy-3,7,4'-trimethoxyflavone and 5,7-dimethoxyflavone enhanced the expression of GLUT4 and PGC-1alpha. Moreover, KPE and 5,7-dimethoxyflavone enhanced the phosphorylation of 5'AMP-activated protein kinase (AMPK). In conclusion, KPE and its polymethoxy flavonoids were found to enhance energy metabolism in myocytes. KPE may improve the dysfunction of muscle metabolism that leads to metabolic syndrome and locomotive dysfunction.
Anti-austeritic Constituents of the Congolese Medicinal Plant Aframomum melegueta.[Pubmed:26197535]
Nat Prod Commun. 2015 Jun;10(6):997-9.
In the course of our search for anticancer agents based on a novel anti-austerity strategy, we found that the CHCl3 extract of the roots of Aflamomum melegueta (Zingiberaceae), collected in the Democratic Republic of Congo, killed PANC-1 human pancreatic cancer cells preferentially in nutrient-deprived medium (NDM). Phytochemical investigation of the CHCl3 extract led to the isolation of seven known compounds [(-)-buplerol (1), (-)-arctigenin (2), (E)-14-hydroxy-15-norlabda-8(17),12-dien-16-al (3), labda-8(17),12-dien-15,16-dial (4), 16-oxo-8(17),12(E)-labdadien-15-oic acid (5), 5-hydroxy-7-Methoxyflavone (6), and apigenin (7)]. In addition to the previously reported preferentially cytotoxic compound, (-)-arctigenin (2, PC50 0.5 muM), (-)-buplerol (1) also displayed potent preferential cytotoxicity with a PC50 value of 8.42 muM and triggered apoptosis-like PANC-1 cell death in NDM.


