Angelica decursiva
Angelica decursiva
1. The products in our compound library are selected from thousands of unique natural products; 2. It has the characteristics of diverse structure, diverse sources and wide coverage of activities; 3. Provide information on the activity of products from major journals, patents and research reports around the world, providing theoretical direction and research basis for further research and screening; 4. Free combination according to the type, source, target and disease of natural product; 5. The compound powder is placed in a covered tube and then discharged into a 10 x 10 cryostat; 6. Transport in ice pack or dry ice pack. Please store it at -20 °C as soon as possible after receiving the product, and use it as soon as possible after opening.
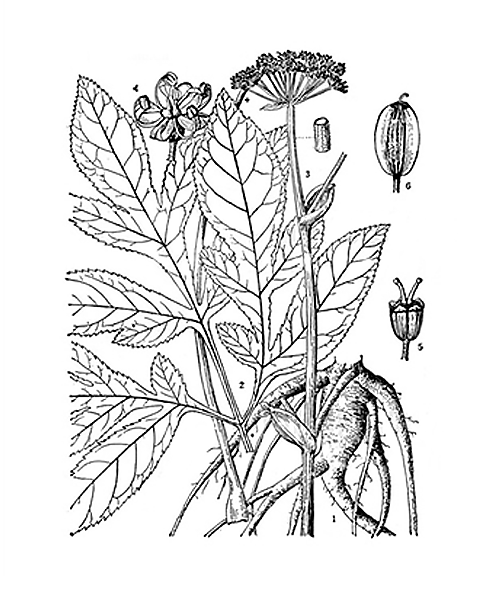
Natural products/compounds from Angelica decursiva
- Cat.No. Product Name CAS Number COA
-
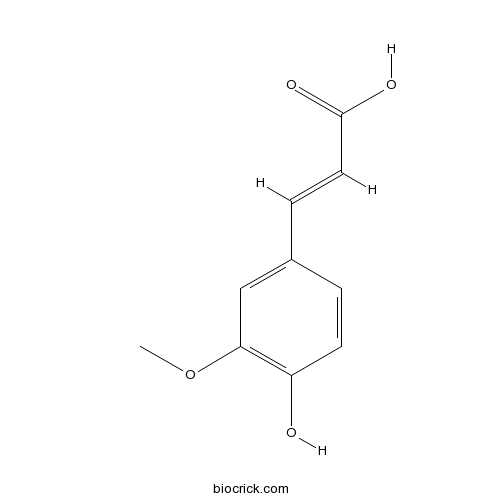
BCN5948
Ferulic acid1135-24-6
Instructions
-
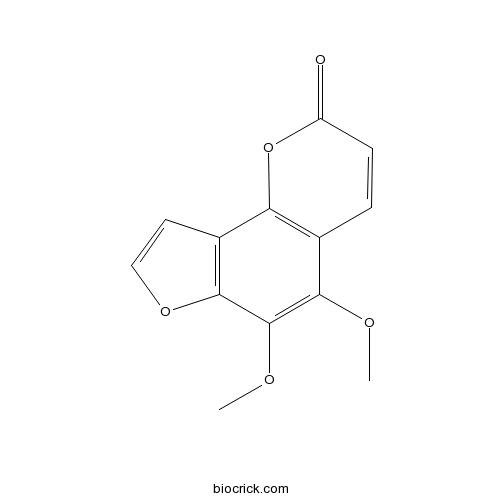
BCN6168
Pimpinellin131-12-4
Instructions
-
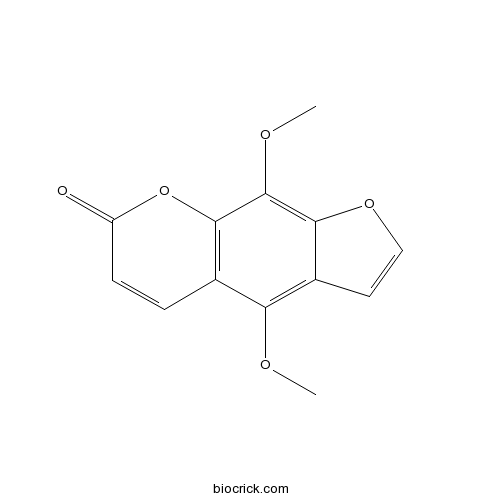
BCN5568
Isopimpinellin482-27-9
Instructions
-
BCN5574
Imperatorin482-44-0
Instructions
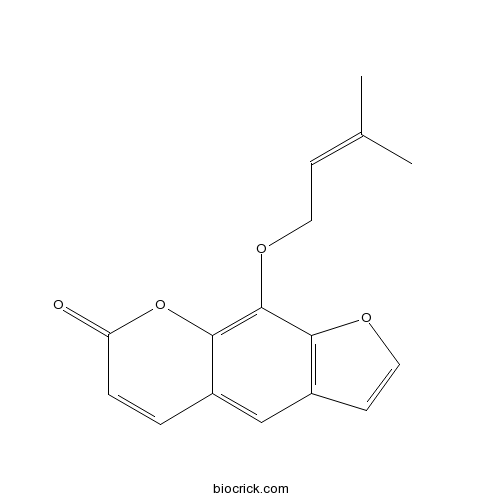
-
BCN2377
Isobergapten482-48-4
Instructions
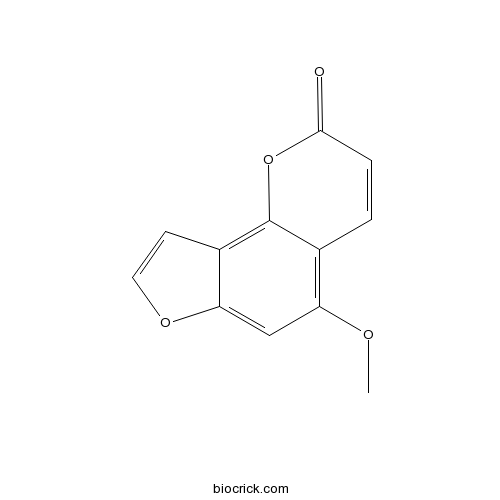
-
BCN5604
Nodakenetin495-32-9
Instructions
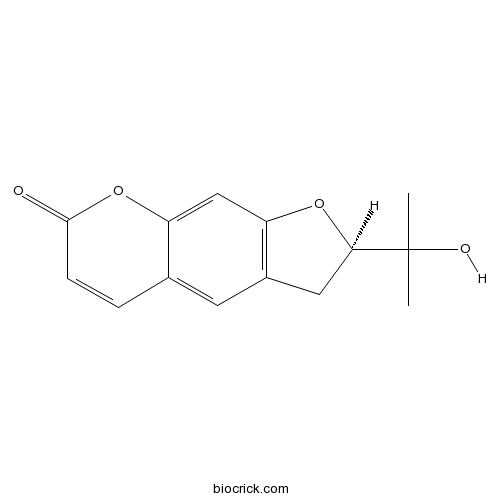
-
BCN1275
Columbianadin5058-13-9
Instructions
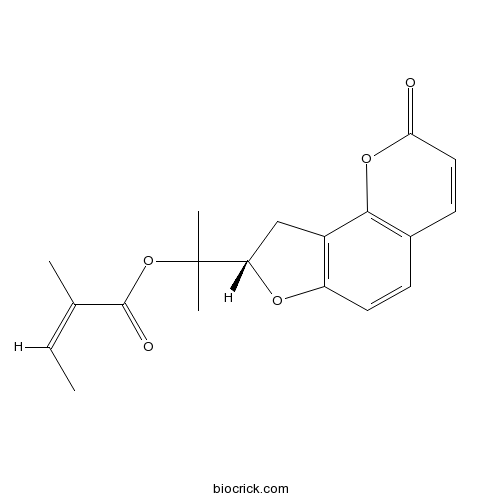
-
BCN4219
Psoralen66-97-7
Instructions
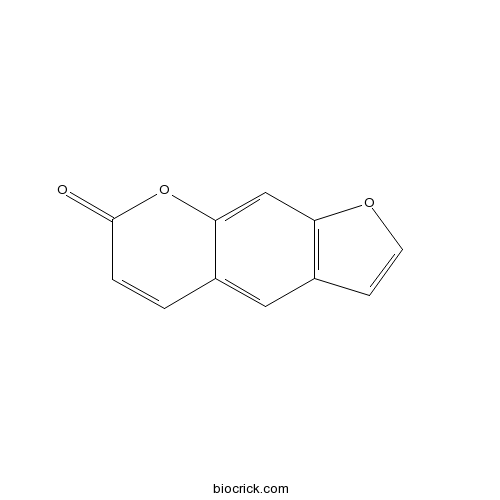
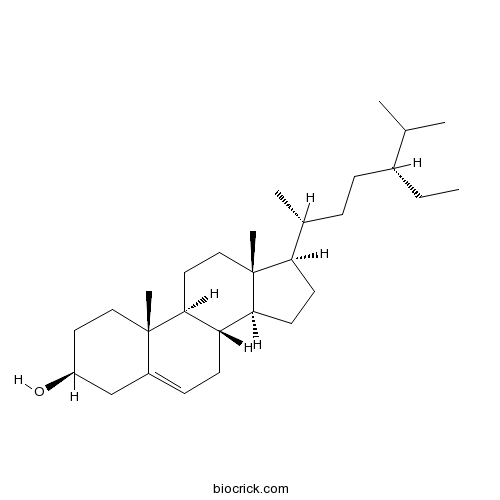
-
BCN1015
Beta-Sitosterol83-46-5
Instructions
Kinetics and molecular docking of dihydroxanthyletin-type coumarins from Angelica decursiva that inhibit cholinesterase and BACE1.[Pubmed: 30047040]
None
Dihydroxanthyletin-type coumarins from Angelica decursiva that inhibits the formation of advanced glycation end products and human recombinant aldose reductase.[Pubmed: 29230690]
None
[Chemical constituents from root of Angelica decursiva].[Pubmed: 29139270]
The compounds were isolated and purified by HP20 macroporous adsorption resin, ODS, silica gel, and Sephadex LH-20 column chromatography, as well as semi-preparative HPLC chromatography from the 80% ethanol extract of the root of Angelica decursiva, and their structures were identified based on their physiochemical properties and spectroscopic data. Twelve compounds were structures were identified as (9R,10R)-9-acetoxy-8,8-dimethyl-9,10-dihydro-2H,8H-benzo[1,2-b:3,4-b']dipyran-2-one-10-yl ester (1), bakuchicin (2), (3', S,4'S)-disenecioyloxy-3',4'-dihydroseselin (3), (3'R,4'R)-3'-angeloyloxy-4'-senecioyloxy-3',4'-dihydroseselincalipteryxin (4), (+)-8,9-dihydro-8-(2-hydroxypropan-2-yl)-2-oxo-2H-furo[2,3h]chromen-9-yl-3-methylbut-2-enoate (5), libanoridin (6), selinidin (7), suberosin (8), crocatone (9), peujaponisinol B (10), peujaponisinol A (11), and ostenol (12), respectively. Compounds 1-5 were isolated from the plants of Angelica genus for the first time. Compounds 7-12 were isolated from A. decursiva for the first time.
Vasorelaxant effects of Angelica decursiva root on isolated rat aortic rings.[Pubmed: 28969672]
Hypertension is one of the most important risk factors for cardiovascular disease (CVD) and a worldwide problem. Despite increases in the development of synthetic drugs for hypertension treatment, the rate of untreated and uncontrolled hypertension remains high. These drugs are effective, but can also cause side effects. Approximately 80% of the world population uses herbal medicines because of their low toxicity and better acceptability by the human body. Therefore, we attempted to identify natural medications for treating hypertension. The 70% ethanol extract of Angelica decursiva root (ADE) shows strong vasorelaxant potential, but no studies have investigated the mechanisms underlying the vasorelaxation effect of A. decursiva.
Kinetics and Molecular Docking Studies of 6-Formyl Umbelliferone Isolated from Angelica decursiva as an Inhibitor of Cholinesterase and BACE1.[Pubmed: 28946641]
None
A Nucleotide Signature for the Identification of Angelicae Sinensis Radix (Danggui) and Its Products.[Pubmed: 27713564]
It is very difficult to identify Angelicae sinensis radix (Danggui) when it is processed into Chinese patent medicines. The proposed internal transcribed spacer 2 (ITS2) is not sufficient to resolve heavily processed materials. Therefore, a short barcode for the identification of processed materials is urgently needed. In this study, 265 samples of Angelicae sinensis radix and adulterants were collected. The ITS2 region was sequenced, and based on one single nucleotide polymorphism(SNP) site unique to Angelica sinensis, a nucleotide signature consisting of 37-bp (5'-aatccgcgtc atcttagtga gctcaaggac ccttagg-3') was developed. It is highly conserved and specific within Angelica sinensis while divergent among other species. Then, we designed primers (DG01F/DG01R) to amplify the nucleotide signature region from processed materials. 15 samples procured online were analysed. By seeking the signature, we found that 7 of them were counterfeits. 28 batches of Chinese patent medicines containing Danggui were amplified. 19 of them were found to contain the signature, and adulterants such as Ligusticum sinense, Notopterygium incisum, Angelica decursiva and Angelica gigas were detected in other batches. Thus, this nucleotide signature, with only 37-bp, will broaden the application of DNA barcoding to identify the components in decoctions, Chinese patent medicines and other products with degraded DNA.
Columbianadin Inhibits Cell Proliferation by Inducing Apoptosis and Necroptosis in HCT116 Colon Cancer Cells.[Pubmed: 27098859]
Columbianadin (CBN), a natural coumarin from Angelica decursiva (Umbelliferae), is known to have various biological activities including anti-inflammatory and anti-cancer effects. In this study, the anti-proliferative mechanism of actions mediated by CBN was investigated in HCT-116 human colon cancer cells. CBN effectively suppressed the growth of colon cancer cells. Low concentration (up to 25 µM) of CBN induced apoptosis, and high concentration (50 µM) of CBN induced necroptosis. The induction of apoptosis by CBN was correlated with the modulation of caspase-9, caspase-3, Bax, Bcl-2, Bim and Bid, and the induction of necroptosis was related with RIP-3, and caspase-8. In addition, CBN induced the accumulation of ROS and imbalance in the intracellular antioxidant enzymes such as SOD-1, SOD-2, catalase and GPx-1. These findings demonstrate that CBN has the potential to be a candidate in the development of anti-cancer agent derived from natural products.
Coumarins from Angelica decursiva inhibit α-glucosidase activity and protein tyrosine phosphatase 1B.[Pubmed: 27085377]
In the present study, we investigated the anti-diabetic potential of six natural coumarins, 4-hydroxy Pd-C-III (1), 4'-methoxy Pd-C-I (2), decursinol (3), decursidin (4), umbelliferone 6-carboxylic acid (5), and 2'-isopropyl psoralene (6) isolated from Angelica decursiva and evaluated their inhibitory activities against protein tyrosine phosphatase 1B (PTP1B), α-glucosidase, and ONOO(-)-mediated protein tyrosine nitration. Coumarins 1-6 showed potent PTP1B and α-glucosidase inhibitory activities with ranges of IC50 values of 5.39-58.90 μM and 65.29-172.10 μM, respectively. In the kinetic study for PTP1B enzyme inhibition, compounds 1, 5, and 6 were competitive, whereas 2 and 4 showed mixed type, and 3 displayed noncompetitive type inhibition. For α-glucosidase enzyme inhibition, compounds 1 and 3 exhibited good mixed-type, while 2, 5, and 6 showed noncompetitive and 4 displayed competitive type inhibition. Furthermore, these coumarins also effectively suppressed ONOO(-)-mediated tyrosine nitration in a dose-dependent manner. To further investigate PTP1B inhibition, we generated a 3D structure of PTP1B using Autodock 4.2 and simulated the binding of compounds 1-6. Docking simulations showed that different residues of PTP1B interacted with different functional groups of compounds 1-6 through hydrogen and hydrophobic interactions. In addition, the binding energies of compounds 1-6 were negative, suggesting that hydrogen bonding may stabilize the open form of the enzyme and potentiate tight binding of the active site of PTP1B, thereby resulting in more effective PTP1B inhibition. These results demonstrate that the whole plant of A. decursiva and its coumarins are useful as potential functional food ingredients for the prevention and treatment of type 2 diabetes.


