ZaleplonBenzodiazepine site agonist CAS# 151319-34-5 |
- VX-765
Catalog No.:BCC3648
CAS No.:273404-37-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 151319-34-5 | SDF | Download SDF |
| PubChem ID | 5719 | Appearance | Powder |
| Formula | C17H15N5O | M.Wt | 305.33 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | CL 284846 | ||
| Solubility | Soluble to 100 mM in DMSO | ||
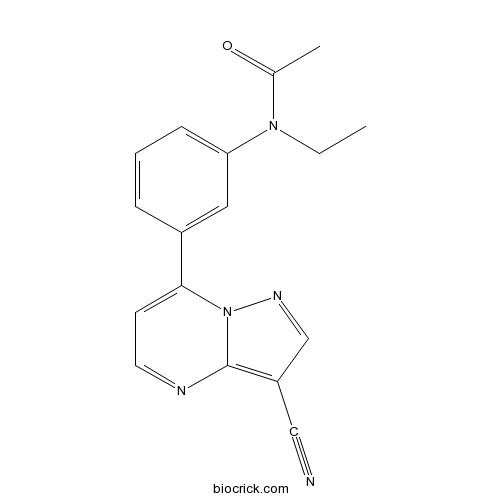
| Chemical Name | N-[3-(3-cyanopyrazolo[1,5-a]pyrimidin-7-yl)phenyl]-N-ethylacetamide | ||
| SMILES | CCN(C1=CC=CC(=C1)C2=CC=NC3=C(C=NN23)C#N)C(=O)C | ||
| Standard InChIKey | HUNXMJYCHXQEGX-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C17H15N5O/c1-3-21(12(2)23)15-6-4-5-13(9-15)16-7-8-19-17-14(10-18)11-20-22(16)17/h4-9,11H,3H2,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Non-benzodiazepine agent that acts as an agonist at the benzodiazepine site. Displays hypnotic, anxiolytic, myorelaxant and anticonvulsant activity. |

Zaleplon Dilution Calculator

Zaleplon Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.2751 mL | 16.3757 mL | 32.7514 mL | 65.5029 mL | 81.8786 mL |
| 5 mM | 0.655 mL | 3.2751 mL | 6.5503 mL | 13.1006 mL | 16.3757 mL |
| 10 mM | 0.3275 mL | 1.6376 mL | 3.2751 mL | 6.5503 mL | 8.1879 mL |
| 50 mM | 0.0655 mL | 0.3275 mL | 0.655 mL | 1.3101 mL | 1.6376 mL |
| 100 mM | 0.0328 mL | 0.1638 mL | 0.3275 mL | 0.655 mL | 0.8188 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Zaleplon(CL-284846) is a sedative/hypnotic, mainly used for insomnia.
- Inokosterone
Catalog No.:BCN3431
CAS No.:15130-85-5
- Genkwanol C
Catalog No.:BCN8012
CAS No.:151283-11-3
- CP 135807
Catalog No.:BCC7774
CAS No.:151272-90-1
- Primin
Catalog No.:BCN2729
CAS No.:15121-94-5
- Poricoic acid AM
Catalog No.:BCN8499
CAS No.:151200-92-9
- Borapetoside F
Catalog No.:BCN6413
CAS No.:151200-50-9
- Borapetoside E
Catalog No.:BCN6571
CAS No.:151200-49-6
- Borapetoside D
Catalog No.:BCN6612
CAS No.:151200-48-5
- 4'-Hydroxy-2,4-dimethoxychalcone
Catalog No.:BCC8708
CAS No.:151135-64-7
- CL 316243 disodium salt
Catalog No.:BCC7091
CAS No.:151126-84-0
- 8-(6-Hydroperoxy-3,7-dimethyl-2,7-octadienyloxy)psoralen
Catalog No.:BCN1558
CAS No.:151121-39-0
- 4-Difluoromethoxy-3-hydroxybenzaldehyde
Catalog No.:BCC8706
CAS No.:151103-08-1
- 3-O-p-Coumaroyloleanolic acid
Catalog No.:BCN3952
CAS No.:151334-06-4
- Ro 32-0432 hydrochloride
Catalog No.:BCC7122
CAS No.:151342-35-7
- Pseudolarolide B
Catalog No.:BCN8093
CAS No.:151368-43-3
- Estradiol hexahydrobenzoate
Catalog No.:BCC8962
CAS No.:15140-27-9
- Swietemahalactone
Catalog No.:BCN6886
CAS No.:1514669-21-6
- Ampelopsin F
Catalog No.:BCN3305
CAS No.:151487-08-0
- N-[[1-[(2-Nitrophenyl)sulfonyl]-1H-indole-3-yl]methyl]-N-[1-[1-[(2-nitrophenyl)sulfonyl]-1H-indole-3-yl]-2-oxo-2-(tert-butylamino)ethyl]-1-(2-diazo-3-oxobutyryl)-2-oxo-3-methylpiperidine-3beta-carboxamide
Catalog No.:BCC8335
CAS No.:151513-70-1
- Levomefolate calcium
Catalog No.:BCC1702
CAS No.:151533-22-1
- RU 58668
Catalog No.:BCC7608
CAS No.:151555-47-4
- ent-3-Oxokauran-17-oic acid
Catalog No.:BCN1674
CAS No.:151561-88-5
- JJKK 048
Catalog No.:BCC5610
CAS No.:1515855-97-6
- XEN445
Catalog No.:BCC5382
CAS No.:1515856-92-4
[Evaluation of the conservability of zolpidem and zaleplon in the hepatic tissue].[Pubmed:26710513]
Sud Med Ekspert. 2015 Sep-Oct;58(5):36-38.
This paper reports the experimental data on the conservability of zolpidem and zalepion in the samples of the cadaveric liver tissue stored under different conditions. The authors describe the method that includes isolation of the substances of interest from the hepatic tissue by means of acetone extraction, their solvent and chromatographic purification, and subsequent identification with the help of the spectrophotometric technique. It is shown that zolpidem and Zaleplon remain in the cadaveric hepatic tissue within 6 months after intoxication regardless of temperature which makes possible their detection, isolation, and identification by the proposed method.
Variability of Zaleplon 5-Oxidase Activity in Mice and Humans, and Inhibition by Raloxifene.[Pubmed:28029084]
Drug Metab Lett. 2017;10(4):278-285.
BACKGROUND: Zaleplon (ZAL) is a sedative-hypnotic agent, which is mainly metabolized to inactive 5-oxidized Zaleplon (5-oxo-ZAL) and N-des-ethylated ZAL (des-ethyl-ZAL) in mice and humans. The former reaction is considered to be catalyzed by aldehyde oxidase present in liver cytosol. METHODS: Here, we examined sex and strain differences of ZAL metabolism to 5-oxo-ZAL among four strains of mice, as well as the inter-individual variation in humans, in order to evaluate the variability of 5-oxo-ZAL-forming activity and its relationship with aldehyde oxidase activity. In mice, the activity in C57BL/6J strain was the highest, followed by C3H/He and BALB/c. The activity in DBA/2J was the lowest, being 2.3-fold lower than that of C57BL/6J mice. The activity of male mice was higher than that of female mice. Large inter-individual variations were observed among humans, with a range of 10- fold. Raloxifene, an inhibitor of aldehyde oxidase, markedly decreased the formation of 5-oxo-ZAL by liver cytosol of mice and humans. Further, the plasma level of 5-oxo-ZAL in mice was decreased when raloxifene was co-administered with ZAL. RESULTS: Our results indicate that the formation of 5-oxo-ZAL from ZAL is mainly catalyzed by aldehyde oxidase in mice and humans, and the variability of 5-oxo-ZAL formation is due primarily to differences of aldehyde oxidase activity. CONCLUSION: High inter-individual variability of ZAL 5-oxidase activity and potential for interaction of ZAL with other medicines that are inhibitors of aldehyde oxidase should be taken into consideration in clinical usage of ZAL.
Investigation of the usefulness of zaleplon at two doses to induce afternoon-sleep under noise interference and its effects on psychomotor performance and vestibular function.[Pubmed:26937286]
Mil Med Res. 2016 Mar 1;3:5.
BACKGROUND: Military operation personnel often suffer from sleep difficulty because of their work requirements. In this study, we investigated the efficacy of Zaleplon at two doses to induce afternoon-sleep under noise interference and its effects on psychomotor performance and vestibular function; we subsequently established the optimal dosage regimen for military operation personnel. METHODS: Twenty-two healthy young male volunteers were recruited for the study. Eight subjects took 10 mg or 15 mg of Zaleplon and placebo alternately and then were exposed to noise. Changes in polysomnography (PSG) indices, including sleep latency (SL), sleep efficiency (SE) and sleep structure, were recorded after drug administration. After awakening, the volunteers' subjective judgments of sleep quality and sleepiness were measured. Eight volunteers underwent 3 psychomotor performance tests at a one-week interval, and the psychomotor performance tests were conducted before and after taking Zaleplon and placebo. Six volunteers participated in the vestibular function test session, and parameters, including optokinetic nystagmus (OKN), vestibular ocular reflex (VOR), visual-vestibular ocular reflex (VVOR) and vestibular ocular reflex fixation suppression (VOR-Fix), were detected by the same experimental design as described above. The data of sleep observations were subjected to one-way variance analysis. RESULTS: Compared with the placebo group, SL was shortened significantly, and the scores of subjective sleep quality and sleep depth were clearly increased in the Zaleplon 10 mg group (P < 0.05). Moreover, the SE and the percent of REM (rapid eye movement) sleep were increased remarkably in the Zaleplon 15 mg group (P < 0.01). Furthermore, the SE, percent of REM sleep and scores of subjective sleep depth in the Zaleplon 15 mg group were significantly higher than in the Zaleplon 10 mg group (P < 0.05). The psychomotor performance did not change significantly after ingestion of 10 mg or 15 mg of Zaleplon, whereas the OKN and VOR gains were lower in the two dose groups of Zaleplon (P < 0.05) and restored to normal 3 h after drug ingestion. CONCLUSION: Zaleplon is an ideal hypnotic for military personnel, and its hypnotic efficiency is dose-related under noise interference; a 15 mg dose of Zaleplon could provide significantly better sleep than a 10 mg dose of Zaleplon.
Lipid nanoparticles of zaleplon for improved oral delivery by Box-Behnken design: optimization, in vitro and in vivo evaluation.[Pubmed:28274147]
Drug Dev Ind Pharm. 2017 Jul;43(7):1205-1214.
PURPOSE: Zaleplon (ZL) is a hypnotic drug prescribed for the management of insomnia and convulsions. The oral bioavailability of ZL was low ( approximately 30%) owing to poor water solubility and hepatic first-pass metabolism. The cornerstone of this investigation is to develop and optimize solid lipid nanoparticles (SLNs) of ZL with the aid of Box-Behnken design (BBD) to improve the oral bioavailability. METHODS: A design space with three formulation variables at three levels were evaluated in BBD. Amount of lipid (A1), amount of surfactant (A2) and concentration of co-surfactant (%) (A3) were selected as independent variables, whereas, particle size (B1), entrapment efficiency (B2) and zeta potential (ZP, B3) as responses. ZL-SLNs were prepared by hot homogenization with ultrasonication method and evaluated for responses to obtain optimized formulation. Morphology of nanoparticles was observed under SEM. DSC and XRD studies were examined to understand the native crystalline behavior of drug in SLN formulations. Further, in vivo studies were performed in Wistar rats. RESULTS: The optimized formulation with 132.89 mg of lipid, 106.7 mg of surfactant and 0.2% w/v of co-surfactant ensued in the nanoparticles with 219.9 +/- 3.7 nm of size, -25.66 +/- 2.83 mV surface charge and 86.83 +/- 2.65% of entrapment efficiency. SEM studies confirmed the spherical shape of SLN formulations. The DSC and XRD studies revealed the transformation of crystalline drug to amorphous form in SLN formulation. In conclusion, in vivo studies in male Wistar rats demonstrated an improvement in the oral bioavailability of ZL from SLN over control ZL suspension. CONCLUSIONS: The enhancement in the oral bioavailability of ZL from SLNs, developed with the aid of BBD, explicated the potential of lipid-based nanoparticles as a potential carrier in improving the oral delivery of this poorly soluble drug.
Comparison of the pharmacological profiles of the hypnotic drugs, zaleplon and zolpidem.[Pubmed:8905326]
Eur J Pharmacol. 1996 Oct 10;313(1-2):35-42.
The BZ1 (omega 1)-selective compound, zolpidem, is a clinically effective hypnotic drug with a pharmacological profile which differs from those of benzodiazepine anxiolytics and hypnotics. Zaleplon (CL 284,846) has recently been described as a hypnotic agent which also has BZ1 (omega 1) receptor selectivity. The pharmacological effects of zolpidem and Zaleplon were therefore compared in mice and rats. Both drugs blocked tonic convulsions induced in mice by pentylenetetrazole and electroconvulsive shock and clonic convulsions induced by isoniazid. Zaleplon was more potent than zolpidem but the maximal effect of zolpidem for increasing the latency to isoniazid-induced convulsions was greater than that of Zaleplon. Little tolerance developed to the anticonvulsant effect of Zaleplon against isoniazid-induced seizures following twice daily administration of 10 or 30 mg/kg for 10 days. Both compounds reduced locomotor activity and produced motor deficits in the rotarod and loaded grid tests in mice. However, while Zaleplon produced all three effects at similar doses, zolpidem showed the greatest potency for reducing locomotion. Zaleplon and zolpidem also decreased locomotion and produced a rotarod deficit in rats. Again, the difference between the doses giving rise to these two effects was greater for zolpidem than for Zaleplon. In a drug discrimination procedure using rats trained to discriminate a dose (5 mg/kg) of chlordiazepoxide, Zaleplon produced partial substitution for chlordiazepoxide at doses which greatly reduced response rates. These results show that Zaleplon and zolpidem have similar pharmacological profiles, presumably related to their BZ1 (omega 1) receptor selectivity. However, the difference between doses producing motor deficits (rotarod, loaded grid) and those giving rise to other effects (anticonvulsant, decreased locomotion) was greater for zolpidem than for Zaleplon. This difference may be related to a greater in vivo intrinsic activity of zolpidem as indicated by the different efficacies of the two drugs to antagonise isoniazid-induced convulsions.
Pharmacokinetics and anticonvulsant effect of a new hypnotic, CL 284,846, in rats.[Pubmed:8592655]
Pharm Res. 1995 Nov;12(11):1592-7.
PURPOSE: CL 284,846 (CL846) is an investigational non-benzodiazepine agent with hypnotic, anxiolytic, myorelaxant and anticonvulsant properties. This study assessed the pharmacokinetics and anticonvulsant action of CL846 in female Sprague-Dawley rats. METHODS: CL846 pharmacokinetics were examined after either an iv bolus dose (2.5 mg/kg) or a 6-hr infusion (0.4 mg/kg/hr). CL846 pharmacodynamics were evaluated with a pentylenetetrazol (PTZ) infusion 5 min after a CL846 in bolus dose (0 to 10 mg/kg). CL846 and the derived metabolite CL 284,859 (CL859) concentrations in serum and brain tissue were determined by HPLC with fluorescence detection. RESULTS: Both the steady-state volume of distribution (1636 +/- 162 and 1804 +/- 293 ml/kg, after bolus and infusion administration, respectively) and systemic clearance (19.1 +/- 7.1 and 22.2 +/- 4.3 ml/min/kg for bolus and infusion administration, respectively) were high. No differences in pharmacokinetic parameters were noted between the two modes of administration. The relationship between anticonvulsant effect and brain/serum concentrations was well described by an Emax model. CL846 was as effective as triazolam in antagonizing PTZ-induced seizures. CONCLUSIONS: Under the conditions of the present study, CL846 pharmacokinetics were linear and stationary. Further evaluation of the anticonvulsant properties of CL846 is warranted, including the potential development of tolerance, which is well known for benzodiazepines.


