Sodium taurocholateCAS# 145-42-6 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 145-42-6 | SDF | Download SDF |
| PubChem ID | 137705085 | Appearance | Powder |
| Formula | C26H44NNaO7S | M.Wt | 537.7 |
| Type of Compound | Other Steroids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
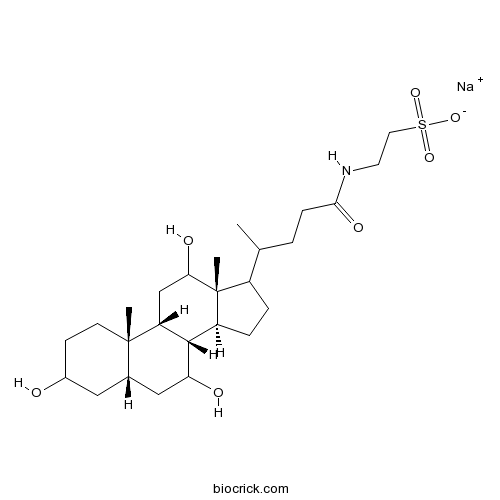
| Chemical Name | sodium;2-[4-[(5S,8R,9R,10S,13R,14S)-3,7,12-trihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl]pentanoylamino]ethanesulfonate | ||
| SMILES | CC(CCC(=O)NCCS(=O)(=O)[O-])C1CCC2C1(C(CC3C2C(CC4C3(CCC(C4)O)C)O)O)C.[Na+] | ||
| Standard InChIKey | JAJWGJBVLPIOOH-KYSLWWRTSA-M | ||
| Standard InChI | InChI=1S/C26H45NO7S.Na/c1-15(4-7-23(31)27-10-11-35(32,33)34)18-5-6-19-24-20(14-22(30)26(18,19)3)25(2)9-8-17(28)12-16(25)13-21(24)29;/h15-22,24,28-30H,4-14H2,1-3H3,(H,27,31)(H,32,33,34);/q;+1/p-1/t15?,16-,17?,18?,19-,20+,21?,22?,24-,25-,26+;/m0./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Sodium taurocholate Dilution Calculator

Sodium taurocholate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.8598 mL | 9.2989 mL | 18.5977 mL | 37.1955 mL | 46.4943 mL |
| 5 mM | 0.372 mL | 1.8598 mL | 3.7195 mL | 7.4391 mL | 9.2989 mL |
| 10 mM | 0.186 mL | 0.9299 mL | 1.8598 mL | 3.7195 mL | 4.6494 mL |
| 50 mM | 0.0372 mL | 0.186 mL | 0.372 mL | 0.7439 mL | 0.9299 mL |
| 100 mM | 0.0186 mL | 0.093 mL | 0.186 mL | 0.372 mL | 0.4649 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- (S)-(+)-Carvone
Catalog No.:BCN9077
CAS No.:2244-16-8
- (1S)-(-)-α-Pinene
Catalog No.:BCN9076
CAS No.:7785-26-4
- (-)-Fenchone
Catalog No.:BCN9075
CAS No.:7787-20-4
- 3-(β-D-Glucopyranosyloxy)-1,6-dihydroxy-2-methyl-9,10-anthracenedione
Catalog No.:BCN9074
CAS No.:125906-49-2
- Ferulic Acid Methyl Ester
Catalog No.:BCN9073
CAS No.:2309-07-1
- Alphalipoic acid
Catalog No.:BCN9072
CAS No.:1077-28-7
- (+)-Fenchone
Catalog No.:BCN9071
CAS No.:4695-62-9
- (-)-Menthone
Catalog No.:BCN9070
CAS No.:14073-97-3
- Benzyl alcohol
Catalog No.:BCN9069
CAS No.:100-51-6
- Citronellal
Catalog No.:BCN9068
CAS No.:106-23-0
- 10-hydroxydec-2-enoic acid
Catalog No.:BCN9067
CAS No.:14113-05-4
- Citral
Catalog No.:BCN9066
CAS No.:5392-40-5
- Sodium deoxycholate
Catalog No.:BCN9079
CAS No.:302-95-4
- Lanosta-8,20(22)-dien-26-oic acid, 15-hydroxy-3,11,23-trioxo-, (15α,20Z)-
Catalog No.:BCN9080
CAS No.:1961358-01-9
- Chol-8-en-24-oic acid, 7,15-dihydroxy-4,4,14-trimethyl-3,11-dioxo-, (5α)-
Catalog No.:BCN9081
CAS No.:942936-54-1
- Calenduloside E
Catalog No.:BCN9082
CAS No.:26020-14-4
- (3β,7β,12β,20Z )- 3,7,12- trihydroxy-11,15,23-trioxo-lanost-8,20-dien-26-oic acid
Catalog No.:BCN9083
CAS No.:1961358-02-0
- (-)-Sesamin
Catalog No.:BCN9084
CAS No.:13079-95-3
- (3r)-7,2'-Dihydroxy-3',4'-dimethoxyisoflavan
Catalog No.:BCN9085
CAS No.:64474-51-7
- Cucurbitacin Q1
Catalog No.:BCN9086
CAS No.:99530-82-2
- Cyclohexanecarboxylic acid, 3-[[(2E)-3-[4-(D-glucopyranosyloxy)-3-hydroxyphenyl]-1-oxo-2-propen-1-yl...
Catalog No.:BCN9087
CAS No.:1629852-63-6
- (-)-Isobicyclogermacrenal
Catalog No.:BCN9088
CAS No.:73256-82-3
- Delphinidin 3-O-galactoside
Catalog No.:BCN9089
CAS No.:197250-28-5
- Bisisorhapontigenin E
Catalog No.:BCN9090
CAS No.:
Ratite oils for local transdermal therapy of 4-OH Tamoxifen: Development, Characterization and Ex-vivo Evaluation.[Pubmed:32648792]
J Liposome Res. 2020 Jul 10:1-38.
The anti-inflammatory property of ratite oils as well as its ability to act as a penetration enhancer makes it an ideal agent to be used in transdermal formulations. The present study aims to develop an effective transfersomal delivery of 4-hydroxytamoxifen, an anti-cancer drug, using ratite oil as a carrier agent for the treatment of breast cancer. The 4-hydroxytamoxifen transfersomes were prepared with and without ratite oils using soy phosphatidylcholine and three different edge activators in five different molar ratios using the rotary evaporation-ultrasonication method. Optimal transfersome formulations were selected using physical-chemical characterization and ex vivo studies. Results from physical-chemical characterisation of the developed formulations found Sodium taurocholate to be the most suitable edge activator, whichrecorded highest entrapment efficiency of 95.1 +/- 2.70% with 85:15, (w/w) and lowest vesicle size of 82.3 +/- 0.02 nmwith 75:25, (w/w) molar ratios. TEM and DSC studies showed that the vesicles were readily identified and present in a nearly perfect spherical shape. In addition, formulations with emu oil had better stability than formulations with ostrich oil. Physical stability studies at 4 degrees C showed that ratite oil transfersomes were stable up to 4 weeks, while transfersomes without ratite oils were stable for 8 weeks. Ex vivo permeability studies using porcine skin concluded that 4-hydroxytamoxifen transfersomal formulations with (85:15, w/w) without emu oil have the potential to be used in transdermal delivery approach to enhance permeation of 4-hydroxytamoxifen, which may be beneficial in the treatment of breast cancer.
Molecular docking and statistical optimization of taurocholate-stabilized galactose anchored bilosomes for the enhancement of sofosbuvir absorption and hepatic relative targeting efficiency.[Pubmed:32611266]
Drug Deliv. 2020 Dec;27(1):996-1009.
The work aimed to improve both absorption and hepatic availability of sofosbuvir. Bilosomes and galactose-anchored bilosomes were investigated as potential nanocarriers for this purpose. Sofosbuvir is a class III drug with high solubility and low permeability. Thus, the drug entrapment into lipid-based galactose-anchored carriers would enhance drug permeability and improve its liver availability. The galactosylated taurocholate was designed and synthesized based on molecular docking studies, where both galactose and taurocholate molecules were connected in a way to avoid affecting crucial interactions and avoid steric clashes with their cellular uptake receptors. The suggested nano-carriers were prepared using a thin-film hydration technique with Sodium taurocholate and span 60 as stabilizers. The prepared formulae were statistically optimized using a central composite design. The optimized plain and galactosylated formulae, composed of SAA to drug ratio of 1:1 w/w and Sodium taurocholate to span ratio of 10:1 w/w, have a vesicular size, zeta potential and entrapment efficiency in the range of 140-150 nm, -50 mV and 85%, respectively. The optimized formulae were lyophilized to increase their physical stability and facilitate accurate drug dosing. In vivo results showed that Sofosbuvir availability in the liver was significantly increased after oral administration of the plain and the galactosylated bilosomal formulae when compared to the oral drug solution with relative targeting efficiencies (RTIs) of 1.51 and 3.66, respectively. These findings confirmed the hypothesis of considering the galactosylated bilosomes a promising nanocarrier to efficiently target sofosbuvir to the liver.
Naturally occurring 5' preS1 deletions markedly enhance replication and infectivity of HBV genotype B and genotype C.[Pubmed:32571971]
Gut. 2020 Jun 22. pii: gutjnl-2019-320096.
BACKGROUND AND AIMS: Deletion of 15-nucleotide or 18-nucleotide (nt) covering preS1 ATG frequently arises during chronic infection with HBV genotypes B and C. Since the second ATG is 33nt downstream, they truncate large (L) envelope protein by 11 residues like wild-type genotype D. This study characterised their functional consequences. METHODS: HBV genomes with or without deletion were amplified from a patient with advanced liver fibrosis and assembled into replication competent 1.1mer construct. Deletion, insertion or point mutation was introduced to additional clones of different genotypes. Viral particles concentrated from transfected HepG2 cells were inoculated to Sodium taurocholate cotransporting polypeptide (NTCP)-reconstituted HepG2 (HepG2/NTCP) cells or differentiated HepaRG cells, and HBV RNA, DNA, proteins were monitored. RESULTS: From transfected HepG2 cells, the 15-nt and 18-nt deletions increased HBV RNA, replicative DNA and extracellular virions. When same number of viral particles was inoculated to HepG2/NTCP cells, the deletion mutants showed higher infectivity. Conversely, HBV infectivity was diminished by putting back the 18nt into naturally occurring genotype C deletion mutants and by adding 33nt to genotype D. Infectivity of full-length genotype C clones was also enhanced by mutating the first ATG codon of the preS1 region but diminished by mutating the second in-frame ATG. Removing N-terminal 11 residues from preS1 peptide 2-59 of genotype C potentiated inhibition of HBV infection and enhanced binding to HepG2/NTCP cells. CONCLUSIONS: The 15-nt and 18-nt deletions somehow increase HBV RNA, replicative DNA and virion production. Shortened L protein is more efficient at mediating HBV infection.
Hepatitis B Virus Entry into Cells.[Pubmed:32570893]
Cells. 2020 Jun 18;9(6). pii: cells9061486.
Hepatitis B virus (HBV), an enveloped partially double-stranded DNA virus, is a widespread human pathogen responsible for more than 250 million chronic infections worldwide. Current therapeutic strategies cannot eradicate HBV due to the persistence of the viral genome in a special DNA structure (covalently closed circular DNA, cccDNA). The identification of Sodium taurocholate co-transporting polypeptide (NTCP) as an entry receptor for both HBV and its satellite virus hepatitis delta virus (HDV) has led to great advances in our understanding of the life cycle of HBV, including the early steps of infection in particular. However, the mechanisms of HBV internalization and the host factors involved in this uptake remain unclear. Improvements in our understanding of HBV entry would facilitate the design of new therapeutic approaches targeting this stage and preventing the de novo infection of naive hepatocytes. In this review, we provide an overview of current knowledge about the process of HBV internalization into cells.
Modeling Hepatitis B Virus Infection in Non-Hepatic 293T-NE-3NRs Cells.[Pubmed:32568248]
J Vis Exp. 2020 Jun 5;(160).
HBV mainly infects human hepatocytes, but it has also been found to infect extrahepatic tissues such as kidney and testis. Nonetheless, cell-based HBV models are limited to hepatoma cell lines (such as HepG2 and Huh7) overexpressing a functional HBV receptor, Sodium taurocholate co-transporting polypeptide (NTCP). Here, we used 293T-NE-3NRs (293T overexpressing human NTCP, HNF4alpha, RXRalpha and PPARalpha) and HepG2-NE (HepG2 overexpressing NTCP) as model cell lines. HBV infection in these cell lines was performed either by using concentrated HBV virus particles from HepG2.2.15 or co-culturing HepG2.2.15 with the target cell lines. HBcAg immunofluorescence for HBcAg was performed to confirm HBV infection. The two methods presented here will help us study HBV infection in non-hepatic cell lines.
Ghrelin inhibits IKKbeta/NF-kappaB activation and reduces pro-inflammatory cytokine production in pancreatic acinar AR42J cells treated with cerulein.[Pubmed:32553660]
Hepatobiliary Pancreat Dis Int. 2020 Jun 2. pii: S1499-3872(20)30108-9.
BACKGROUND: Previous studies have provided conflicting results regarding whether the serum ghrelin concentration can reflect the severity of acute pancreatitis (AP). The present study examined the correlation between the serum ghrelin concentration and AP severity in animal models and investigated whether altered ghrelin expression in pancreatic acinar cells influences IKKbeta/NF-kappaB signaling and pro-inflammatory cytokine production. METHODS: Mild or severe AP was induced in rats by intraperitoneal injection of cerulein or retrograde cholangiopancreatic duct injection of Sodium taurocholate, respectively. After successful model induction, serum ghrelin, tumor necrosis factor-alpha (TNF-alpha), and interleukin-6 (IL-6) concentrations were determined by enzyme-linked immunosorbent assay, and IKKbeta/NF-kappaB activation was assessed by immunohistochemistry. Subsequently, stable overexpression or knockdown of ghrelin in AR42J cells was achieved by lentiviral transfection. After transfected cells and control cells were treated with cerulein for 24 h, the TNF-alpha and IL-1beta levels in the supernatants were determined by enzyme-linked immunosorbent assay, and the expression levels of p-p65, IKKbeta, and p-IKKbeta were detected by Western blotting. RESULTS: In rat AP models, AP severity was correlated with increased IKKbeta/NF-kappaB activation, pro-inflammatory cytokine production, and ghrelin secretion. The levels of pro-inflammatory cytokines TNF-alpha and IL-1beta as well as IKKbeta/NF-kappaB signaling activity were increased upon knockdown of ghrelin in the AP acinar cell model and decreased with ghrelin overexpression. CONCLUSIONS: Serum ghrelin is related to the severity of AP. Ghrelin may play a protective role in the pathogenesis of AP by inhibiting the pro-inflammatory cytokines and the activation of the IKKbeta/NF-kappaB signaling pathway.
Emodin Protects Against Acute Pancreatitis-Associated Lung Injury by Inhibiting NLPR3 Inflammasome Activation via Nrf2/HO-1 Signaling.[Pubmed:32546964]
Drug Des Devel Ther. 2020 May 21;14:1971-1982.
Aim: Lung injury is a common complication of acute pancreatitis (AP), which leads to the development of acute respiratory distress syndrome and causes high mortality. In the present study, we investigated the therapeutic effect of emodin on AP-induced lung injury and explored the molecular mechanisms involved. Materials and Methods: Thirty male Sprague-Dawley rats were randomly divided into AP (n=24) and normal (n=6) groups. Rats in the AP group received a retrograde injection of 5% Sodium taurocholate into the biliary-pancreatic duct and then randomly assigned to untreated, emodin, combined emodin and ML385, and dexamethasone (DEX) groups. Pancreatic and pulmonary injury was assessed using H&E staining. In in vitro study, rat alveolar epithelial cell line L2 cells were exposed to lipopolysaccharide and treated with emodin. Nrf2 siRNA pool was applied for the knockdown of Nrf2. The contents of the pro-inflammatory cytokines in the bronchoalveolar lavage fluid and lung were determined using enzyme-linked immunosorbent assay. The expressions of related mRNAs and proteins in the lung or L2 cells were detected using real-time polymerase chain reaction, Western blot, immunohistochemistry and immunofluorescence. Key Findings: Emodin administration alleviated pancreatic and pulmonary injury of rats with AP. Emodin administration suppressed the production of proinflammatory cytokines, downregulated NLRP3, ASC and caspase-1 expressions and inhibited NF-kappaB nuclear accumulation in the lung. In addition, Emodin increased Nrf2 nuclear translocation and upregulated HO-1 expression. Moreover, the anti-inflammatory effect of emodin was blocked by Nrf2 inhibitor ML385. Conclusion: Emodin effectively protects rats against AP-associated lung injury by inhibiting NLRP3 inflammasome activation via Nrf2/HO-1 signaling.
Interactions Between Neutrophils and Platelets in the Progression of Acute Pancreatitis.[Pubmed:32541639]
Pancreas. 2020 Jul;49(6):830-836.
OBJECTIVE: Severe acute pancreatitis is a serious disease, but its detailed mechanism has not yet been elucidated. We aimed to clarify the interaction between neutrophils and platelets in the pathogenesis of acute pancreatitis. METHODS: We induced acute pancreatitis in rats by injection of Sodium taurocholate into the biliopancreatic duct and killed them over time. We observed the histological changes in pancreatic tissue with special attention to the dynamics of neutrophils and platelets. We also measured the concentrations of neutrophil- and platelet-derived factors in pancreatic tissue and blood samples. RESULTS: Neutrophils and platelets in the pancreatic tissue showed a similar pattern of migration. They initially spread in the interlobular connective tissue and finally into the lobules. The concentration of myeloperoxidase gradually increased in the inflamed pancreas until 24 hours and the concentration of thromboxane B2, plasminogen activator inhibitor 1, and CD41 also increased with time. Finally, the concentration of serum myeloperoxidase, citrullinated histone H3, and high-mobility group box 1 increased over time. CONCLUSIONS: The interaction between neutrophils and platelets in pancreatic tissue plays an important role in the mechanism of advancing severity in acute pancreatitis. Circulating damage-associated molecular patterns induced by excessive local inflammation may lead to other organ injuries.


