SB 224289 hydrochlorideSelective 5-HT1B antagonist CAS# 180084-26-8 |
- Talnetant
Catalog No.:BCC1981
CAS No.:174636-32-9
- Talnetant hydrochloride
Catalog No.:BCC1982
CAS No.:204519-66-4
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 180084-26-8 | SDF | Download SDF |
| PubChem ID | 11226716 | Appearance | Powder |
| Formula | C32H33ClN4O3 | M.Wt | 557.09 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 10 mM in DMSO with gentle warming | ||
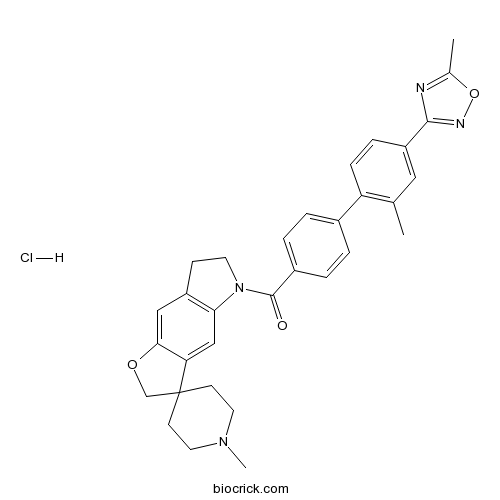
| Chemical Name | [4-[2-methyl-4-(5-methyl-1,2,4-oxadiazol-3-yl)phenyl]phenyl]-(1'-methylspiro[6,7-dihydro-2H-furo[2,3-f]indole-3,4'-piperidine]-5-yl)methanone;hydrochloride | ||
| SMILES | CC1=C(C=CC(=C1)C2=NOC(=N2)C)C3=CC=C(C=C3)C(=O)N4CCC5=C4C=C6C(=C5)OCC67CCN(CC7)C.Cl | ||
| Standard InChIKey | GKGKBZYMDILCOF-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C32H32N4O3.ClH/c1-20-16-25(30-33-21(2)39-34-30)8-9-26(20)22-4-6-23(7-5-22)31(37)36-13-10-24-17-29-27(18-28(24)36)32(19-38-29)11-14-35(3)15-12-32;/h4-9,16-18H,10-15,19H2,1-3H3;1H | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Selective 5-HT1B receptor antagonist (pKi = 8.2). Displays > 60-fold selectivity over 5-HT1D, 5-HT1A, 5-HT1E, 5-HT1F, 5-HT2A and 5-HT2C receptors in radioligand binding and functional assays. Centrally active following oral administration in vivo. |

SB 224289 hydrochloride Dilution Calculator

SB 224289 hydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.795 mL | 8.9752 mL | 17.9504 mL | 35.9008 mL | 44.8761 mL |
| 5 mM | 0.359 mL | 1.795 mL | 3.5901 mL | 7.1802 mL | 8.9752 mL |
| 10 mM | 0.1795 mL | 0.8975 mL | 1.795 mL | 3.5901 mL | 4.4876 mL |
| 50 mM | 0.0359 mL | 0.1795 mL | 0.359 mL | 0.718 mL | 0.8975 mL |
| 100 mM | 0.018 mL | 0.0898 mL | 0.1795 mL | 0.359 mL | 0.4488 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Voafinidine
Catalog No.:BCN6738
CAS No.:180059-77-2
- SDZ NKT 343
Catalog No.:BCC7349
CAS No.:180046-99-5
- Ketohakonanol
Catalog No.:BCN7427
CAS No.:18004-20-1
- 7-Chlorokynurenic acid
Catalog No.:BCC6577
CAS No.:18000-24-3
- 3-oxo-Olean-12-en-28-oic acid
Catalog No.:BCN3171
CAS No.:17990-42-0
- 27-Hydroxymangiferolic acid
Catalog No.:BCN4689
CAS No.:17983-82-3
- Aflastatin A
Catalog No.:BCN1822
CAS No.:179729-59-0
- N-Benzoyl-leucine
Catalog No.:BCC9092
CAS No.:17966-67-5
- Macrocarpal J
Catalog No.:BCN1139
CAS No.:179603-47-5
- Amabiline
Catalog No.:BCN1950
CAS No.:17958-43-9
- Cynaustine
Catalog No.:BCN1951
CAS No.:17958-39-3
- Cynaustraline
Catalog No.:BCN2048
CAS No.:17958-37-1
- Bupivacaine HCl
Catalog No.:BCC4406
CAS No.:18010-40-7
- Quercetin 3-O-glucoside-7-O-rhamnoside
Catalog No.:BCN1520
CAS No.:18016-58-5
- Megastigm-7-ene-3,4,6,9-tetrol
Catalog No.:BCN6511
CAS No.:180164-14-1
- Anthracophyllone
Catalog No.:BCN7606
CAS No.:1801750-22-0
- Ganoderlactone D
Catalog No.:BCN7849
CAS No.:1801934-15-5
- Phytolaccagenin
Catalog No.:BCN1140
CAS No.:1802-12-6
- NCT-501
Catalog No.:BCC6539
CAS No.:1802088-50-1
- Cyclo(Leu-Ala)
Catalog No.:BCN2428
CAS No.:1803-60-7
- Perillaldehyde
Catalog No.:BCN8294
CAS No.:18031-40-8
- Solifenacin hydrochloride
Catalog No.:BCC5193
CAS No.:180468-39-7
- Fmoc-D-Asn(Trt)-OH
Catalog No.:BCC3084
CAS No.:180570-71-2
- Gentioflavin
Catalog No.:BCN3619
CAS No.:18058-50-9
The selective 5-HT1B receptor inverse agonist 1'-methyl-5-[[2'-methyl-4'-(5-methyl-1,2, 4-oxadiazol-3-yl)biphenyl-4-yl]carbonyl]-2,3,6,7-tetrahydro- spiro[furo[2,3-f]indole-3,4'-piperidine] (SB-224289) potently blocks terminal 5-HT autoreceptor function both in vitro and in vivo.[Pubmed:9548813]
J Med Chem. 1998 Apr 9;41(8):1218-35.
5-HT1 receptors are members of the G-protein-coupled receptor superfamily and are negatively linked to adenylyl cyclase activity. The human 5-HT1B and 5-HT1D receptors (previously known as 5-HT1Dbeta and 5-HT1Dalpha, respectively), although encoded by two distinct genes, are structurally very similar. Pharmacologically, these two receptors have been differentiated using nonselective chemical tools such as ketanserin and ritanserin, but the absence of truly selective agents has meant that the precise function of the 5-HT1B and 5-HT1D receptors has not been defined. In this paper we describe how, using computational chemistry models as a guide, the nonselective 5-HT1B/5-HT1D receptor antagonist 4 was structurally modified to produce the selective 5-HT1B receptor inverse agonist 5, 1'-methyl-5-[[2'-methyl-4'-(5-methyl-1,2, 4-oxadiazol-3-yl)biphenyl-4-yl]carbonyl]-2,3,6, 7-tetrahydrospiro[furo[2,3-f]indole-3,4'-piperidine] (SB-224289). This compound is a potent antagonist of terminal 5-HT autoreceptor function both in vitro and in vivo.
Differential effects of 5-HT1B/1D receptor antagonists in dorsal and median raphe innervated brain regions.[Pubmed:9652357]
Eur J Pharmacol. 1998 Apr 10;346(2-3):175-80.
The effect of SB-224289 (2,3,6.7-tetrahydro-1'-methyl-5- inverted question mark2'-methyl-4'-[(5-methyl-1,2,4-oxadiazol e-3-yl)biphenyl-4-yl]carbonyl inverted question markFuro[2,3-F]-indole-3-spiro-4'-piperidine oxalate) (4 mg/kg i.p., 5-HT1B receptor antagonist), GR 127935 (N-[4-methoxy-3-(4-methyl-1-piperizinyl)phenyl]-2'-methyl-4'-(5-me thyl-1,2,4-oxadiazole-3-yl)[1,1'-biphenyl]-carboxamide) (0.3 mg/kg i.p., 5-HT1B/1D receptor antagonist), and paroxetine (10 mg/kg p.o.) were investigated on extracellular 5-hydroxytryptamine (5-HT) levels in the frontal cortex, striatum and dentate gyrus of the freely moving guinea-pig with microdialysis. In the frontal cortex and striatum (dorsal raphe innervated areas), GR 127935 evoked a significant decrease in extracellular 5-HT, reaching minima of 41+/-12% and 32+/-6% of basal, respectively. This decrease may be explained by antagonism of inhibitory 5-HT1B/1D receptors on raphe cell bodies, leading to a local increase in 5-HT, which, in turn, stimulated 5-HT1A receptors to decrease cell firing, and hence 5-HT release from terminals. In contrast, SB-224289 had no effect on 5-HT levels in either region. In the dentate gyrus (median raphe innervated area), GR 127935 and SB-224289 significantly increased extracellular 5-HT, reaching maxima of 146+/-11% and 151+/-19% of basal, respectively. The ability of both compounds to increase 5-HT levels in the dentate gyrus suggests a lack of 5-HT1B/1D receptors in the median raphe nucleus. Paroxetine produced a small but non-significant increase in extracellular 5-HT in the frontal cortex, and a small decrease in the dentate gyrus. The lack of effect of paroxetine in terminal areas may be due to the limiting effects of cell body 5-HT autoreceptors. In summary, the above data demonstrate that 5-HT1B/1D receptor antagonists increase 5-HT levels in the dentate gyrus, implying that acute administration of 5-HT1B/1D receptor antagonists will achieve a similar effect to chronic selective serotonin re-uptake inhibitor treatment in median raphe innervated areas. This, in turn, suggests that such compounds may be efficacious in the treatment of depression.
SB-224289--a novel selective (human) 5-HT1B receptor antagonist with negative intrinsic activity.[Pubmed:9776361]
Br J Pharmacol. 1998 Sep;125(1):202-8.
1. Human 5-HT1B (h5-HT1B) and human 5-HT1D (h5-HT1D) receptors show remarkably similar pharmacology with few compounds discriminating the receptors. We report here on a novel compound, SB-224289 (1'-Methyl-5-[[2'-methyl-4'-(5-methyl- 1,2,4-oxadiazol-3-yl)biphenyl-4-yl]carbonyl]-2,3,6,7-tetrahydro spiro [furo [2,3-f]indole-3,4'-piperidine] oxalate), which has high affinity for h5-HT1B receptors (pK1=8.16+/-0.06) and displays over 75 fold selectivity for the h5-HT1B receptor over all other 5-HT receptors including the h5-HT1D receptor and all other receptors tested thus far. 2. Functional activity of SB-224289 was measured in a [15S]GTPgammaS binding assay on recombinant h5-HT1B and h5-HT1D receptors expressed in Chinese Hamster Ovary (CHO) cells. SB-224289 displayed negative intrinsic activity at both receptors with higher potency at h5-HT1B receptors. SB-224289 caused a rightward shift of agonist concentration response curves consistent with competitive antagonism and generated affinities comparable with those obtained from competition radioligand receptor binding studies. 3. SB-224289 potentiated [3H]5-HT release from electrically stimulated guinea-pig cerebral cortical slices to the same extent as as the non-selective 5-HT1 antagonist methiothepin. SB-224289 also fully reversed the inhibitory effect of exogenously superfused 5-HT on electrically stimulated release. 4. Using SB-224289 as a tool compound, we confirm that in guinea-pig cerebral cortex the terminal 5-HT autoreceptor is of the 5-HT1B subtype.
Stimulation of 5-HT1B receptors causes hypothermia in the guinea pig.[Pubmed:9274976]
Eur J Pharmacol. 1997 Jul 23;331(2-3):169-74.
The selective, brain penetrant, 5-HT(1B/D) (formerly 5-HT(1D beta/alpha)) receptor agonist SKF-99101H (3-(2-dimethylaminoethyl)-4-chloro-5-propoxyindole hemifumarate) (30 mg/kg i.p.) causes a dose related fall in rectal temperature in guinea pigs which previous studies have shown to be blocked by the non-selective 5-HT(1B/D) receptor antagonist GR-127935 (N-[4-methoxy-3-(4-methyl-1-piperazinyl) phenyl]-2'-methyl-4'-(5-methyl-1,2,4-oxadiazol-3-yl) [1,1'biphenyl]-4-carboxamide oxalate). The present study shows that the hypothermic response to SKF-99101H is dose-dependently blocked by SB-224289G (1'-methyl-5-(2'-methyl-4'-[(5-methyl-1,2,4-oxadiazol-3-yl)bipheny l-4-yl]carbonyl)-2,3,6,7-tetrahydrospiro[furo[2,3-f]indole-3,4'-pi peridone] hemioxalate) (0.3-10.0 mg/kg p.o.) (ED50 3.62 mg/kg), which is the first compound to be described which is more than 60 fold selective for the 5-HT1B receptor over the 5-HT1D receptor. SB-216641A (N-[3-(2-dimethylamino) ethoxy-4-methoxy-phenyl] 2'-methyl-4'-(5-methyl-1,2,4-oxadiazol-3-yl)-(1,1'-biphenyl)-4-car boxamide hydrochloride) (0.6-20.0 mg/kg i.p.), which is somewhat less selective (30 fold) for the 5-HT1B receptor over the 5-HT1D receptor had a similar effect (ED50 4.43 mg/kg). The brain penetrant 5-HT1D selective receptor antagonist, BRL-15572 (4-(3-chlorophenyl)-alpha-(diphenylmethyl)-1-piperazineethanol+ ++ dihydrochloride) (0.3-100.0 mg/kg i.p.) was inactive. When administered alone neither BRL-15572 (0.1-10 mg/kg i.p.) nor SB-224289G (2.2-22 mg/kg p.o.) had an effect on body temperature. These data demonstrate that 5-HT1B (formerly 5-HT(1D beta)) and not 5-HT1D (formerly 5-HT(1D alpha)) receptors mediate the hypothermic response to SKF-99101H (30 mg/kg i.p.) in guinea pigs. The compounds described are useful pharmacological tools for distinguishing responses to 5-HT1B and 5-HT1D receptors.


