PterostilbeneCAS# 537-42-8 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 537-42-8 | SDF | Download SDF |
| PubChem ID | 5281727 | Appearance | White powder |
| Formula | C16H16O3 | M.Wt | 256.30 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Synonyms | trans-4-Hydroxy 3',5'-dimethoxystilbene | ||
| Solubility | >11.4mg/mL in DMSO | ||
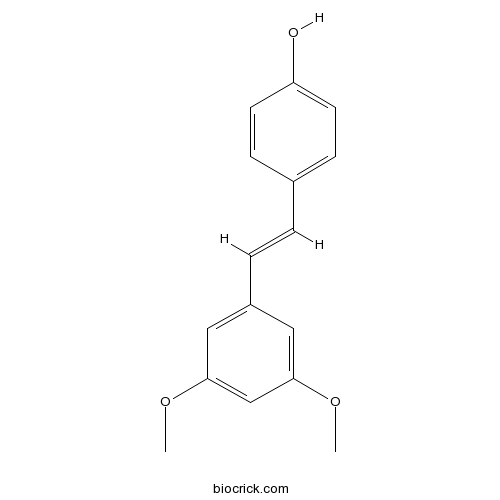
| Chemical Name | 4-[(E)-2-(3,5-dimethoxyphenyl)ethenyl]phenol | ||
| SMILES | COC1=CC(=CC(=C1)C=CC2=CC=C(C=C2)O)OC | ||
| Standard InChIKey | VLEUZFDZJKSGMX-ONEGZZNKSA-N | ||
| Standard InChI | InChI=1S/C16H16O3/c1-18-15-9-13(10-16(11-15)19-2)4-3-12-5-7-14(17)8-6-12/h3-11,17H,1-2H3/b4-3+ | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Pterostilbene acts as a peroxisome proliferator-activated receptor alpha (PPARalpha) agonist, it has been implicated in anticarcinogenesis, antioxidant, modulation of neurological disease, anti-inflammation, attenuation of vascular disease, and amelioration of diabetes. Pterostilbene downregulates inflammatory iNOS and COX-2 gene expression in macrophages by inhibiting the activation of NFkappaB by interfering with the activation of PI3K/Akt/IKK and MAPK. Pterostilbene may protect HUVECs against oxLDL-induced apoptosis by downregulating LOX-1-mediated activation through a pathway involving oxidative stress, p53, mitochondria, cytochrome c and caspase protease. |
| Targets | NOS | COX | p65 | NF-kB | PI3K | Akt | p38MAPK | IkB | GLUT | PPAR | LDL | LOX | IKK |
| In vitro | Pterostilbene suppressed lipopolysaccharide-induced up-expression of iNOS and COX-2 in murine macrophages.[Pubmed: 18656926 ]J Agric Food Chem. 2008 Aug 27;56(16):7502-9.Pterostilbene, an active constituent of blueberries, is known to possess anti-inflammatory activity and also to induce apoptosis in various types of cancer cells. Pterostilbene, an Active Constituent of Blueberries, Stimulates Nitric Oxide Production via Activation of Endothelial Nitric Oxide Synthase in Human Umbilical Vein Endothelial Cells.[Pubmed: 26008990]Plant Foods Hum Nutr. 2015 May 26.Endothelial dysfunction, a key process in development of cardiovascular diseases, is largely due to reduced nitric oxide (NO) derived from endothelial NO synthase (eNOS). Resveratrol has been reported to stimulate NO production via estrogen receptor α (ERα) activation in endothelial cells. |
| In vivo | Pterostilbene improves glycaemic control in rats fed an obesogenic diet: involvement of skeletal muscle and liver.[Pubmed: 25998070]Food Funct. 2015 Jun 10;6(6):1968-76.This study aims to determine whether Pterostilbene improves glycaemic control in rats showing insulin resistance induced by an obesogenic diet. Pterostilbene protects vascular endothelial cells against oxidized low-density lipoprotein-induced apoptosis in vitro and in vivo.[Pubmed: 21928089 ]Apoptosis. 2012 Jan;17(1):25-36.Vascular endothelial cell (VEC) apoptosis is the main event occurring during the development of atherosclerosis. Pterostilbene (PT), a natural dimethylated analog of resveratrol, has been the subject of intense research in cancer and inflammation. However, the protective effects of PT against oxidized low-density lipoprotein (oxLDL)-induced apoptosis in VECs have not been clarified. |
| Kinase Assay | Pterostilbene, a new agonist for the peroxisome proliferator-activated receptor alpha-isoform, lowers plasma lipoproteins and cholesterol in hypercholesterolemic hamsters.[Pubmed: 15853379 ]J Agric Food Chem. 2005 May 4;53(9):3403-7.Resveratrol, a stilbenoid antioxidant found in grapes, wine, peanuts and other berries, has been reported to have hypolipidemic properties. |
| Animal Research | Association between Pterostilbene and Quercetin Inhibits Metastatic Activity of B16 Melanoma1[Pubmed: 15736313]Neoplasia. 2005 Jan; 7(1): 37–47.Inhibition of cancer growth by resveratrol (trans-3,5,4'-trihydroxystilbene; RESV), a phytoalexin present in many plant species, is limited by its low bioavailability. Pterostilbene (3,5-dimethoxy-4'-hydroxystilbene; PTER) and quercetin (3,3',4',5,6-pentahydroxyflavone; QUER), two structurally related and naturally occurring small polyphenols, show longer half-life in vivo. |

Pterostilbene Dilution Calculator

Pterostilbene Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.9017 mL | 19.5084 mL | 39.0168 mL | 78.0336 mL | 97.5419 mL |
| 5 mM | 0.7803 mL | 3.9017 mL | 7.8034 mL | 15.6067 mL | 19.5084 mL |
| 10 mM | 0.3902 mL | 1.9508 mL | 3.9017 mL | 7.8034 mL | 9.7542 mL |
| 50 mM | 0.078 mL | 0.3902 mL | 0.7803 mL | 1.5607 mL | 1.9508 mL |
| 100 mM | 0.039 mL | 0.1951 mL | 0.3902 mL | 0.7803 mL | 0.9754 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Chlorophorin
Catalog No.:BCN3288
CAS No.:537-41-7
- Convolvine
Catalog No.:BCN1904
CAS No.:537-30-4
- Isoporoidine
Catalog No.:BCN1890
CAS No.:537-28-0
- Tropacocaine
Catalog No.:BCN1934
CAS No.:537-26-8
- Diperodon HCl
Catalog No.:BCC3766
CAS No.:537-12-2
- 3-Acetyl-5-Hydroxymethyl-7-Hydroxycoumarin
Catalog No.:BCC9201
CAS No.:53696-74-5
- QX 314 chloride
Catalog No.:BCC7326
CAS No.:5369-03-9
- QX 222
Catalog No.:BCC6906
CAS No.:5369-00-6
- ICI 89406
Catalog No.:BCC6807
CAS No.:53671-71-9
- O-1918
Catalog No.:BCC7313
CAS No.:536697-79-7
- 4,6-Dimethoxy-2H-1-benzopyran-2-one
Catalog No.:BCN3452
CAS No.:53666-78-7
- Dehydrobruceantin
Catalog No.:BCN7617
CAS No.:53662-98-9
- N-Acetyl-L-tyrosine
Catalog No.:BCC9082
CAS No.:537-55-3
- Soyasaponin Be Methyl Ester
Catalog No.:BCN5925
CAS No.:117210-13-6
- N-Acetyl-m-toluidine
Catalog No.:BCC9083
CAS No.:537-92-8
- beta-Amyrenonol methylthiomethyl ether
Catalog No.:BCN3354
CAS No.:
- Mexiletine HCl
Catalog No.:BCC4677
CAS No.:5370-01-4
- 2-Acetyl-5-bromothiophene
Catalog No.:BCC8513
CAS No.:5370-25-2
- BML-210(CAY10433)
Catalog No.:BCC6479
CAS No.:537034-17-6
- Tubacin
Catalog No.:BCC2428
CAS No.:537049-40-4
- Carprofen
Catalog No.:BCC4645
CAS No.:53716-49-7
- Oxfendazole
Catalog No.:BCC4817
CAS No.:53716-50-0
- Bruceantinol
Catalog No.:BCN7616
CAS No.:53729-52-5
- Luteolin-7-O-glucoside
Catalog No.:BCN5388
CAS No.:5373-11-5
Association between pterostilbene and quercetin inhibits metastatic activity of B16 melanoma.[Pubmed:15736313]
Neoplasia. 2005 Jan;7(1):37-47.
Inhibition of cancer growth by resveratrol (trans-3,5,4'-trihydroxystilbene; RESV), a phytoalexin present in many plant species, is limited by its low bioavailability. Pterostilbene (3,5-dimethoxy-4'-hydroxystilbene; PTER) and quercetin (3,3',4',5,6-pentahydroxyflavone; QUER), two structurally related and naturally occurring small polyphenols, show longer half-life in vivo. In vitro growth of highly malignant B16 melanoma F10 cells (B16M-F10) is inhibited (56%) by short-time exposure (60 min/day) to PTER (40 microm) and QUER (20 microm) (approximate mean values of plasma concentrations measured within the first hour after intravenous administration of 20 mg/kg each polyphenol). Intravenous administration of PTER and QUER (20 mg/kg per day) to mice inhibits (73%) metastatic growth of B16M-F10 cell in the liver, a common site for metastasis development. The anti-metastatic mechanism involves: 1) a PTER-induced inhibition of vascular adhesion molecule 1 expression in the hepatic sinusoidal endothelium, which consequently decreases B16M-F10 cell adhesion to the endothelium through very late activation antigen 4; and 2) a QUER- and PTER-induced inhibition of Bcl-2 expression in metastatic cells, which sensitizes them to vascular endothelium-induced cytotoxicity. Our findings demonstrate that the association of PTER and QUER inhibits metastatic melanoma growth and extends host survival.
Pterostilbene suppressed lipopolysaccharide-induced up-expression of iNOS and COX-2 in murine macrophages.[Pubmed:18656926]
J Agric Food Chem. 2008 Aug 27;56(16):7502-9.
Pterostilbene, an active constituent of blueberries, is known to possess anti-inflammatory activity and also to induce apoptosis in various types of cancer cells. Here, we investigated the inhibitory effects of Pterostilbene on the induction of NO synthase (NOS) and cyclooxygenase-2 (COX-2) in murine RAW 264.7 cells activated with lipopolysaccharide (LPS). Western blotting and real-time polymerase chain reaction (PCR) analyses demonstrated that Pterostilbene significantly blocked the protein and mRNA expression of iNOS and COX-2 in LPS-induced macrophages. Treatment with Pterostilbene resulted in the reduction of LPS-induced nuclear translocation of the nuclear factor-kappaB (NFkappaB) subunit and the dependent transcriptional activity of NFkappaB by blocking phosphorylation of inhibitor kappaB (IkappaB)alpha and p65 and subsequent degradation of IkappaB alpha. Transient transfection experiments using NFkappaB reporter constructs indicated that Pterostilbene inhibits the transcriptional activity of NFkappaB in LPS-stimulated mouse macrophages. We found that Pterostilbene also inhibited LPS-induced activation of PI3K/Akt, extracellular signal-regulated kinase 1/2 and p38 MAPK. Taken together, these results show that Pterostilbene down regulates inflammatory iNOS and COX-2 gene expression in macrophages by inhibiting the activation of NFkappaB by interfering with the activation of PI3K/Akt/IKK and MAPK. These results have an important implication for using Pterostilbene toward the development of an effective anti-inflammatory agent.
Pterostilbene, an Active Constituent of Blueberries, Stimulates Nitric Oxide Production via Activation of Endothelial Nitric Oxide Synthase in Human Umbilical Vein Endothelial Cells.[Pubmed:26008990]
Plant Foods Hum Nutr. 2015 Sep;70(3):263-8.
Endothelial dysfunction, a key process in development of cardiovascular diseases, is largely due to reduced nitric oxide (NO) derived from endothelial NO synthase (eNOS). Resveratrol has been reported to stimulate NO production via estrogen receptor alpha (ERalpha) activation in endothelial cells. Here, we investigated whether two natural methylated analogs of resveratrol, Pterostilbene (Pts) and trans-3,5,4'-trimethoxystilbene (TMS), similarly to resveratrol, could influence endothelial NO release in human umbilical vein endothelial cells (HUVECs). In HUVECs exposed to Pts or TMS, NO production and phosphorylation of eNOS, protein kinase B (Akt), and ERalpha were measured by using a fluorimetric NO assay kit and Western blot analysis, respectively. Dimethylated Pts, but not trimethylated TMS, stimulated dose-dependent NO production via eNOS phosphorylation. Pts also stimulated dose-dependent phosphorylation of Akt, but not of ERalpha. NO production and eNOS phosphorylation in response to Pts were significantly abolished by the phosphoinositide 3-kinase (PI3K)/Akt inhibitor LY294002, but not by the ERalpha antagonist ICI182780. Our results suggest that Pts, but not TMS, is capable of inducing eNOS phosphorylation and the subsequent NO release, presumably, by activating PI3K/Akt pathway. The potential efficacy of Pts, an active constituent of blueberries, may aid in the prevention of cardiovascular diseases characterized by endothelial dysfunction.
Pterostilbene improves glycaemic control in rats fed an obesogenic diet: involvement of skeletal muscle and liver.[Pubmed:25998070]
Food Funct. 2015 Jun;6(6):1968-76.
This study aims to determine whether Pterostilbene improves glycaemic control in rats showing insulin resistance induced by an obesogenic diet. Rats were divided into 3 groups: the control group and two groups treated with either 15 mg kg(-1) d(-1) (PT15) or 30 mg kg(-1) d(-1) of Pterostilbene (PT30). HOMA-IR was decreased in both Pterostilbene-treated groups, but this reduction was greater in the PT15 group (-45% and -22% respectively vs. the control group). The improvement of glycaemic control was not due to a delipidating effect of Pterostilbene on skeletal muscle. In contrast, GLUT4 protein expression was increased (+58% and +52% vs. the control group), suggesting an improved glucose uptake. The phosphorylated-Akt/total Akt ratio was significantly enhanced in the PT30 group (+25%), and therefore a more efficient translocation of GLUT4 is likely. Additionally, in this group the amount of cardiotrophin-1 was significantly increased (+65%). These data suggest that the effect of Pterostilbene on Akt is mediated by this cytokine. In the liver, glucokinase activity was significantly increased only in the PT15 group (+34%), and no changes were observed in glucose-6-phosphatase activity. The beneficial effect of Pterostilbene on glycaemic control was more evident with the lower dose, probably because in the PT15 group both the muscle and the liver were contributing to this effect, but in the PT30 group only the skeletal muscle was responsible. In conclusion, Pterostilbene improves glycaemic control in rats showing insulin resistance induced by an obesogenic diet. An increase in hepatic glucokinase activity, as well as in skeletal muscle glucose uptake, seems to be involved in the anti-diabetic effect of this phenolic compound.
Pterostilbene protects vascular endothelial cells against oxidized low-density lipoprotein-induced apoptosis in vitro and in vivo.[Pubmed:21928089]
Apoptosis. 2012 Jan;17(1):25-36.
Vascular endothelial cell (VEC) apoptosis is the main event occurring during the development of atherosclerosis. Pterostilbene (PT), a natural dimethylated analog of resveratrol, has been the subject of intense research in cancer and inflammation. However, the protective effects of PT against oxidized low-density lipoprotein (oxLDL)-induced apoptosis in VECs have not been clarified. We investigated the anti-apoptotic effects of PT in vitro and in vivo in mice. PT at 0.1-5 muM possessed antioxidant properties comparable to that of trolox in a cell-free system. Exposure of human umbilical vein VECs (HUVECs) to oxLDL (200 mug/ml) induced cell shrinkage, chromatin condensation, nuclear fragmentation, and cell apoptosis, but PT protected against such injuries. In addition, PT injection strongly decreased the number of TUNEL-positive cells in the endothelium of atherosclerotic plaque from apoE(-/-) mice. OxLDL increased reactive oxygen species (ROS) levels, NF-kappaB activation, p53 accumulation, apoptotic protein levels and caspases-9 and -3 activities and decreased mitochondrial membrane potential (MMP) and cytochrome c release in HUVECs. These alterations were attenuated by pretreatment with PT. PT inhibited the expression of lectin-like oxLDL receptor-1 (LOX-1) expression in vitro and in vivo. Cotreatment with PT and siRNA of LOX-1 synergistically reduced oxLDL-induced apoptosis in HUVECs. Overexpression of LOX-1 attenuated the protection by PT and suppressed the effects of PT on oxLDL-induced oxidative stress. PT may protect HUVECs against oxLDL-induced apoptosis by downregulating LOX-1-mediated activation through a pathway involving oxidative stress, p53, mitochondria, cytochrome c and caspase protease. PT might be a potential natural anti-apoptotic agent for the treatment of atherosclerosis.
Pterostilbene, a new agonist for the peroxisome proliferator-activated receptor alpha-isoform, lowers plasma lipoproteins and cholesterol in hypercholesterolemic hamsters.[Pubmed:15853379]
J Agric Food Chem. 2005 May 4;53(9):3403-7.
Resveratrol, a stilbenoid antioxidant found in grapes, wine, peanuts and other berries, has been reported to have hypolipidemic properties. We investigated whether resveratrol and its three analogues (Pterostilbene, piceatannol, and resveratrol trimethyl ether) would activate the peroxisome proliferator-activated receptor alpha (PPARalpha) isoform. This nuclear receptor is proposed to mediate the activity of lipid-lowering drugs such as the fibrates. The four stilbenes were evaluated at 1, 10, 100, and 300 microM along with ciprofibrate (positive control), for the activation of endogenous PPARalpha in H4IIEC3 cells. Cells were transfected with a peroxisome proliferator response element-AB (rat fatty acyl CoA beta-oxidase response element)-luciferase gene reporter construct. Pterostilbene demonstrated the highest induction of PPARalpha showing 8- and 14-fold increases in luciferase activity at 100 and 300 microM, respectively, relative to the control. The maximal luciferase activity responses to Pterostilbene were higher than those obtained with the hypolipidemic drug, ciprofibrate (33910 and 19460 relative luciferase units, respectively), at 100 microM. Hypercholesterolemic hamsters fed with Pterostilbene at 25 ppm of the diet showed 29% lower plasma low density lipoprotein (LDL) cholesterol, 7% higher plasma high density lipoprotein (HDL) cholesterol, and 14% lower plasma glucose as compared to the control group. The LDL/HDL ratio was also statistically significantly lower for Pterostilbene, as compared to results for the control animals, at this diet concentration. Results from in vitro studies showed that Pterostilbene acts as a PPARalpha agonist and may be a more effective PPARalpha agonist and hypolipidemic agent than resveratrol. In vivo studies demonstrate that Pterostilbene possesses lipid and glucose lowering effects.


