PerakineCAS# 4382-56-3 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 4382-56-3 | SDF | Download SDF |
| PubChem ID | 453213 | Appearance | Powder |
| Formula | C21H22N2O3 | M.Wt | 350.4 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
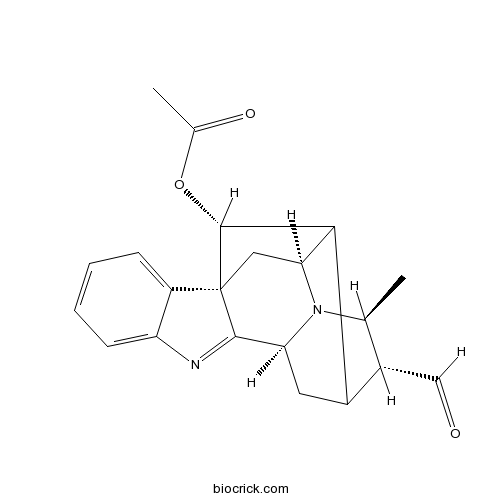
| Chemical Name | [(1R,10S,13R,14S,16S,18R)-13-formyl-14-methyl-8,15-diazahexacyclo[14.2.1.01,9.02,7.010,15.012,17]nonadeca-2,4,6,8-tetraen-18-yl] acetate | ||
| SMILES | CC1C(C2CC3N1C4C2C(C5(C4)C3=NC6=CC=CC=C56)OC(=O)C)C=O | ||
| Standard InChIKey | GDXJMOGWONJRHL-HWUILILVSA-N | ||
| Standard InChI | InChI=1S/C21H22N2O3/c1-10-13(9-24)12-7-16-19-21(14-5-3-4-6-15(14)22-19)8-17(23(10)16)18(12)20(21)26-11(2)25/h3-6,9-10,12-13,16-18,20H,7-8H2,1-2H3/t10-,12?,13-,16-,17-,18?,20+,21+/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Standard reference |
| In vitro | Purification, cloning, functional expression and characterization of perakine reductase: the first example from the AKR enzyme family, extending the alkaloidal network of the plant Rauvolfia.[Pubmed: 18409028]Plant Mol Biol. 2008 Jul;67(5):455-67.Perakine reductase (PR) catalyzes an NADPH-dependent step in a side-branch of the 10-step biosynthetic pathway of the alkaloid ajmaline.
|
| Structure Identification | J Biol Chem. 2012 Mar 30;287(14):11213-21.Crystal structure of perakine reductase, founding member of a novel aldo-keto reductase (AKR) subfamily that undergoes unique conformational changes during NADPH binding.[Pubmed: 22334702]
Natural Product Research & Development,2007,19 (10) :235-239.Indole Alkaloids from Rauwolfia vomitoria[Reference: WebLink]Fifteen indole alkaloids were isolated from the roots of Rauwolfia vomitoria.
|

Perakine Dilution Calculator

Perakine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.8539 mL | 14.2694 mL | 28.5388 mL | 57.0776 mL | 71.347 mL |
| 5 mM | 0.5708 mL | 2.8539 mL | 5.7078 mL | 11.4155 mL | 14.2694 mL |
| 10 mM | 0.2854 mL | 1.4269 mL | 2.8539 mL | 5.7078 mL | 7.1347 mL |
| 50 mM | 0.0571 mL | 0.2854 mL | 0.5708 mL | 1.1416 mL | 1.4269 mL |
| 100 mM | 0.0285 mL | 0.1427 mL | 0.2854 mL | 0.5708 mL | 0.7135 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Robtin
Catalog No.:BCN5490
CAS No.:4382-34-7
- Dihydrorobinetin
Catalog No.:BCN5489
CAS No.:4382-33-6
- Quercetin 3,3'-dimethyl ether
Catalog No.:BCN7781
CAS No.:4382-17-6
- SMI-4a
Catalog No.:BCC2233
CAS No.:438190-29-5
- 4-(4-Aminophenyl)morpholin-3-one
Catalog No.:BCC8650
CAS No.:438056-69-0
- H-Thr(tBu)-OH
Catalog No.:BCC3106
CAS No.:4378-13-6
- Salinosporamide A (NPI-0052, Marizomib)
Catalog No.:BCC2094
CAS No.:437742-34-2
- Crategolic acid
Catalog No.:BCN5487
CAS No.:4373-41-5
- Xanthinol nicotinate
Catalog No.:BCC9191
CAS No.:437-74-1
- Genkwanin
Catalog No.:BCN5488
CAS No.:437-64-9
- Gentisin
Catalog No.:BCN7518
CAS No.:437-50-3
- MRS 2365
Catalog No.:BCC5879
CAS No.:436847-09-5
- PFK-015
Catalog No.:BCC5280
CAS No.:4382-63-2
- 3(20)-Phytene-1,2-diol
Catalog No.:BCN6589
CAS No.:438536-34-6
- JIP-1 (153-163)
Catalog No.:BCC5777
CAS No.:438567-88-5
- 2-Amino-3-methylbenzoic acid
Catalog No.:BCC8528
CAS No.:4389-45-1
- Gentianine
Catalog No.:BCN5492
CAS No.:439-89-4
- O-2093
Catalog No.:BCC7070
CAS No.:439080-01-0
- Afatinib
Catalog No.:BCC3656
CAS No.:439081-18-2
- Bay 60-7550
Catalog No.:BCC1405
CAS No.:439083-90-6
- Lasmiditan
Catalog No.:BCC4077
CAS No.:439239-90-4
- GW 627368
Catalog No.:BCC7961
CAS No.:439288-66-1
- ITK inhibitor
Catalog No.:BCC1662
CAS No.:439574-61-5
- BMS-509744
Catalog No.:BCC1424
CAS No.:439575-02-7
Crystal structure of perakine reductase, founding member of a novel aldo-keto reductase (AKR) subfamily that undergoes unique conformational changes during NADPH binding.[Pubmed:22334702]
J Biol Chem. 2012 Mar 30;287(14):11213-21.
Perakine reductase (PR) catalyzes the NADPH-dependent reduction of the aldehyde Perakine to yield the alcohol raucaffrinoline in the biosynthetic pathway of ajmaline in Rauvolfia, a key step in indole alkaloid biosynthesis. Sequence alignment shows that PR is the founder of the new AKR13D subfamily and is designated AKR13D1. The x-ray structure of methylated His(6)-PR was solved to 2.31 A. However, the active site of PR was blocked by the connected parts of the neighbor symmetric molecule in the crystal. To break the interactions and obtain the enzyme-ligand complexes, the A213W mutant was generated. The atomic structure of His(6)-PR-A213W complex with NADPH was determined at 1.77 A. Overall, PR folds in an unusual alpha(8)/beta(6) barrel that has not been observed in any other AKR protein to date. NADPH binds in an extended pocket, but the nicotinamide riboside moiety is disordered. Upon NADPH binding, dramatic conformational changes and movements were observed: two additional beta-strands in the C terminus become ordered to form one alpha-helix, and a movement of up to 24 A occurs. This conformational change creates a large space that allows the binding of substrates of variable size for PR and enhances the enzyme activity; as a result cooperative kinetics are observed as NADPH is varied. As the founding member of the new AKR13D subfamily, PR also provides a structural template and model of cofactor binding for the AKR13 family.
Purification, cloning, functional expression and characterization of perakine reductase: the first example from the AKR enzyme family, extending the alkaloidal network of the plant Rauvolfia.[Pubmed:18409028]
Plant Mol Biol. 2008 Jul;67(5):455-67.
Perakine reductase (PR) catalyzes an NADPH-dependent step in a side-branch of the 10-step biosynthetic pathway of the alkaloid ajmaline. The enzyme was cloned by a "reverse-genetic" approach from cell suspension cultures of the plant Rauvolfia serpentina (Apocynaceae) and functionally expressed in Escherichia coli as the N-terminal His(6)-tagged protein. PR displays a broad substrate acceptance, converting 16 out of 28 tested compounds with reducible carbonyl function which belong to three substrate groups: benzaldehyde, cinnamic aldehyde derivatives and monoterpenoid indole alkaloids. The enzyme has an extraordinary selectivity in the group of alkaloids. Sequence alignments define PR as a new member of the aldo-keto reductase (AKR) super family, exhibiting the conserved catalytic tetrad Asp52, Tyr57, Lys84, His126. Site-directed mutagenesis of each of these functional residues to an alanine residue results in >97.8% loss of enzyme activity, in compounds of each substrate group. PR represents the first example of the large AKR-family which is involved in the biosynthesis of plant monoterpenoid indole alkaloids. In addition to a new esterase, PR significantly extends the Rauvolfia alkaloid network to the novel group of peraksine alkaloids.


