PalmitoylethanolamideFAAH and PAA substrate. Selective GPR55 agonist CAS# 544-31-0 |
- Dexpramipexole dihydrochloride
Catalog No.:BCC1528
CAS No.:104632-27-1
- Dexpramipexole
Catalog No.:BCC1527
CAS No.:104632-28-2
- Cariprazine hydrochloride
Catalog No.:BCC1454
CAS No.:1083076-69-0
- Cariprazine
Catalog No.:BCC1453
CAS No.:839712-12-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 544-31-0 | SDF | Download SDF |
| PubChem ID | 4671 | Appearance | Powder |
| Formula | C18H37NO2 | M.Wt | 299.5 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | PEA | ||
| Solubility | Soluble to 20 mM in DMSO and to 25 mM in ethanol | ||
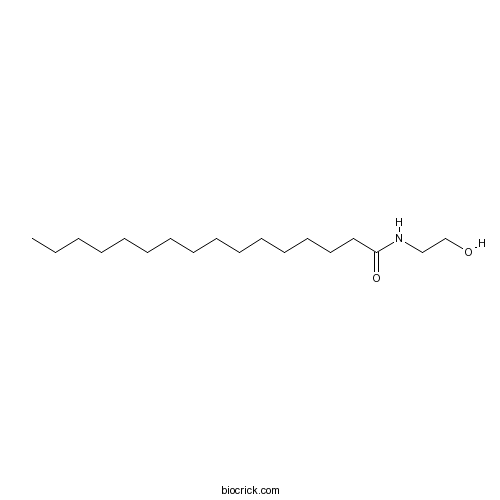
| Chemical Name | N-(2-hydroxyethyl)hexadecanamide | ||
| SMILES | CCCCCCCCCCCCCCCC(=O)NCCO | ||
| Standard InChIKey | HXYVTAGFYLMHSO-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C18H37NO2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-18(21)19-16-17-20/h20H,2-17H2,1H3,(H,19,21) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Endogenous lipid that acts as a selective GPR55 agonist (EC50 values are 4, 19 800 and > 30 000 nM at GPR55, CB2 and CB1 receptors respectively). Substrate for fatty acid amide hydrolase (FAAH) and PEA-preferring acid amidase (PAA) and exhibits antinociceptive and anticonvulsant in vivo. Directly activates PPARα (EC50 = 3 μM) producing robust anti-inflammatory actions. |

Palmitoylethanolamide Dilution Calculator

Palmitoylethanolamide Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.3389 mL | 16.6945 mL | 33.389 mL | 66.778 mL | 83.4725 mL |
| 5 mM | 0.6678 mL | 3.3389 mL | 6.6778 mL | 13.3556 mL | 16.6945 mL |
| 10 mM | 0.3339 mL | 1.6694 mL | 3.3389 mL | 6.6778 mL | 8.3472 mL |
| 50 mM | 0.0668 mL | 0.3339 mL | 0.6678 mL | 1.3356 mL | 1.6694 mL |
| 100 mM | 0.0334 mL | 0.1669 mL | 0.3339 mL | 0.6678 mL | 0.8347 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Lirinidine
Catalog No.:BCN8274
CAS No.:54383-28-7
- N,N-Bis(2-hydroxyethyl)-p-phenylenediamine sulphate
Catalog No.:BCN8366
CAS No.:54381-16-7
- 7-Hydroxy-5,8-dimethoxyflavanone
Catalog No.:BCN5722
CAS No.:54377-24-1
- BMS-538203
Catalog No.:BCC4136
CAS No.:543730-41-2
- 4'-Methoxyacetoacetanilide
Catalog No.:BCC8712
CAS No.:5437-98-9
- Decarine
Catalog No.:BCN5721
CAS No.:54354-62-0
- Eriodictyol 7,3'-dimethyl ether
Catalog No.:BCN8105
CAS No.:54352-60-2
- 4beta-Hydroxywithanolide E
Catalog No.:BCN7572
CAS No.:54334-04-2
- Protopseudohypericin
Catalog No.:BCN2813
CAS No.:54328-09-5
- Amsacrine hydrochloride
Catalog No.:BCC4310
CAS No.:54301-15-4
- Ac-Gly-OH
Catalog No.:BCC2943
CAS No.:543-24-8
- 2',4'-Dihydroxy-3',6'-dimethoxydihydrochalcone
Catalog No.:BCN1421
CAS No.:54299-52-4
- Myristic acid
Catalog No.:BCN8390
CAS No.:544-63-8
- Norcantharidin
Catalog No.:BCN1281
CAS No.:5442-12-6
- Capadenoson
Catalog No.:BCC1450
CAS No.:544417-40-5
- HIV-1 integrase inhibitor
Catalog No.:BCC1618
CAS No.:544467-07-4
- c-di-AMP
Catalog No.:BCC8054
CAS No.:54447-84-6
- MRS 1845
Catalog No.:BCC7198
CAS No.:544478-19-5
- UBP 282
Catalog No.:BCC7171
CAS No.:544697-47-4
- JNJ 10181457 dihydrochloride
Catalog No.:BCC7842
CAS No.:544707-20-2
- 3alpha-dihydrocadambine
Catalog No.:BCN8151
CAS No.:54483-84-0
- Jolkinolide E
Catalog No.:BCN3772
CAS No.:54494-34-7
- 5-Glutinen-3-ol
Catalog No.:BCN5723
CAS No.:545-24-4
- Uvaol
Catalog No.:BCN5724
CAS No.:545-46-0
Ultra-micronized Palmitoylethanolamide: An Efficacious Adjuvant Therapy for Parkinson's Disease.[Pubmed:28325153]
CNS Neurol Disord Drug Targets. 2017;16(6):705-713.
BACKGROUND: Parkinson's disease (PD) is the subject of intense efforts to develop strategies that slow down or stop disease progression and disability. Substantial evidence points to a prominent role for neuroinflammation in the underlying dopaminergic cell death. Ultramicronized Palmitoylethanolamide (um-PEA) is well-known for its ability to promote the resolution of neuroinflammation and exert neuroprotection. This study was designed to assess the efficacy of um-PEA as adjuvant therapy in patients with advanced PD. METHOD: Thirty PD patients receiving levodopa were included in the study. The revised- Movement Disorder Society/Unified Parkinson's Disease Rating Scale (MDS-UPDRS) questionnaire was used to assess motor and non-motor symptoms. Clinical assessments were carried out before and after addition of um-PEA (600 mg). MDS-UPDRS questionnaire total score for parts I, II, III, and IV was analyzed using the Generalized Linear Mixed Model, followed by the Wilcoxon signed-rank test to evaluate the difference of each item's mean score between baseline and end of um-PEA treatment. RESULTS: Addition of um-PEA to PD patients receiving levodopa therapy elicited a significant and progressive reduction in the total MDS-UPDRS score (parts I, II, III and IV). For each item, the mean score difference between baseline and end of um-PEA treatment showed a significant reduction in most nonmotor and motor symptoms. The number of patients with symptoms at basal was reduced after one year of um-PEA treatment. None of the participants reported side effects attributable to the addition of um-PEA. CONCLUSION: um-PEA slowed down disease progression and disability in PD patients, suggesting that um-PEA may be an efficacious adjuvant therapy for PD.
Palmitoylethanolamide induces microglia changes associated with increased migration and phagocytic activity: involvement of the CB2 receptor.[Pubmed:28336953]
Sci Rep. 2017 Mar 23;7(1):375.
The endogenous fatty acid amide Palmitoylethanolamide (PEA) has been shown to exert anti-inflammatory actions mainly through inhibition of the release of pro-inflammatory molecules from mast cells, monocytes and macrophages. Indirect activation of the endocannabinoid (eCB) system is among the several mechanisms of action that have been proposed to underlie the different effects of PEA in vivo. In this study, we used cultured rat microglia and human macrophages to evaluate whether PEA affects eCB signaling. PEA was found to increase CB2 mRNA and protein expression through peroxisome proliferator-activated receptor-alpha (PPAR-alpha) activation. This novel gene regulation mechanism was demonstrated through: (i) pharmacological PPAR-alpha manipulation, (ii) PPAR-alpha mRNA silencing, (iii) chromatin immunoprecipitation. Moreover, exposure to PEA induced morphological changes associated with a reactive microglial phenotype, including increased phagocytosis and migratory activity. Our findings suggest indirect regulation of microglial CB2R expression as a new possible mechanism underlying the effects of PEA. PEA can be explored as a useful tool for preventing/treating the symptoms associated with neuroinflammation in CNS disorders.
Palmitoylethanolamide Reduces Neuropsychiatric Behaviors by Restoring Cortical Electrophysiological Activity in a Mouse Model of Mild Traumatic Brain Injury.[Pubmed:28321191]
Front Pharmacol. 2017 Mar 6;8:95.
Traumatic brain injury (TBI) represents a major public health problem, which is associated with neurological dysfunction. In severe or moderate cases of TBI, in addition to its high mortality rate, subjects may encounter diverse behavioral dysfunctions. Previous reports suggest that an association between TBI and chronic pain syndromes tends to be more common in patients with mild forms of brain injury. Despite causing minimal brain damage, mild TBI (mTBI) often leads to persistent psychologically debilitating symptoms, which can include anxiety, various forms of memory and learning deficits, and depression. At present, no effective treatment options are available for these symptoms, and little is known about the complex cellular activity affecting neuronal activity that occurs in response to TBI during its late phase. Here, we used a mouse model to investigate the effect of Palmitoylethanolamide (PEA) on both the sensorial and neuropsychiatric dysfunctions associated with mTBI through behavioral, electrophysiological, and biomolecular approaches. Fourteen-day mTBI mice developed anxious, aggressive, and reckless behavior, whilst depressive-like behavior and impaired social interactions were observed from the 60th day onward. Altered behavior was associated with changes in interleukin 1 beta (IL-1beta) expression levels and neuronal firing activity in the medial prefrontal cortex. Compared with vehicle, PEA restored the behavioral phenotype and partially normalized the biochemical and functional changes occurring at the supraspinal level. In conclusion, our findings reveal some of the supraspinal modifications responsible for the behavioral alterations associated with mTBI and suggest PEA as a pharmacological tool to ameliorate neurological dysfunction induced by the trauma.
The anti-inflammatory compound palmitoylethanolamide inhibits prostaglandin and hydroxyeicosatetraenoic acid production by a macrophage cell line.[Pubmed:28357126]
Pharmacol Res Perspect. 2017 Feb 27;5(2):e00300.
The anti-inflammatory agent Palmitoylethanolamide (PEA) reduces cyclooxygenase (COX) activity in vivo in a model of inflammatory pain. It is not known whether the compound reduces prostaglandin production in RAW264.7 cells, whether such an action is affected by compounds preventing the breakdown of endogenous PEA, whether other oxylipins are affected, or whether PEA produces direct effects upon the COX-2 enzyme. RAW264.7 cells were treated with lipopolysaccharide and interferon-gamma to induce COX-2. At the level of mRNA, COX-2 was induced >1000-fold following 24 h of the treatment. Coincubation with PEA (10 mumol/L) did not affect the levels of COX-2, but reduced the levels of prostaglandins D2 and E2 as well as 11- and 15-hydroxyeicosatetraenoic acid, which can also be synthesised by a COX-2 pathway in macrophages. These effects were retained when hydrolysis of PEA to palmitic acid was blocked. Linoleic acid-derived oxylipin levels were not affected by PEA. No direct effects of PEA upon the oxygenation of either arachidonic acid or 2-arachidonoylglycerol by COX-2 were found. It is concluded that in lipopolysaccharide and interferon-gamma-stimulated RAW264.7 cells, PEA reduces the production of COX-2-derived oxylipins in a manner that is retained when its metabolism to palmitic acid is inhibited.
The orphan receptor GPR55 is a novel cannabinoid receptor.[Pubmed:17876302]
Br J Pharmacol. 2007 Dec;152(7):1092-101.
BACKGROUND: The endocannabinoid system functions through two well characterized receptor systems, the CB1 and CB2 receptors. Work by a number of groups in recent years has provided evidence that the system is more complicated and additional receptor types should exist to explain ligand activity in a number of physiological processes. EXPERIMENTAL APPROACH: Cells transfected with the human cDNA for GPR55 were tested for their ability to bind and to mediate GTPgammaS binding by cannabinoid ligands. Using an antibody and peptide blocking approach, the nature of the G-protein coupling was determined and further demonstrated by measuring activity of downstream signalling pathways. KEY RESULTS: We demonstrate that GPR55 binds to and is activated by the cannabinoid ligand CP55940. In addition endocannabinoids including anandamide and virodhamine activate GTPgammaS binding via GPR55 with nM potencies. Ligands such as cannabidiol and abnormal cannabidiol which exhibit no CB1 or CB2 activity and are believed to function at a novel cannabinoid receptor, also showed activity at GPR55. GPR55 couples to Galpha13 and can mediate activation of rhoA, cdc42 and rac1. CONCLUSIONS: These data suggest that GPR55 is a novel cannabinoid receptor, and its ligand profile with respect to CB1 and CB2 described here will permit delineation of its physiological function(s).
The search for the palmitoylethanolamide receptor.[Pubmed:15963531]
Life Sci. 2005 Aug 19;77(14):1685-98.
Palmitoylethanolamide (PEA), the naturally occurring amide of ethanolamine and palmitic acid, is an endogenous lipid that modulates pain and inflammation. Although the anti-inflammatory effects of PEA were first characterized nearly 50 years ago, the identity of the receptor mediating these actions has long remained elusive. We recently identified the ligand-activated transcription factor, peroxisome proliferator-activated receptor-alpha (PPAR-alpha), as the receptor mediating the anti-inflammatory actions of this lipid amide. Here we outline the history of PEA, starting with its initial discovery in the 1950s, and discuss the pharmacological properties of this compound, particularly in regards to its ability to activate PPAR-alpha.
Palmitoylethanolamide, endocannabinoids and related cannabimimetic compounds in protection against tissue inflammation and pain: potential use in companion animals.[Pubmed:16324856]
Vet J. 2007 Jan;173(1):21-30.
Endocannabinoids have analgesic/anti-inflammatory properties. The biology of endocannabinoids, their receptors, signalling mechanisms and role in the regulation of physiological processes have been extensively reviewed. This review focuses on the role of Palmitoylethanolamide (PEA), an endogenous fatty acid amide analogue of the endocannabinoid anandamide, in tissue protective mechanisms. PEA was first identified almost five decades ago in lipid extracts of various natural products, and its anti-inflammatory and antinociceptive effects were established later. Evidence exists that PEA is synthesised during inflammation and tissue damage and a number of beneficial effects, including the relief of inflammation and pruritus, have been shown to be useful in the control of neurogenic and neuropathic pain. The postulated hypotheses as to the mode of action of PEA include a possible local autacoid-like mediator activity regulating mast-cell activity and putative activation of cannabinoids and vanilloid TRPV1 receptors via "entourage" effects. The large number of scientific investigations into the effects of PEA and PEA-related compounds has given rise to new therapeutic opportunities. In spite of the multitude of therapies currently employed to control inflammation, pain, pruritus and tissue damage, the possibility of using a natural compound, such as PEA to manipulate endogenous protective mechanisms may be considered a beneficial novel therapeutic strategy in veterinary medicine.
The palmitoylethanolamide family: a new class of anti-inflammatory agents?[Pubmed:11945130]
Curr Med Chem. 2002 Mar;9(6):663-74.
The discovery of anandamide as an endogenous ligand for the cannabinoid receptors has led to a resurgence of interest in the fatty acid amides. However, N-palmitoylethanolamine (PEA), a shorter and fully saturated analogue of anandamide, has been known since the fifties. This endogenous compound is a member of the N-acylethanolamines, found in most mammalian tissues. PEA is accumulated during inflammation and has been demonstrated to have a number of anti-inflammatory effects, including beneficial effects in clinically relevant animal models of inflammatory pain. It is now engaged in phase II clinical development, and two studies regarding the treatment of chronic lumbosciatalgia and multiple sclerosis are in progress. However, its precise mechanism of action remains debated. In the present review, the biochemical and pharmacological properties of PEA are discussed, in particular with respect to its analgesic and anti-inflammatory properties.
Anticonvulsant activity of N-palmitoylethanolamide, a putative endocannabinoid, in mice.[Pubmed:11442148]
Epilepsia. 2001 Mar;42(3):321-7.
PURPOSE: The purpose of this study was to evaluate in mice the anticonvulsant potential of N-Palmitoylethanolamide, a putative endocannabinoid that accumulates in the body during inflammatory processes. METHODS: N-Palmitoylethanolamide was injected intraperitoneally (i.p.) in mice and evaluated for anticonvulsant activity [in maximal electroshock seizure (MES) and chemical-induced convulsions] and for neurologic impairment (rotorod). It was compared with anandamide and with different palmitic acid analogues as well as with reference anticonvulsants (AEDs) injected under the same conditions. RESULTS: The MES test showed, after i.p. administration to mice, that N-palmitoy]ethanolamide had an median effective dose (ED50) value comparable to that of phenytoin (PHT; 8.9 and 9.2 mg/kg, respectively). In the subcutaneous pentylenetetrazol test and in the 3-mercaptropropionic acid test, it was effective only against tonic convulsions. N-Palmitoylethanolamide was devoid of neurologic impairment < or = 250 mg/kg, yielding a high protective index. CONCLUSIONS: N-Palmitoylethanolamide, an endogenous compound with antiinflammatory and analgesic activities, is a potent AED in mice. Its precise mechanism of action remains to be elucidated.


