N-CaffeoyltryptophanCAS# 109163-69-1 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 109163-69-1 | SDF | Download SDF |
| PubChem ID | 15228042 | Appearance | Powder |
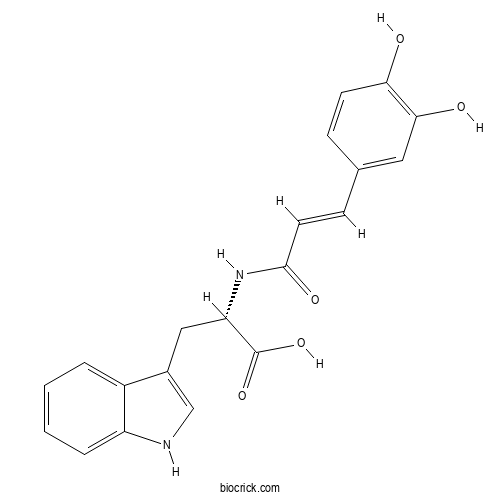
| Formula | C20H18N2O5 | M.Wt | 366.4 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (2S)-2-[[(E)-3-(3,4-dihydroxyphenyl)prop-2-enoyl]amino]-3-(1H-indol-3-yl)propanoic acid | ||
| SMILES | C1=CC=C2C(=C1)C(=CN2)CC(C(=O)O)NC(=O)C=CC3=CC(=C(C=C3)O)O | ||
| Standard InChIKey | XITPERBRJNUFSB-BVBGJJFLSA-N | ||
| Standard InChI | InChI=1S/C20H18N2O5/c23-17-7-5-12(9-18(17)24)6-8-19(25)22-16(20(26)27)10-13-11-21-15-4-2-1-3-14(13)15/h1-9,11,16,21,23-24H,10H2,(H,22,25)(H,26,27)/b8-6+/t16-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

N-Caffeoyltryptophan Dilution Calculator

N-Caffeoyltryptophan Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.7293 mL | 13.6463 mL | 27.2926 mL | 54.5852 mL | 68.2314 mL |
| 5 mM | 0.5459 mL | 2.7293 mL | 5.4585 mL | 10.917 mL | 13.6463 mL |
| 10 mM | 0.2729 mL | 1.3646 mL | 2.7293 mL | 5.4585 mL | 6.8231 mL |
| 50 mM | 0.0546 mL | 0.2729 mL | 0.5459 mL | 1.0917 mL | 1.3646 mL |
| 100 mM | 0.0273 mL | 0.1365 mL | 0.2729 mL | 0.5459 mL | 0.6823 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 3alpha-Hydroxytirucalla-7,24-dien-21-oic acid
Catalog No.:BCX2175
CAS No.:82509-40-8
- Acacetin 7-O-glucuronide
Catalog No.:BCX2174
CAS No.:38226-83-4
- 3-O-Acetyl-11-methoxy-alpha-boswellic acid
Catalog No.:BCX2173
CAS No.:1619260-05-7
- Tetrandrine 2' beta-oxide (Tetrandrine-2'-N-beta-oxide)
Catalog No.:BCX2172
CAS No.:62828-25-5
- 1-(4,7-Dimethoxy-1,3-benzodioxol-5-yl)-1-propanone
Catalog No.:BCX2171
CAS No.:107882-48-4
- Peiioside A
Catalog No.:BCX2170
CAS No.:1610618-94-4
- Obtusichromoneside A
Catalog No.:BCX2169
CAS No.:2414481-20-0
- 6,9,10-Trihydroxyoctadec-7-enoic acid
Catalog No.:BCX2168
CAS No.:1134486-96-6
- Steponine
Catalog No.:BCX2167
CAS No.:17132-69-3
- Meliasendanin D 4'-O-beta-D-glucopyranoside
Catalog No.:BCX2166
CAS No.:1931100-79-6
- Styraxlignolide G
Catalog No.:BCX2165
CAS No.:41607-21-0
- Cistanoside H
Catalog No.:BCX2164
CAS No.:104806-92-0
- Crenatoside
Catalog No.:BCX2177
CAS No.:Crenatoside
- Na-p-Hydroxycoumaroyltryptophan
Catalog No.:BCX2178
CAS No.:171741-93-8
- 5-Methoxy-[8]-gingerol
Catalog No.:BCX2179
CAS No.:863780-81-8
- 3-O-Feruloyl-5-caffeoylquinic acid
Catalog No.:BCX2180
CAS No.:173658-86-1
- Angustone A
Catalog No.:BCX2181
CAS No.:90686-13-8
- Pulsatiloside B
Catalog No.:BCX2182
CAS No.:178825-03-1
- 7alpha-Hydroxydehydroabietic acid
Catalog No.:BCX2183
CAS No.:76235-98-8
- Verticilla-4(20),7,11-triene
Catalog No.:BCX2184
CAS No.:386223-19-4
- Angelol D
Catalog No.:BCX2185
CAS No.:83199-39-7
- 6-Methyl citrate
Catalog No.:BCX2186
CAS No.:26163-65-5
- Angelol E
Catalog No.:BCX2187
CAS No.:83156-03-0
- Tricin 4'-O-(erythro-beta-guaiacylglyceryl)ether
Catalog No.:BCX2188
CAS No.:1292294-32-6
[An Investigation of Substances Influencing Adipocyte Function].[Pubmed:39085056]
Yakugaku Zasshi. 2024;144(8):799-804.
Adipose tissue plays a central metabolic role in systemic energy metabolism via nutrient exchange and secretion of adipose-derived hormones and cytokines. Adipose tissue dysfunction increases the risk of developing conditions, including type 2 diabetes, coronary artery disease, stroke, and cancer, ultimately shortening healthy lifespan. Maintaining adipose tissue functions has recently garnered attention as a means to extend healthy life expectancy. We previously developed a T-cell activation-inhibitory assay, which facilitates efficient selection of candidate substances for extending healthy lifespan. Using this assay, we identified two candidate substances: Cynandione A (CA), a major component of Cynanchum wilfordii, and N-Caffeoyltryptophan (NCT) found in coffee. This review summarizes recent findings regarding the effect of CA and NCT on adipocyte (the primary cells in adipose tissue) function, and their potential contribution to extending healthy life expectancy.
N-Caffeoyltryptophan enhances adipogenic differentiation in preadipocytes and improves glucose tolerance in mice.[Pubmed:36460233]
Biochim Biophys Acta Gen Subj. 2023 Feb;1867(2):130277.
Coffee consumption has been shown to reduce the risk of developing type 2 diabetes mellitus (T2DM) in humans; however, the exact mechanism is not completely understood. Here, we demonstrate that N-Caffeoyltryptophan (CTP), an ingredient of coffee, enhances adipogenic differentiation and promotes glucose uptake into adipocytes. CTP increased lipid accumulation and adipogenic markers (PPARgamma, C/EBPalpha, and FABP4) expression in mouse 3T3-L1 preadipocyte cell lines and primary preadipocytes. In addition, CTP promoted glucose uptake in 3T3-L1 cells. In the oral glucose tolerance test, daily administration of CTP (30 mg/kg/day, i.p.) for a week reduced blood glucose levels in mice. In 3T3-L1 cells, adipogenic differentiation and increased adipogenic markers expression induced by CTP were inhibited by U0126, a selective MEK1/2 inhibitor. Furthermore, mRNA induction of Pparg by CTP was abrogated in SIRT1 siRNA-transfected 3T3-L1 cells. These results suggest the involvement of the MEK/ERK signaling and SIRT1 in the mechanism of adipogenic function of CTP. Taken together, CTP might contribute to the reduction in postprandial glycemia and a subsequent reduction in onset risk for T2DM.
C/EBPbeta Promotes LPS-Induced IL-1beta Transcription and Secretion in Alveolar Macrophages via NOD2 Signaling.[Pubmed:36120185]
J Inflamm Res. 2022 Sep 11;15:5247-5263.
OBJECTIVE: C/EBPbeta, a crucial transcription factor, regulates innate immunity and inflammatory responses. However, the role played by C/EBPbeta in alveolar macrophage (AM) inflammatory responses remains unknown. This study aimed to investigate the role and mechanism of C/EBPbeta in alveolar macrophages (AMs) from the transcriptional level and to search for natural compounds targeting C/EBPbeta. METHODS: Rat AMs were infected with Lv-sh-C/EBPbeta and treated with LPS, and the expression levels of iNOS, TNF-alpha, IL-6, and IL-1beta were measured by RT-qPCR, Western blotting, and ELISA. Mechanistically, transcriptome sequencing (RNA-seq) revealed changes in gene expression patterns in AMs after LPS stimulation and C/EBPbeta knockdown. Functional enrichment analyses and rescue experiments identified and validated inflammation-associated cell signaling pathways regulated by C/EBPbeta. Furthermore, virtual screening was used to search for natural compounds that inhibit C/EBPbeta with the structure of helenalin as a reference. RESULTS: Following stimulation with LPS, AMs exhibited an increased expression of C/EBPbeta. C/EBPbeta knockdown significantly decreased the expression levels of inflammatory mediators. A total of 374 differentially expressed genes (DEGs) were identified between LPS-stimulated C/EBPbeta knockdown and negative control cells. The NOD-like receptor signaling may be a key target for C/EBPbeta, according to functional enrichment analyses of the DEGs. Further experiments showed that the muramyl dipeptide (MDP, NOD2 agonist) reversed the downregulation of inflammatory mediators and the NF-kappaB pathway caused by the C/EBPbeta knockdown. The virtual screening revealed that N-Caffeoyltryptophan, orilotimod, and petasiphenone have comparable pharmacological properties to helenalin (a known C/EBPbeta inhibitor) and demonstrate a great binding capacity to C/EBPbeta. CONCLUSION: Ablation of C/EBPbeta may attenuate LPS-induced inflammatory damage in AMs by inhibiting the NOD2 receptor signaling pathway. Three natural compounds, N-Caffeoyltryptophan, orilotimod, and petasiphenone, may be potential C/EBPbeta inhibitors.
A Metabolomic Study of the Variability of the Chemical Composition of Commonly Consumed Coffee Brews.[Pubmed:30669279]
Metabolites. 2019 Jan 18;9(1):17.
Coffee drinking has been associated with a lower risk of certain chronic diseases and overall mortality. Its effects on disease risk may vary according to the type of coffee brew consumed and its chemical composition. We characterized variations in the chemical profiles of 76 coffee brew samples representing different brew methods, roast levels, bean species, and caffeine types, either prepared or purchased from outlets in Rockville, Maryland, United States of America. Samples were profiled using liquid chromatography coupled with high-resolution mass spectrometry, and the main sources of chemical variability identified by the principal component partial R-square multivariable regression were found to be brew methods (R(partial)(2) = 36%). A principal component analysis (PCA) was run on 18 identified coffee compounds after normalization for total signal intensity. The three first principal components were driven by roasting intensity (41% variance), type of coffee beans (29%), and caffeine (8%). These variations were mainly explained by hydroxycinnamoyl esters and diketopiperazines (roasting), N-Caffeoyltryptophan, N-p-coumaroyltryptophan, feruloylquinic acids, and theophylline (coffee bean variety) and theobromine (decaffeination). Instant coffees differed from all coffee brews by high contents of diketopiperazines, suggesting a higher roast of the extracted beans. These variations will be important to consider for understanding the effects of different coffee brews on disease risk.
Finding Potent Sirt Inhibitor in Coffee: Isolation, Confirmation and Synthesis of Javamide-II (N-Caffeoyltryptophan) as Sirt1/2 Inhibitor.[Pubmed:26986569]
PLoS One. 2016 Mar 17;11(3):e0150392.
Recent studies suggest that Sirt inhibition may have beneficial effects on several human diseases such as neurodegenerative diseases and cancer. Coffee is one of most popular beverages with several positive health effects. Therefore, in this paper, potential Sirt inhibitors were screened using coffee extract. First, HPLC was utilized to fractionate coffee extract, then screened using a Sirt1/2 inhibition assay. The screening led to the isolation of a potent Sirt1/2 inhibitor, whose structure was determined as javamide-II (N-Caffeoyltryptophan) by NMR. For confirmation, the amide was chemically synthesized and its capacity of inhibiting Sirt1/2 was also compared with the isolated amide. Javamide-II inhibited Sirt2 (IC50; 8.7 muM) better than Sirt1(IC50; 34muM). Since javamide-II is a stronger inhibitor for Sirt2 than Sirt1. The kinetic study was performed against Sirt2. The amide exhibited noncompetitive Sirt2 inhibition against the NAD+ (Ki = 9.8 muM) and showed competitive inhibition against the peptide substrate (Ki = 5.3 muM). Also, a docking simulation showed stronger binding pose of javamide-II to Sirt2 than AGK2. In cellular levels, javamide-II was able to increase the acetylation of total lysine, cortactin and histone H3 in neuronal NG108-15 cells. In the same cells, the amide also increased the acetylation of lysine (K382) in p53, but not (K305). This study suggests that Javamide-II found in coffee may be a potent Sirt1/2 inhibitor, probably with potential use in some conditions of human diseases.


