MPPGGroup III/group II mGlu antagonist. More selective for group III than group II CAS# 169209-65-8 |
- MTEP hydrochloride
Catalog No.:BCC1780
CAS No.:1186195-60-7
- mGlu2 agonist
Catalog No.:BCC1745
CAS No.:1311385-32-6
- LY341495
Catalog No.:BCC1724
CAS No.:201943-63-7
- CPPHA
Catalog No.:BCC1501
CAS No.:693288-97-0
- Dipraglurant
Catalog No.:BCC1531
CAS No.:872363-17-2
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 169209-65-8 | SDF | Download SDF |
| PubChem ID | 3972752 | Appearance | Powder |
| Formula | C9H12NO5P | M.Wt | 245.17 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in 1eq. NaOH | ||
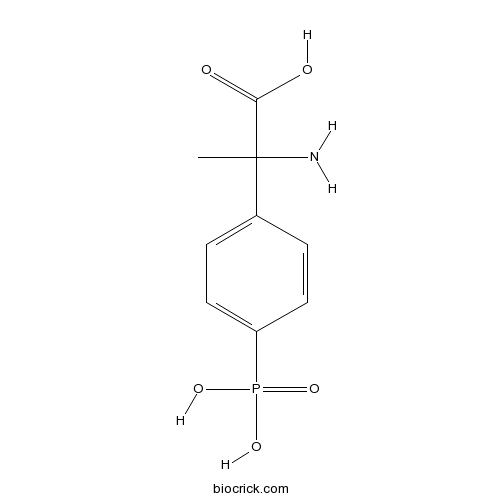
| Chemical Name | 2-amino-2-(4-phosphonophenyl)propanoic acid | ||
| SMILES | CC(C1=CC=C(C=C1)P(=O)(O)O)(C(=O)O)N | ||
| Standard InChIKey | PAONCRJPUQXPRW-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C9H12NO5P/c1-9(10,8(11)12)6-2-4-7(5-3-6)16(13,14)15/h2-5H,10H2,1H3,(H,11,12)(H2,13,14,15) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Potent antagonist of L-AP4-induced effects in rat spinal cord, thalamic and hippocampal neurons, showing selectivity over (1S,3S)-ACPD-induced effects. Potent antagonist of mGlu receptors linked to adenylyl cyclase in adult rat cortical slices. |

MPPG Dilution Calculator

MPPG Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.0788 mL | 20.394 mL | 40.788 mL | 81.576 mL | 101.9701 mL |
| 5 mM | 0.8158 mL | 4.0788 mL | 8.1576 mL | 16.3152 mL | 20.394 mL |
| 10 mM | 0.4079 mL | 2.0394 mL | 4.0788 mL | 8.1576 mL | 10.197 mL |
| 50 mM | 0.0816 mL | 0.4079 mL | 0.8158 mL | 1.6315 mL | 2.0394 mL |
| 100 mM | 0.0408 mL | 0.2039 mL | 0.4079 mL | 0.8158 mL | 1.0197 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- MSPG
Catalog No.:BCC6819
CAS No.:169209-64-7
- (2R,4R)-APDC
Catalog No.:BCC6969
CAS No.:169209-63-6
- 4'-O-Methylcoumestrol
Catalog No.:BCN7226
CAS No.:1690-62-6
- Tropanyl phenylacetate
Catalog No.:BCN1927
CAS No.:1690-22-8
- RS 67506 hydrochloride
Catalog No.:BCC6878
CAS No.:168986-61-6
- RS 67333 hydrochloride
Catalog No.:BCC5693
CAS No.:168986-60-5
- Z-Tyr(tBu)-OH.DCHA
Catalog No.:BCC2745
CAS No.:16879-90-6
- SGC707
Catalog No.:BCC6543
CAS No.:1687736-54-4
- H-Tyr-OtBu
Catalog No.:BCC3128
CAS No.:16874-12-7
- Ezatiostat
Catalog No.:BCC3638
CAS No.:168682-53-9
- Conivaptan HCl
Catalog No.:BCC3756
CAS No.:168626-94-6
- TPEN
Catalog No.:BCC7913
CAS No.:16858-02-9
- MTPG
Catalog No.:BCC6820
CAS No.:169209-66-9
- Trametol
Catalog No.:BCN6924
CAS No.:169217-47-4
- Dihydropedicin
Catalog No.:BCN4692
CAS No.:169234-89-3
- Fmoc-Phe(4-F)-OH
Catalog No.:BCC3220
CAS No.:169243-86-1
- Ro 25-6981
Catalog No.:BCC4158
CAS No.:169274-78-6
- Nebivolol hydrochloride
Catalog No.:BCC9099
CAS No.:169293-50-9
- Boc-D-Orn(Z)-OH
Catalog No.:BCC3431
CAS No.:16937-92-1
- Boc-D-Leu-OH.H2O
Catalog No.:BCC3409
CAS No.:16937-99-8
- 3β,7β,15β-trihydroxy-11-oxo-lanosta-8-en-24->20 lactone
Catalog No.:BCC8643
CAS No.:1694587-15-9
- Boc-N-Me-Ala-OH
Catalog No.:BCC3209
CAS No.:16948-16-6
- RS 17053 hydrochloride
Catalog No.:BCC6874
CAS No.:169505-93-5
- Protostemotinine
Catalog No.:BCN8314
CAS No.:169534-85-4
Asymmetric Strecker-Type Reaction of alpha-Aryl Ketones. Synthesis of (S)-alphaM4CPG, (S)-MPPG, (S)-AIDA, and (S)-APICA, the Antagonists of Metabotropic Glutamate Receptors.[Pubmed:11674092]
J Org Chem. 1999 Jan 8;64(1):120-125.
Heating a mixture of alpha-aryl ketone with (R)-phenylglycinol produces a mixture of imine and 1,3-dioxazolidine. Treatment of this mixture with trimethylsilyl cyanide followed by transformation of nitrile to ester gives Strecker-type reaction products. The diastereoselectivity of the generated alpha-amino esters is from 2/1 to 7/1, and the (R,S)isomer is found as the major product. The (R,S) and (R,R)isomers can be separated by conversion to their N-Cbz or cyclization derivatives. Using this methodology, four antagonists of metabotropic glutamate receptors, (S)-alphaM4CPG, (S)-MPPG, (S)-AIDA, and (S)-APICA, are synthesized.
Implementation of the validation testing in MPPG 5.a "Commissioning and QA of treatment planning dose calculations-megavoltage photon and electron beams".[Pubmed:28291929]
J Appl Clin Med Phys. 2017 Jan;18(1):115-127.
The AAPM Medical Physics Practice Guideline (MPPG) 5.a provides concise guidance on the commissioning and QA of beam modeling and dose calculation in radiotherapy treatment planning systems. This work discusses the implementation of the validation testing recommended in MPPG 5.a at two institutions. The two institutions worked collaboratively to create a common set of treatment fields and analysis tools to deliver and analyze the validation tests. This included the development of a novel, open-source software tool to compare scanning water tank measurements to 3D DICOM-RT Dose distributions. Dose calculation algorithms in both Pinnacle and Eclipse were tested with MPPG 5.a to validate the modeling of Varian TrueBeam linear accelerators. The validation process resulted in more than 200 water tank scans and more than 50 point measurements per institution, each of which was compared to a dose calculation from the institution's treatment planning system (TPS). Overall, the validation testing recommended in MPPG 5.a took approximately 79 person-hours for a machine with four photon and five electron energies for a single TPS. Of the 79 person-hours, 26 person-hours required time on the machine, and the remainder involved preparation and analysis. The basic photon, electron, and heterogeneity correction tests were evaluated with the tolerances in MPPG 5.a, and the tolerances were met for all tests. The MPPG 5.a evaluation criteria were used to assess the small field and IMRT/VMAT validation tests. Both institutions found the use of MPPG 5.a to be a valuable resource during the commissioning process. The validation testing in MPPG 5.a showed the strengths and limitations of the TPS models. In addition, the data collected during the validation testing is useful for routine QA of the TPS, validation of software upgrades, and commissioning of new algorithms.
Antagonism of the presumed presynaptic action of L-AP4 on GABAergic transmission in the ventrobasal thalamus by the novel mGluR antagonist MPPG.[Pubmed:8734494]
Neuropharmacology. 1996 Feb;35(2):239-41.
The metabotropic glutamate receptor (mGluR) agonists CCG-I and L-AP4, acting at Group II and Group III mGluRs respectively, can reduce GABAergic synaptic inhibition on single neurones in the rat thalamus in vivo via a presumed presynaptic mechanism. The actions of L-AP4 were antagonized by (+/-)-alpha-methyl-4-phosphonophenylglycine (MPPG), whereas CCG-I was significantly less affected. Thus MPPG may be a useful tool for detecting physiological roles for Group III mGluRs.
Novel potent selective phenylglycine antagonists of metabotropic glutamate receptors.[Pubmed:8864696]
Eur J Pharmacol. 1996 Aug 1;309(1):71-8.
The metabotropic glutamate (mGlu) receptor antagonist properties of novel phenylglycine analogues were investigated in adult rat cortical slices (mGlu receptors negatively coupled to adenylyl cyclase), neonatal rat cortical slices and in cultured rat cerebellar granule cells (mGlu receptors coupled to phosphoinositide hydrolysis). (RS)-alpha-methyl-4-phosphonophenylglycine (MPPG), (RS)-alpha-methyl-4-sulphonophenylglycine (MSPG), (RS)-alpha-methyl-4-tetrazolylphenylglycine (MTPG), (RS)-alpha-methyl-3-carboxymethyl-4-hydroxyphenylglycine (M3CM4HPG) and (RS)-alpha-methyl-4-hydroxy-3-phosphonomethylphenylglycine (M4H3PMPG) were demonstrated to have potent and selective effects against 10 microM L-2-amino-4-phosphonobutyrate (L-AP4)- and 0.3 microM (2S,1'S,2'S)-2-(2-carboxycyclopropyl)glycine (L-CCG-1)-mediated inhibition of forskolin-stimulated cAMP accumulation in the adult rat cortex. In contrast, these compounds demonstrated either weak or no antagonism at mGlu receptors coupled to phosphoinositide hydrolysis in either neonatal rat cortex or in cultured cerebellar granule cells. These compounds thus appear to be useful discriminatory pharmacological tools for mGlu receptors and form the basis for the further development of novel antagonists.
Pharmacological antagonism of the actions of group II and III mGluR agonists in the lateral perforant path of rat hippocampal slices.[Pubmed:8730739]
Br J Pharmacol. 1996 Apr;117(7):1457-62.
1. An understanding of the physiological and pathological roles of metabotropic glutamate receptors (mGluRs) is currently hampered by the lack of selective antagonists. Standard extracellular recording techniques were used to investigate the activity of recently reported mGluR antagonists on agonist-induced depressions of synaptic transmission in the lateral perforant path of hippocampal slices obtained from 12-16 day-old rats. 2. The group III specific mGluR agonist, (S)-2-amino-4-phosphonobutanoate (L-AP4) depressed basal synaptic transmission in a reversible and dose-dependent manner. The mean (+/-s.e. mean) depression obtained with 100 microM L-AP4 (the maximum concentration tested) was 74 +/- 3% and the IC50 value was 3 +/- 1 microM (n = 5). 3. The selective group II mGluR agonists, (1S,3S)-1-aminocyclopentane-1, 3-dicarboxylate ((1S,3s)-ACPD) and (2S, 1'R, 2'R, 3'R)-2-(2',3'-dicarboxycyclopropyl)glycine (DCG-IV) also depressed basal synaptic transmission in a reversible and dose-dependent manner. The mean depression obtained with 200 microM (1S,3S)-ACPD was 83 +/- 8% and the IC50 value was 12 +/- 3 microM (n = 5). The mean depression obtained with 1 microM DCG-IV was 73 +/- 7% and the IC50 value was 88 +/- 15 nM (n = 4). 4. Synaptic depressions induced by the actions of 20 microM (1S,3S)-ACPD and 10 microM L-AP4 were antagonized by the mGluR antagonists (+)-alpha-methyl-4-carboxyphenylglycine ((+)-MCPG), (S)-2-methyl-2-amino-4-phosphonobutanoate (MAP4), (2S,1'S,2'S)-2-methyl-2(2'-carboxycyclopropyl)glycine (MCCG), (RS)-alpha-methyl-4-tetrazolylphenylglycine (MTPG), (RS)-alpha-methyl-4-sulphonophenylglycine (MSPG) and (RS)-alpha-methyl-4-phosphonophenylglycine (MPPG) (all tested at 500 microM). 5. (+)-MCPG was a weak antagonist of both L-AP4 and (1S,3S)-ACPD-induced depressions. MCCG was selective towards (1S,3S)-ACPD, but analysis of its effects were complicated by apparent partial agonist activity. MAP4 showed good selectivity for L-AP4-induced effects. 6. The most effective antagonist tested against 10 microM L-AP4 was MPPG (mean reversal 90 +/- 3%; n = 4). In contrast, the most effective antagonist tested against 20 microM (1S,3S)-ACPD induced depressions was MTPG (mean reversal 64 +/- 4%; n = 4). Both antagonists produced parallel shifts in agonist dose-response curves. Schild analysis yielded estimated KD values of 11.7 microM and 27.5 microM, respectively. Neither antagonist had any effect on basal transmission or on depressions induced by the adenosine receptor agonist, 2-chloroadenosine (500 nM; n = 3). 7. We conclude that both group II and group III mGluRs can mediate synaptic depressions induced by mGluR agonists in the lateral perforant path. The mGlur antagonists MTPG, MPPG and MAP4 should be useful in determining the roles of group II and III mGluRs in the central nervous system.
New phenylglycine derivatives with potent and selective antagonist activity at presynaptic glutamate receptors in neonatal rat spinal cord.[Pubmed:8532166]
Neuropharmacology. 1995 Aug;34(8):851-6.
The depression of the monosynaptic excitation of neonatal rat motoneurones produced by the metabotropic glutamate receptor (mGluR) agonists (1S,3S)-1-aminocyclopentane-1, 3-dicarboxylate (ACPD) or L-2-amino-4-phosphonobutyrate (L-AP4) was antagonized by three novel phenylglycine analogues: (RS)-alpha-methyl-4-sulphonophenylglycine (MSPG), (RS)-alpha-methyl-4-phosphonophenylglycine (MPPG) and (RS)-alpha-methyl-4-tetrazolylphenylglycine (MTPG). The potencies of all the new compounds were greater than that of the previously reported (RS)-alpha-methyl-4-carboxyphenylglycine (MCPG). For L-AP4-sensitive presynaptic mGluRs, the order of antagonist potency found was MPPG > MSPG > MTPG > MCPG. In contrast, the order of antagonist potency found for (1S,3S)-ACPD-sensitive presynaptic mGluRs was MTPG > MPPG > MSPG > MCPG. To date, MPPG (KD 9.2 microM) is the most potent L-AP4-sensitive receptor antagonist yet tested on the neonatal rat spinal cord. In addition, MTPG (KD 77 microM) is the most potent antagonist yet tested for (1S,3S)-ACPD-sensitive receptors in this preparation.


