L-741,626High affinity D2 antagonist CAS# 81226-60-0 |
- BMS-708163 (Avagacestat)
Catalog No.:BCC2104
CAS No.:1146699-66-2
- DAPT (GSI-IX)
Catalog No.:BCC3618
CAS No.:208255-80-5
- YO-01027 (Dibenzazepine, DBZ)
Catalog No.:BCC2100
CAS No.:209984-56-5
- Semagacestat (LY450139)
Catalog No.:BCC3610
CAS No.:425386-60-3
- Flurizan
Catalog No.:BCC2342
CAS No.:51543-40-9
- E 2012
Catalog No.:BCC1540
CAS No.:870843-42-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 81226-60-0 | SDF | Download SDF |
| PubChem ID | 133633 | Appearance | Powder |
| Formula | C20H21ClN2O | M.Wt | 340.85 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in DMSO and to 100 mM in ethanol | ||
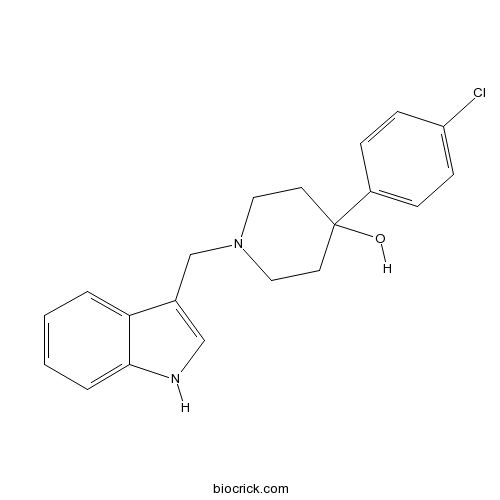
| Chemical Name | 4-(4-chlorophenyl)-1-(1H-indol-3-ylmethyl)piperidin-4-ol | ||
| SMILES | C1CN(CCC1(C2=CC=C(C=C2)Cl)O)CC3=CNC4=CC=CC=C43 | ||
| Standard InChIKey | LLBLNMUONVVVPG-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H21ClN2O/c21-17-7-5-16(6-8-17)20(24)9-11-23(12-10-20)14-15-13-22-19-4-2-1-3-18(15)19/h1-8,13,22,24H,9-12,14H2 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | A potent D2 dopamine receptor selective antagonist, with affinities of 2.4, 100 and 220 nM for D2, D3 and D4 receptors respectively. Centrally active following systemic administration in vivo. |

L-741,626 Dilution Calculator

L-741,626 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.9338 mL | 14.6692 mL | 29.3384 mL | 58.6768 mL | 73.346 mL |
| 5 mM | 0.5868 mL | 2.9338 mL | 5.8677 mL | 11.7354 mL | 14.6692 mL |
| 10 mM | 0.2934 mL | 1.4669 mL | 2.9338 mL | 5.8677 mL | 7.3346 mL |
| 50 mM | 0.0587 mL | 0.2934 mL | 0.5868 mL | 1.1735 mL | 1.4669 mL |
| 100 mM | 0.0293 mL | 0.1467 mL | 0.2934 mL | 0.5868 mL | 0.7335 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Panaxynol
Catalog No.:BCN3833
CAS No.:81203-57-8
- Boc-D-Tyr(2-Br-Z)-OH
Catalog No.:BCC3464
CAS No.:81189-61-9
- Apatinib
Catalog No.:BCC5099
CAS No.:811803-05-1
- Imiloxan hydrochloride
Catalog No.:BCC6875
CAS No.:81167-22-8
- RR-src
Catalog No.:BCC6956
CAS No.:81156-93-6
- Pravastatin sodium
Catalog No.:BCC2321
CAS No.:81131-70-6
- Cilastatin sodium
Catalog No.:BCC7457
CAS No.:81129-83-1
- (Z)-Lachnophyllum lactone
Catalog No.:BCN4746
CAS No.:81122-95-4
- N-Nonyldeoxynojirimycin
Catalog No.:BCC7752
CAS No.:81117-35-3
- Racecadotril
Catalog No.:BCC4614
CAS No.:81110-73-8
- Clarithromycin
Catalog No.:BCC9219
CAS No.:81103-11-9
- Cisapride
Catalog No.:BCC4207
CAS No.:81098-60-4
- 15-Deoxoeucosterol
Catalog No.:BCN4348
CAS No.:81241-53-4
- ent-9-Hydroxy-15-oxo-16-kauren-19-oic acid beta-D-glucopyranosyl ester
Catalog No.:BCN1345
CAS No.:81263-96-9
- ent-6,11-Dihydroxy-15-oxo-16-kauren-19-oic acid beta-D-glucopyranosyl ester
Catalog No.:BCN1344
CAS No.:81263-97-0
- ent-6,9-Dihydroxy-15-oxo-16-kauren-19-oic acid beta-D-glucopyranosyl ester
Catalog No.:BCN1343
CAS No.:81263-98-1
- ent-6,9-Dihydroxy-15-oxo-16-kauren-19-oic acid
Catalog No.:BCN1342
CAS No.:81264-00-8
- D-AP7
Catalog No.:BCC6559
CAS No.:81338-23-0
- Amisulpride hydrochloride
Catalog No.:BCC4252
CAS No.:81342-13-4
- Schisandrin C epoxide
Catalog No.:BCN3744
CAS No.:81345-36-0
- Cyclo(Tyr-Hpro)
Catalog No.:BCN2424
CAS No.:813461-21-1
- Momordicoside F1
Catalog No.:BCN3273
CAS No.:81348-81-4
- Momordicoside L
Catalog No.:BCN3274
CAS No.:81348-83-6
- Momordicoside K
Catalog No.:BCN3272
CAS No.:81348-84-7
Human D2 and D4 dopamine receptors couple through betagamma G-protein subunits to inwardly rectifying K+ channels (GIRK1) in a Xenopus oocyte expression system: selective antagonism by L-741,626 and L-745,870 respectively.[Pubmed:9833627]
Neuropharmacology. 1998 Aug;37(8):983-7.
To examine the effects of a novel selective D4 receptor ligand, L-745,870 (3-[4-(4-chlorophenyl)piperazin-1-yl]methyl-1H-pyrrolo[2,3-b]pyrid ine), on human dopamine receptor function, the ability of this ligand to antagonise G-protein gated inwardly rectifying K+ (GIRK/Kir3) currents activated by cloned human D2 and D4 receptors expressed in Xenopus oocytes was examined using voltage-clamp recording. Its effects were also contrasted with that of a selective D2 receptor antagonist L-741,626. L-745,870 had no detectable agonist activity on human D4 receptors and selectively blocked currents activated by D4 but not D2 receptors. The role of G-protein subunits in dopamine receptor modulation of GIRK currents was also examined by co-expression of beta1 and/or gamma2 subunits on spontaneously active and receptor-activated currents. Currents activated by both D2 and D4 receptors were occluded by direct activation of GIRK currents following co-transfection with the cDNA encoding G-protein betagamma subunits. These data demonstrate that L-745,870 and L-741,626 act as antagonists on human D4 and D2 receptors respectively, and that activation of GIRK channels by these dopamine receptors can be disrupted by direct stimulation of K+ currents by G-protein betagamma subunits.
Antagonism of the effects of (+)-PD 128907 on midbrain dopamine neurones in rat brain slices by a selective D2 receptor antagonist L-741,626.[Pubmed:8968560]
Br J Pharmacol. 1996 Dec;119(7):1491-7.
1. The ability of PD 128907 to activate dopamine receptors in the ventral tegmental area, substantia nigra pars compacta, and striatum was investigated by use of in vitro electrophysiological recording and fast cyclic voltammetry. The affinity of a novel D2 selective antagonist L-741,626 for receptors activated by this agonist was measured to determine if its effects were mediated by D2 or D3 receptors. 2. The active (+) enantiomer of PD 128907 bound with high affinity and selectivity to rat D3 dopamine receptors. The Ki values for (+)-PD 128907 were 620 nM at D2, 1 nM at D3 and 720 nM at D4 receptors. 3. (+)-PD 128907 inhibited cell firing in both the ventral tegmental area and substantia nigra pars compacta with EC50 values of 33 nM (pEC50 = 7.48 +/- 0.10, n = 10) and 38 nM (pEC50 = 7.42 +/- 0.15, n = 5), respectively. No effects of (+)-PD 128907 (100 nM) were observed on glutamate or GABA-mediated synaptic potentials elicited by focal bipolar stimulation. 4. L-741,626 antagonized these effects of (+)-PD 128907 in a concentration-dependent and surmountable manner with an affinity, determined from Schild analysis, of 20 nM (pKB = 7.71 +/- 0.14) in the ventral tegmental area and 11 nM (pKB = 7.95 +/- 0.18) in the substantia nigra pars compacta. 5. (+)-PD 128907 also inhibited dopamine release in the caudate-putamen with an EC50 of 66 nM (n = 5). The affinity of L-741,626 for these nerve terminal autoreceptors (pKB = 7.71 +/- 0.06; = 20 nM) was identical to that observed on midbrain dopamine neurones. 6. These data demonstrate that the D3 receptor ligand (+)-PD 128907 is a potent agonist on rat midbrain dopamine neurones. However, its lack of regional selectivity, and the high affinity of the selective D2 receptor antagonist L-741,626 for receptors activated by (+)-PD 128907, was more consistent with an action on D2 autoreceptors rather than upon a D3 dopamine receptor subtype.
Synthesis and characterization of selective dopamine D2 receptor antagonists. 2. Azaindole, benzofuran, and benzothiophene analogs of L-741,626.[Pubmed:20542439]
Bioorg Med Chem. 2010 Jul 15;18(14):5291-300.
A series of indole, 7-azaindole, benzofuran, and benzothiophene compounds have been prepared and evaluated for affinity at D2-like dopamine receptors. These compounds share structural elements with the classical D2-like dopamine receptor antagonists haloperidol, N-methylspiperone and benperidol. Two new compounds, 4-(4-iodophenyl)-1-((4-methoxy-1H-indol-3-yl)methyl)piperidin-4-ol (6) and 4-(4-iodophenyl)-1-((5-methoxy-1H-indol-3-yl)methyl)piperidin-4-ol (7), were found to have high affinity to and selectivity for D2 versus D3 receptors. Changing the aromatic ring system from an indole to other heteroaromatic ring systems reduced the D2 binding affinity and the D2 versus D3 selectivity.
S33084, a novel, potent, selective, and competitive antagonist at dopamine D(3)-receptors: II. Functional and behavioral profile compared with GR218,231 and L741,626.[Pubmed:10869411]
J Pharmacol Exp Ther. 2000 Jun;293(3):1063-73.
The selective dopamine D(3)-receptor antagonist S33084 dose dependently attenuated induction of hypothermia by 7-hydroxy-2-dipropylaminotetralin (7-OH-DPAT) and PD128,907. S33084 also dose dependently reduced 7-OH-DPAT-induced penile erections (PEs) but had little effect on 7-OH-DPAT-induced yawning and hypophagia, and it did not block contralateral rotation elicited by the preferential D(3) agonist quinpirole in unilateral substantia nigra-lesioned rats. In models of potential antipsychotic activity, S33084 had little effect on conditioned avoidance behavior and the locomotor response to amphetamine and cocaine in rats, and weakly inhibited apomorphine-induced climbing in mice. Moreover, S33084 was inactive in models of potential extrapyramidal activity in rats: induction of catalepsy and prolactin secretion and inhibition of methylphenidate-induced gnawing. Another selective D(3) antagonist, GR218,231, mimicked S33084 in inhibiting 7-OH-DPAT-induced PEs and hypothermia but neither hypophagia nor yawning behavior. Similarly, it was inactive in models of potential antipsychotic and extrapyramidal activity. In distinction to S33084 and GR218,231, the preferential D(2) antagonist L741,626 inhibited all responses elicited by 7-OH-DPAT. Furthermore, it displayed robust activity in models of antipsychotic and, at slightly higher doses, extrapyramidal activity. In summary, S33084 was inactive in models of potential antipsychotic and extrapyramidal activity and failed to modify spontaneous locomotor behavior. Furthermore, it did not affect hypophagia or yawns, but attenuated hypothermia and PEs, elicited by 7-OH-DPAT. This profile was shared by GR218,231, whereas L741,626 was effective in all models. Thus, D(2)-receptors are principally involved in these paradigms, although D(3)-receptors may contribute to induction of hypothermia and PEs. S33084 should comprise a useful tool for further exploration of the pathophysiological significance of D(3)- versus D(2)-receptors.


