L-651,582Ca2+ channel blocker CAS# 99519-84-3 |
- GW1929
Catalog No.:BCC1611
CAS No.:196808-24-9
- Balaglitazone
Catalog No.:BCC1395
CAS No.:199113-98-9
- Inolitazone
Catalog No.:BCC1652
CAS No.:223132-37-4
- Inolitazone dihydrochloride
Catalog No.:BCC1653
CAS No.:223132-38-5
- Aleglitazar
Catalog No.:BCC1337
CAS No.:475479-34-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 99519-84-3 | SDF | Download SDF |
| PubChem ID | 108144 | Appearance | Powder |
| Formula | C17H12Cl3N5O2 | M.Wt | 424.67 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | Carboxyamidotriazole | ||
| Solubility | Soluble to 100 mM in DMSO | ||
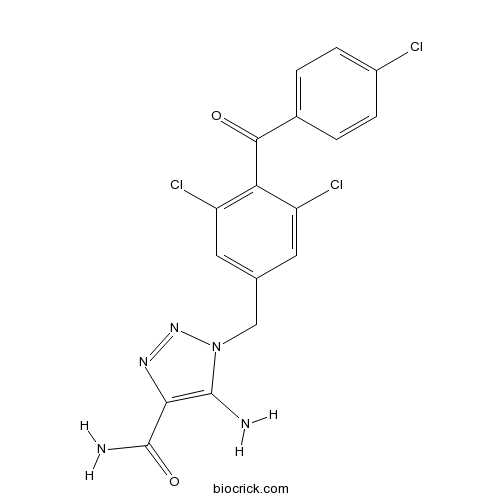
| Chemical Name | 5-amino-1-[[3,5-dichloro-4-(4-chlorobenzoyl)phenyl]methyl]triazole-4-carboxamide | ||
| SMILES | C1=CC(=CC=C1C(=O)C2=C(C=C(C=C2Cl)CN3C(=C(N=N3)C(=O)N)N)Cl)Cl | ||
| Standard InChIKey | WNRZHQBJSXRYJK-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C17H12Cl3N5O2/c18-10-3-1-9(2-4-10)15(26)13-11(19)5-8(6-12(13)20)7-25-16(21)14(17(22)27)23-24-25/h1-6H,7,21H2,(H2,22,27) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Orally active Ca2+ channel blocker; inhibits K+- and carbachol-stimulated Ca2+ influx (IC50 values are 500 and 935 nM respectively). Exhibits antiproliferative, antiangiogenic and antimetastatic activity in vivo and displays selectivity towards numerous mismatch repair-deficient tumor cell lines in vitro. |

L-651,582 Dilution Calculator

L-651,582 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.3548 mL | 11.7738 mL | 23.5477 mL | 47.0954 mL | 58.8692 mL |
| 5 mM | 0.471 mL | 2.3548 mL | 4.7095 mL | 9.4191 mL | 11.7738 mL |
| 10 mM | 0.2355 mL | 1.1774 mL | 2.3548 mL | 4.7095 mL | 5.8869 mL |
| 50 mM | 0.0471 mL | 0.2355 mL | 0.471 mL | 0.9419 mL | 1.1774 mL |
| 100 mM | 0.0235 mL | 0.1177 mL | 0.2355 mL | 0.471 mL | 0.5887 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 3-Ethoxy-4-ethoxycarbonyl phenylacetic acid
Catalog No.:BCC8629
CAS No.:99469-99-5
- 1-Chloroethyl cyclohexyl carbonate
Catalog No.:BCC8463
CAS No.:99464-83-2
- Ampiroxicam
Catalog No.:BCC4426
CAS No.:99464-64-9
- BTZO 1
Catalog No.:BCC7886
CAS No.:99420-15-2
- Methyl rosmarinate
Catalog No.:BCN4536
CAS No.:99353-00-1
- Kadsurin A
Catalog No.:BCN6515
CAS No.:99340-07-5
- Venlafaxine Hydrochloride
Catalog No.:BCC2513
CAS No.:99300-78-4
- Levodropropizine
Catalog No.:BCC4520
CAS No.:99291-25-5
- Proglumide sodium salt
Catalog No.:BCC5768
CAS No.:99247-33-3
- Mulberrofuran G pentaacetate
Catalog No.:BCN6518
CAS No.:99217-75-1
- Broussoflavonol B
Catalog No.:BCN3679
CAS No.:99217-70-6
- Caesalpin J
Catalog No.:BCC8305
CAS No.:99217-67-1
- K 252a
Catalog No.:BCC7152
CAS No.:99533-80-9
- Neuropeptide FF
Catalog No.:BCC5983
CAS No.:99566-27-5
- Sertaconazole
Catalog No.:BCC9146
CAS No.:99592-32-2
- Sertaconazole nitrate
Catalog No.:BCC4716
CAS No.:99592-39-9
- Ondansetron HCl
Catalog No.:BCC2493
CAS No.:99614-01-4
- Ondansetron
Catalog No.:BCC5043
CAS No.:99614-02-5
- Leucanthogenin
Catalog No.:BCN7932
CAS No.:99615-00-6
- Isothymonin
Catalog No.:BCN3393
CAS No.:99615-01-7
- Kazinol B
Catalog No.:BCN4538
CAS No.:99624-27-8
- Kazinol A
Catalog No.:BCN3388
CAS No.:99624-28-9
- ent-3beta,18-Dihydroxylabda-8(17),13E-dien-15-oic acid
Catalog No.:BCN7669
CAS No.:99624-39-2
- Uncinatone
Catalog No.:BCN4547
CAS No.:99624-92-7
Anti-proliferative activity of L-651,582 correlates with calcium-mediated regulation of nucleotide metabolism at phosphoribosyl pyrophosphate synthetase.[Pubmed:2167901]
J Cell Physiol. 1990 Sep;144(3):457-66.
L-651,582, 5-amino-[4-(4-chlorobenzoyl)-3,5-dichlorobenzyl]-1, 2,3-triazole-4-carboxamide, is an antiproliferative and antiparasitic agent which inhibits nucleotide metabolism in mammalian cells. The drug equivalently inhibited 3H-hypoxanthine, 14C-adenine, and 14C-formate incorporation into nucleotide pools in Madin-Darby bovine kidney (MDBK) cells, suggesting depletion of the supply of phosphoribosyl pyrophosphate, (PRPP), required for each of these independent pathways. Inhibition of nucleotide metabolism correlated with inhibition of proliferation for three cell types with differing sensitivities toward the drug. L-651,582 inhibited incorporation of 3H-hypoxanthine into nucleotide pools with either glucose, uridine, or ribose as carbon source suggesting a block at PRPP synthetase, rather than a block in a pathway supplying ribose-5-phosphate. PRPP synthetase was not inhibited directly by the compound, indicating regulation of the enzyme in intact cells. Drug treatment did not kill cells but reduced the fraction of cells in S and G2/M while increasing the population in G1. Inhibition of uptake of 45Ca was demonstrated at concentrations identical to those required for inhibition of nucleotide metabolism or proliferation. Inhibition of cellular PRPP biosynthesis rates were also observed using EGTA to lower calcium levels. These data suggest a previously unrecognized link between calcium entry, the regulation of nucleotide biosynthesis at PRPP synthetase, and the rate of proliferation of mammalian cells.
L-651,582 inhibition of intracellular parasitic protozoal growth correlates with host-cell directed effects.[Pubmed:1993990]
J Pharmacol Exp Ther. 1991 Feb;256(2):462-7.
L-651,582 (5-amino-[4-(4-chlorobenzoyl)-3,5-dichlorobenzyl]-1,2,3-triazole-4- carboxamide), which is active in vivo against coccidiosis caused by the intracellular parasitic protozoan Eimeria tenella, is also effective against this organism in tissue culture in Maden Darby bovine kidney (MDBK) host cells. L-651,582 inhibited 45Ca++ uptake, as well as [3H]hypoxanthine incorporation into soluble nucleotide pools, in MDBK cells at concentrations similar to those required for antiparasitic activity (IC50 = 0.3 microgram/ml). However, the drug did not inhibit the [3H] hypoxanthine incorporation into the nucleotide pools of the intracellular E. tenella under the same conditions. The antiparasitic activity of several L-651,582 analogs paralleled their ability to inhibit [3H]hypoxanthine incorporation in MDBK cells. L-651,582 similarly inhibited the growth of the intracellular parasitic protozoan Toxoplasma gondii in vitro with either HeLa cells or Normal Human Fibroblasts as host cells. The IC50 for inhibition of T. gondii growth in normal human fibroblasts was similar to that for [3H]hypoxanthine incorporation inhibition. In HeLa cells, however, 40-fold higher levels were required for the inhibition of [3H]hypoxanthine incorporation than were required to inhibit parasite growth, showing that antiparasitic activity was not a consequence of the alteration in host nucleotide metabolism. This was also consistent with the observation that the effect of L-651,582 against E. tenella in MDBK cells was not reversed by the addition of nutrients involved in nucleotide biosynthesis. This study suggests that L-651,582 has antiparasitic activity which correlates with host cell effects and which may be a result of blocking Ca++ entry in the host cells, but is not the result of an alteration of host nucleotide biosynthesis.
The inhibition of receptor-mediated and voltage-dependent calcium entry by the antiproliferative L-651,582.[Pubmed:1645340]
J Biol Chem. 1991 Jun 5;266(16):10136-42.
L-651,582, 5-amino-[4-(4-chlorobenzoyl)-3,5-dichlorobenzyl]-1,2,3-triazole-4- carboxamide, an antiproliferative and antiparasitic agent previously shown to affect 45Ca2+ uptake into mammalian cells, inhibits both receptor-mediated and voltage-dependent calcium entry in well characterized in vitro systems. Indo 1 fluorescence measurements of cytosolic calcium levels indicate that the drug has no effect on the initial transient release of internal stores of calcium stimulated by fMet-Leu-Phe in rat polymorphonuclear leukocytes. It does decrease the levels maintained subsequently, however, indicating blockage of calcium influx through receptor-operated channels. L-651,582 also blocks the stimulation of leukotriene B4 (LTB4) production by fMet-Leu-Phe with an IC50 = 0.5 micrograms/ml equal to that for calcium entry inhibition. The LTB4 inhibition is likely due to calcium entry inhibition since L-651,582 does not inhibit calmodulin or enzymes producing arachidonate metabolites. L-651,582 also inhibits potassium-stimulated 45Ca2+ influx into GH3 cells with an IC50 of 0.5 microgram/ml, indicating a block of voltage-gated L-type calcium channels. Patch voltage clamp measurements of current through L- and T-type calcium in guinea pig atrial cells also indicate that L-651,582 is a calcium antagonist. Block of L-type calcium channels is voltage-dependent, and the apparent dissociation constant for the high affinity state is 0.2 micrograms/ml. The IC50 for block of T-type calcium channels is 1.4 micrograms/ml. The inhibition of cellular proliferation and the production of arachidonate metabolites by L-651,582 may be the result of the nearly equipotent block of receptor-operated and voltage-gated calcium channels.
Selective sensitivity to carboxyamidotriazole by human tumor cell lines with DNA mismatch repair deficiency.[Pubmed:18464258]
Int J Cancer. 2008 Jul 15;123(2):258-263.
We have previously reported that high-dose nifedipine had a selective antiproliferative effect on colon cancer cell lines deficient in DNA mismatch repair (MMR). We hypothesized that carboxyamidotriazole (CAI), a calcium channel blocker, would also have a selective inhibitory effect on colon cancer cell lines with DNA MMR deficiency. In addition, we speculated that this effect may also be seen in cell lines deficient in DNA MMR derived from other tumor types. Fourteen human cancer cell lines with and without DNA MMR derived from carcinomas of the colon, bladder, ovary and prostate were treated with CAI, vehicle or control drugs (nifedipine and 5-flurouracil). The effect of treatment on growth inhibition, invasion, apoptosis and cell cycle progression was assessed. Selective sensitivity to CAI was observed in all cancer cell lines deficient in MMR. Compared with the MMR-proficient cells, the matched deficient cells were significantly more sensitive to the growth inhibitory effect of CAI and nifedipine, but less sensitive to 5-flurouracil. CAI significantly inhibited the invasive ability of MMR-deficient cancer cells compared to 5-flurouracil. CAI induced more apoptosis but similar level of G(2)/M arrest in MMR (hMLH1- or hMSH6-)-deficient colon cancer cells than MMR-proficient counterparts. CAI selectively inhibits proliferation and invasion in MMR-deficient human cancer cell lines. The antitumor effect is at least partly explained by G2/M cell cycle arrest and induction of apoptosis. These findings may have clinical implications directing clinical trials in selectively targeted patients with DNA MMR tumors.


