Geissoschizine methyl etherCAS# 60314-89-8 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 60314-89-8 | SDF | Download SDF |
| PubChem ID | 6443046 | Appearance | Powder |
| Formula | C22H26N2O3 | M.Wt | 366.45 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
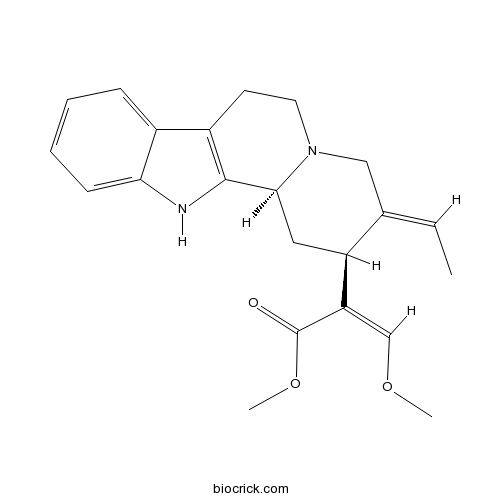
| Chemical Name | methyl (Z)-2-[(2S,3E,12bS)-3-ethylidene-2,4,6,7,12,12b-hexahydro-1H-indolo[2,3-a]quinolizin-2-yl]-3-methoxyprop-2-enoate | ||
| SMILES | CC=C1CN2CCC3=C(C2CC1C(=COC)C(=O)OC)NC4=CC=CC=C34 | ||
| Standard InChIKey | VAMJZLUOKJRHOW-XEASWFAXSA-N | ||
| Standard InChI | InChI=1S/C22H26N2O3/c1-4-14-12-24-10-9-16-15-7-5-6-8-19(15)23-21(16)20(24)11-17(14)18(13-26-2)22(25)27-3/h4-8,13,17,20,23H,9-12H2,1-3H3/b14-4-,18-13-/t17-,20-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Geissoschizine methyl ether is a potent serotonin ₁A receptor agonist and candidate for amelioration of aggressiveness and sociality by yokukansan. 2. Geissoschizine methyl ether has vasorelaxant effect, which is composed of two different mechanisms: endothelial dependency with nitric oxide and endothelial independency with voltage-dependent Ca(2+)-channel blocking. 3. Geissoschizine methyl ether has third-generation antipsychotic-like actions at the dopamine and serotonin receptors. 4. Geissoschizine methyl ether is a strong acetylcholinesterase (AChE) inhibitor, it inhibited 50% of AChE activity at concentrations of 3.7 ± 0.3 ug/mL. 5. Geissoschizine methyl ether protects neurons from oxidative toxicity by shifting energy metabolism from mitochondrial respiration to glycolysis. |
| Targets | 5-HT Receptor | Calcium Channel | Potassium Channel | ROS | NADPH-oxidase | AChR |

Geissoschizine methyl ether Dilution Calculator

Geissoschizine methyl ether Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.7289 mL | 13.6444 mL | 27.2889 mL | 54.5777 mL | 68.2221 mL |
| 5 mM | 0.5458 mL | 2.7289 mL | 5.4578 mL | 10.9155 mL | 13.6444 mL |
| 10 mM | 0.2729 mL | 1.3644 mL | 2.7289 mL | 5.4578 mL | 6.8222 mL |
| 50 mM | 0.0546 mL | 0.2729 mL | 0.5458 mL | 1.0916 mL | 1.3644 mL |
| 100 mM | 0.0273 mL | 0.1364 mL | 0.2729 mL | 0.5458 mL | 0.6822 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Odanacatib (MK-0822)
Catalog No.:BCC1197
CAS No.:603139-19-1
- Sulforhodamine 101
Catalog No.:BCC8019
CAS No.:60311-02-6
- Tamarixetin
Catalog No.:BCN4116
CAS No.:603-61-2
- Chrysosplenetin
Catalog No.:BCN4115
CAS No.:603-56-5
- Auriculasin
Catalog No.:BCN3970
CAS No.:60297-37-2
- Fulvine
Catalog No.:BCN2082
CAS No.:6029-87-4
- Latifoline
Catalog No.:BCN1978
CAS No.:6029-86-3
- Rinderine
Catalog No.:BCN1971
CAS No.:6029-84-1
- 7-Angeloylretronecine
Catalog No.:BCN2036
CAS No.:6029-82-9
- Gestodene
Catalog No.:BCC4490
CAS No.:60282-87-3
- Guvacine hydrochloride
Catalog No.:BCC6574
CAS No.:6027-91-4
- Guanosine-2'(3')-monophosphate disodium salt
Catalog No.:BCC3608
CAS No.:6027-83-4
- NBI 35965 hydrochloride
Catalog No.:BCC7567
CAS No.:603151-83-3
- 4-Methoxyphenyl beta-D-glucopyranoside
Catalog No.:BCN1403
CAS No.:6032-32-2
- Sulprostone
Catalog No.:BCC7547
CAS No.:60325-46-4
- GSK-3 inhibitor 1
Catalog No.:BCC4126
CAS No.:603272-51-1
- LY2090314
Catalog No.:BCC1717
CAS No.:603288-22-8
- 9-O-Feruloyllariciresinol
Catalog No.:BCN4112
CAS No.:60337-67-9
- Leucovorin Calcium
Catalog No.:BCC1198
CAS No.:6035-45-6
- 6,7,8-Trimethoxycoumarin
Catalog No.:BCN4113
CAS No.:6035-49-0
- Doronine
Catalog No.:BCN2106
CAS No.:60367-00-2
- 7,8-Benzoflavone
Catalog No.:BCN6538
CAS No.:604-59-1
- Narcissoside
Catalog No.:BCN1263
CAS No.:604-80-8
- Tombozine
Catalog No.:BCN4117
CAS No.:604-99-9
Geissoschizine methyl ether, a corynanthean-type indole alkaloid from Uncaria rhynchophylla as a potential acetylcholinesterase inhibitor.[Pubmed:21714741]
Nat Prod Res. 2012;26(1):22-8.
Geissoschizine methyl ether (1), a newly discovered strong acetylcholinesterase (AChE) inhibitor, along with six weakly active alkaloids, vallesiachotamine (2), hisuteine (3), hirsutine (4), isorhynchophylline (5), cisocorynoxeine (6) and corynoxeine (7) have been isolated from Uncaria rhynchophylla. Geissoschizine methyl ether (1) inhibited 50% of AChE activity at concentrations of 3.7 +/- 0.3 microg mL(-1) while the IC(50) value of physostigmine as a standard was 0.013 +/- 0.002 microg mL(-1). The mode of AChE inhibition by 1 was reversible and non-competitive. In addition, molecular modelling was performed to explore the binding mode of inhibitor 1 at the active site of AChE.
Geissoschizine methyl ether protects oxidative stress-mediated cytotoxicity in neurons through the 'Neuronal Warburg Effect'.[Pubmed:27114061]
J Ethnopharmacol. 2016 Jul 1;187:249-58.
ETHNOPHARMACOLOGICAL RELEVANCE: The rate of production of reactive oxygen species (ROS) is determined by mitochondrial metabolic rate. In turn, excessive ROS damage mitochondrial function, which is linked to aging and neurodegenerative conditions. One possible path to prevent oxidative stress could be achieved by reducing mitochondrial respiration in favor of less efficient ATP production via glycolysis. Such a shift in energy metabolism is known as the 'Warburg effect'. Geissoschizine methyl ether (GM) is one of the active components responsible for the psychotropic effects of Yokukansan, an herbal preparation widely used in China and Japan. AIM OF THE STUDY: GM protects neurons from glutamate-induced oxidative cytotoxicity through regulating mitochondrial function and suppressing ROS generation. We investigated the protective mechanism of GM against glutamate-induced oxidative stress in neuronal cells. MATERIALS AND METHODS: The current study was performed on primary neurons and HT22 cells, a hippocampus neuronal cell line. Cell viability was measured by Calcein AM assay. H2DCFDA staining was used for intracellular ROS measurement. GSH level was measured using the GSH-Glo luminescence-based assay. Mitochondrial respiration and glycolysis were measured by the Seahorse Bioscience XFe 96 Extracellular Flux Analyzer. Protein levels were analyzed by western blot analysis. RESULTS: GM prevented glutamate-induced cytotoxicity in an HT-22 neuronal cell line even with a 9-hour exposure delay. GM blocked glutamate-induced intracellular ROS accumulation through suppressing mitochondrial respiration. Further, we found that GM up-regulated glycolysis and the pentose-phosphate pathway, which is involved in the production of intracellular reducing agent, NADPH. In addition, GM protected primary cortical neurons from both glutamate and buthioninesulfoximine toxicity. CONCLUSION: GM prevents glutamate-induced oxidative damage through reducing mitochondrial respiration, which further suppresses ROS generation. In addition, GM up-regulates glycolysis which compensate for the energy depletion induced by mitochondrial respiration inhibition. Overall, our study is the first to report that GM protects neurons from oxidative toxicity by shifting energy metabolism from mitochondrial respiration to glycolysis.
Geissoschizine methyl ether, an indole alkaloid extracted from Uncariae Ramulus et Uncus, is a potent vasorelaxant of isolated rat aorta.[Pubmed:12063078]
Eur J Pharmacol. 2002 May 31;444(3):183-9.
Effects of Geissoschizine methyl ether, an indole alkaloid isolated from the hook of Uncariae Ramulus et Uncus, on vascular responses were examined using isolated strips of rat aorta. Geissoschizine methyl ether (10(-7)-10(-4) M) relaxed norepinephrine (5x10(-8) M)-induced contraction in a dose-dependent manner. The potency (50% efficacy concentration, EC(50)=0.744 microM) was approximately 14 times greater than that (EC(50)=10.6 microM) of hirsutine, one of the indole alkaloids isolated from Uncariae Ramulus et Uncus that demonstrates a vasorelaxant effect by Ca(2+)-channel blocking. The vasorelaxant effect of Geissoschizine methyl ether found at the lower concentrations (10(-7)-3x10(-6) M) on the norepinephrine-induced contraction was abolished by pretreatment with N(G)-nitro-L-arginine (10(-4) M), an inhibitor of nitric oxide synthesis, or by denuding aortas of endothelium, while the effects at the higher concentrations (10(-5)-10(-4) M) were not completely prevented by either N(G)-nitro-L-arginine and deendothelialization. Furthermore, Geissoschizine methyl ether did not relax high K(+)-, Ca(2+)- and a Ca(2+)-channel agonist Bay K8644-induced contractions at the lower concentrations that markedly relaxed the norepinephrine-induced contractions, while the higher concentrations of Geissoschizine methyl ether relaxed the high K(+)-, Ca(2+)- and Bay K8644-induced contractions. These results suggest that the vasorelaxant effect of Geissoschizine methyl ether is composed of two different mechanisms: endothelial dependency with nitric oxide and endothelial independency with voltage-dependent Ca(2+)-channel blocking.
Geissoschizine methyl ether, an alkaloid in Uncaria hook, is a potent serotonin (1)A receptor agonist and candidate for amelioration of aggressiveness and sociality by yokukansan.[Pubmed:22314317]
Neuroscience. 2012 Apr 5;207:124-36.
Yokukansan (YKS), a traditional Japanese medicine, is composed of seven kinds of dried herbs. It is widely prescribed in clinical situation for treating psychiatric disorders such as aggressiveness in patients with dementia. We previously demonstrated that YKS and Uncaria hook (UH), which is a constituent herb of YKS, had a partial agonistic effect to 5-HT(1A) receptors in vitro. However, it has still been unclear whether this in vitro effect is reflected in in vivo, and what the active ingredients are. The purpose of the present study is to find the active ingredient in YKS and to demonstrate the effect in in vivo. In the present study, we first studied the effect of YKS and UH on aggressiveness and sociality in socially isolated mice. YKS and UH ameliorated the isolation-induced increased aggressiveness and decreased sociality, and these ameliorative effects were counteracted by coadministration of 5-HT(1A) receptor antagonist WAY-100635, or disappeared by eliminating UH from YKS. These results suggest that the effect of YKS is mainly attributed to UH, and the active ingredient is contained in UH. To find the candidate ingredients, we examined competitive binding assay and [(35)S] guanosine 5'-O-(3-thiotriphosphate) (GTPgammaS) binding assay of seven major alkaloids in UH using Chinese hamster ovary cells expressing 5-HT(1A) receptors artificially. Only Geissoschizine methyl ether (GM) among seven alkaloids potently bound to 5-HT(1A) receptors and acted as a partial agonist. This in vitro result on GM was further demonstrated in the socially isolated mice. As did YKS and UH, GM ameliorated the isolation-induced increased aggressiveness and decreased sociality, and the effect was counteracted by coadministration of WAY-100635. These lines of results suggest that GM in UH is potent 5-HT(1A) receptor agonist and a candidate for pharmacological effect of YKS on aggressiveness and sociality in socially isolated mice.
Geissoschizine methyl ether has third-generation antipsychotic-like actions at the dopamine and serotonin receptors.[Pubmed:21951966]
Eur J Pharmacol. 2011 Dec 5;671(1-3):79-86.
Aripiprazole has made a significant contribution to the treatment of schizophrenia and related disorders. It has improved its safety and tolerability profiles, and these effects have been attributed to its pharmacological profile at the serotonin 5-HT and dopamine D(2) receptors. To discover compounds that have a similar pharmacological profile, we introduced a generic single-cell-based calcium imaging assay that standardizes the readouts from various assays used in previous studies on aripiprazole. In the present assay, the efficacy and potency of known ligands of serotonin 5-HT(1A), 5-HT(2A), 5-HT(2C), 5-HT(7) and dopamine D(2L) receptors were comparable to those found in previous studies using a variety of readouts. The developed assay was also able to reproduce the partial agonist activity, the low intrinsic activity and the selective activation of aripiprazole at the dopamine D(2L) receptors. Under identical experimental conditions, Geissoschizine methyl ether (GM), a plant indole alkaloid, behaved as a partial agonist at the serotonin 5-HT(1A) receptor, a partial agonist/antagonist at the dopamine D(2L) receptor and an antagonist at the serotonin 5-HT(2A), 5-HT(2C) and 5-HT(7) receptors. Interestingly, GM showed a relatively low intrinsic activity and evoked a partial activation response in a subset of cells expressing the dopamine D(2L) receptor; both of these effects were similarly observed for aripiprazole. Although GM is far less potent at the dopamine receptor than aripiprazole at dopamine D(2L) receptors (EC(50)=4.4 muM for GM vs. EC(50)=56 nM for aripiprazole), GM and GM derivatives may comprise a new set of candidates for atypical antipsychotics.


