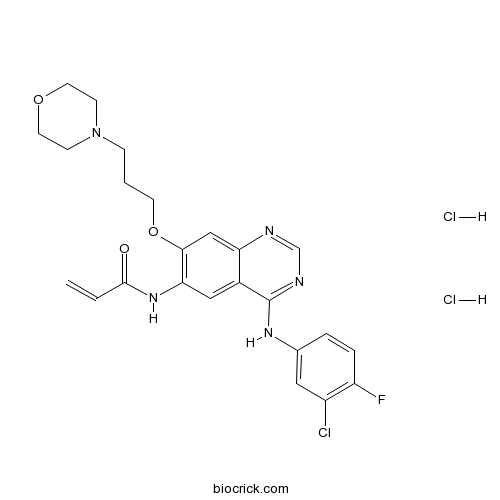
Canertinib dihydrochloridePan-ErbB inhibitor, potent and selective CAS# 289499-45-2 |
- Lapatinib
Catalog No.:BCC3633
CAS No.:231277-92-2
- Lapatinib Ditosylate
Catalog No.:BCC2083
CAS No.:388082-78-8
- Neratinib (HKI-272)
Catalog No.:BCC3685
CAS No.:698387-09-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 289499-45-2 | SDF | Download SDF |
| PubChem ID | 156413 | Appearance | Powder |
| Formula | C24H27Cl3FN5O3 | M.Wt | 558.87 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | CI 1033, PD 183805 | ||
| Solubility | DMSO : ≥ 155 mg/mL (277.35 mM) *"≥" means soluble, but saturation unknown. | ||
| Chemical Name | N-[4-(3-chloro-4-fluoroanilino)-7-(3-morpholin-4-ylpropoxy)quinazolin-6-yl]prop-2-enamide;dihydrochloride | ||
| SMILES | C=CC(=O)NC1=C(C=C2C(=C1)C(=NC=N2)NC3=CC(=C(C=C3)F)Cl)OCCCN4CCOCC4.Cl.Cl | ||
| Standard InChIKey | JZZFDCXSFTVOJY-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C24H25ClFN5O3.2ClH/c1-2-23(32)30-21-13-17-20(14-22(21)34-9-3-6-31-7-10-33-11-8-31)27-15-28-24(17)29-16-4-5-19(26)18(25)12-16;;/h2,4-5,12-15H,1,3,6-11H2,(H,30,32)(H,27,28,29);2*1H | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Potent EGFR kinase inhibitor (IC50 = 7.4 nM). Blocks autophosphorylation of erbB2. Inhibits growth of A431 tumor xenografts in mice. Cell permeable. |

Canertinib dihydrochloride Dilution Calculator

Canertinib dihydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.7893 mL | 8.9466 mL | 17.8932 mL | 35.7865 mL | 44.7331 mL |
| 5 mM | 0.3579 mL | 1.7893 mL | 3.5786 mL | 7.1573 mL | 8.9466 mL |
| 10 mM | 0.1789 mL | 0.8947 mL | 1.7893 mL | 3.5786 mL | 4.4733 mL |
| 50 mM | 0.0358 mL | 0.1789 mL | 0.3579 mL | 0.7157 mL | 0.8947 mL |
| 100 mM | 0.0179 mL | 0.0895 mL | 0.1789 mL | 0.3579 mL | 0.4473 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Canertinib dihydrochloride is a selective inhibitor of Pan-erbB tyrosine kinase with IC50 value of 3.5 nM [1].
ErbB receptor family is a member of the receptor tyrosine kinase superfamily and plays an important role in the normal breast development. It has been shown that ErbB overexpression involves in the pathogenesis and progression of breast cancer and is a negative prognostic indicator for breast cancer [2].
Canertinib dihydrochloride is a potent ErbB inhibitor and has a different selectivity with the reported ErbB inhibitor erlotinib. When tested with BT474 cells overexpressing ErbB2-receptor, treatment with Canertinib dihydrochloride induced cell apoptosis with 47% fraction by inhibiting Akt and MAPK activity [3]. In a panel of neuroblastoma cell lines, treated with Canertinib dihydrochloride resulted in a significant reduction in cell yield with IC50 value ranging from 0.94 μM to 2.45 μM and induced cell apoptosis from 3% to 56% tested with SK-N-SH cells [2].
In 12 week old NOD-SCID-IL2R gamma knockout mouse model with SK-N-SH cells subcutaneous xenograft, administration of Canertinib dihydrochloride for 18 days resulted in a marked reduction in tumor growth and final tumor weight [2].
References:
[1].Smaill, J.B., et al., Tyrosine kinase inhibitors. 17. Irreversible inhibitors of the epidermal growth factor receptor: 4-(phenylamino)quinazoline- and 4-(phenylamino)pyrido[3,2-d]pyrimidine-6-acrylamides bearing additional solubilizing functions. J Med Chem, 2000. 43(7): p. 1380-97.
[2].Richards, K.N., et al., Signaling of ERBB receptor tyrosine kinases promotes neuroblastoma growth in vitro and in vivo. Cancer, 2010. 116(13): p. 3233-43.
[3].Nelson, J.M. and D.W. Fry, Akt, MAPK (Erk1/2), and p38 act in concert to promote apoptosis in response to ErbB receptor family inhibition. J Biol Chem, 2001. 276(18): p. 14842-7.
- Anodendrine
Catalog No.:BCN1956
CAS No.:28942-07-6
- Fmoc-Cl
Catalog No.:BCC2802
CAS No.:28920-43-6
- Triazolam
Catalog No.:BCC5218
CAS No.:28911-01-5
- 3,7,16-Trihydroxystigmast-5-ene
Catalog No.:BCN5191
CAS No.:289056-24-2
- Ergosta-5,24(28)-diene-3,7,16-triol
Catalog No.:BCN5190
CAS No.:289054-34-8
- Z-D-Met-OH
Catalog No.:BCC2758
CAS No.:28862-80-8
- S-(-)-Carbidopa
Catalog No.:BCN8453
CAS No.:28860-95-9
- H-Asp(OBzl)-OBzl.TosOH
Catalog No.:BCC2886
CAS No.:2886-33 -1
- Cediranib (AZD217)
Catalog No.:BCC1121
CAS No.:288383-20-0
- (-)-dicentrine
Catalog No.:BCC8167
CAS No.:28832-07-7
- Lithospermic acid
Catalog No.:BCN5369
CAS No.:28831-65-4
- alpha-Asarone
Catalog No.:BCN3837
CAS No.:2883-98-9
- Allo-Thr-OH
Catalog No.:BCC3101
CAS No.:28954-12-3
- Cassiachromone
Catalog No.:BCN5192
CAS No.:28955-30-8
- Oridonin
Catalog No.:BCN5953
CAS No.:28957-04-2
- Lasiodin
Catalog No.:BCN3387
CAS No.:28957-08-6
- Senicapoc
Catalog No.:BCC1943
CAS No.:289656-45-7
- Boc-HomoArg(NO2)-OH
Catalog No.:BCC2646
CAS No.:28968-64-1
- CB 300919
Catalog No.:BCC1456
CAS No.:289715-28-2
- Pectolinarin
Catalog No.:BCN1217
CAS No.:28978-02-1
- Persicoside
Catalog No.:BCN4780
CAS No.:28978-03-2
- Z-N-Me-Phe-OH
Catalog No.:BCC3349
CAS No.:2899-07-2
- H-Trp-Oet.HCl
Catalog No.:BCC2673
CAS No.:2899-28-7
- H-Tryptophanol
Catalog No.:BCC2701
CAS No.:2899-29-8
Gateways to clinical trials.[Pubmed:18040531]
Methods Find Exp Clin Pharmacol. 2007 Oct;29(8):547-83.
(-)-Epigallocatechin gallate, [188Re]-P2045, 12B75, 89-12; Abacavir sulfate/lamivudine, Abatacept, Abiraterone acetate, ABT-869, Adalimumab, Ad-rh Endostatin, AI-700, Alemtuzumab, Alvimopan hydrate, Amrubicin hydrochloride, AP-12009, Apomab 7.3, Arformoterol tartrate, Aripiprazole, AS-1404, Azacitidine, AZD-0530; Bevacizumab, BHT-3009, Biapenem, Bortezomib, Bosentan, Bremelanotide; CA9-SCAN, Calcitonin gene-related peptide, Canertinib dihydrochloride, Cannabidiol, Carboxyamidotriazole, Caspofungin acetate, Celgosivir, Certolizumab pegol, Cinacalcet hydrochloride, Clevudine, CP-751871, Curcumin, Cx-401, Cypher; Darunavir, Decitabine, Deforolimus, Dexamet, Dipyridamole/prednisolone, Drospirenone, Drospirenone/estradiol, DTPw-HepB-Hib, Duloxetine hydrochloride; Efalizumab, Emtricitabine, Erlotinib hydrochloride, Escitalopram oxalate, Eszopiclone; Ferumoxtran-10, Ferumoxytol, Fondaparinux sodium, Fosaprepitant dimeglumine; gamma-Hydroxybutyrate sodium, Gefitinib, Genistein, Ghrelin (human), Gimatecan, GM-CSF PMED, Golimumab, gp100 PMED; Imatinib mesylate, Immunoglobulin intravenous (human), IV Gamma-globulin; LA-419, Laropiprant, L-BLP-25, Levodopa/carbidopa/entacapone, Lidocaine/prilocaine, Lopinavir/ritonavir, Lumiracoxib, LY-2076962; Mepolizumab, Methylnaltrexone bromide, Mitiglinide calcium hydrate, Mycophenolic acid sodium salt, Myristyl nicotinate; Natalizumab, Nesiritide, Niacin/lovastatin; Oblimersen sodium, Ofatumumab, Olmesartan medoxomil, Olmesartan medoxomil/hydrochlorothiazide, Ozarelix; Palonosetron hydrochloride, Parathyroid hormone (human recombinant), Pazopanib hydrochloride, Pegaptanib octasodium, Pegfilgrastim, Peginterferon alfa- 2a, Peginterferon alfa-2b, Pegvisomant, Pemetrexed disodium, Pexelizumab, Picoplatin, Pimecrolimus, Posaconazole, Pregabalin, PRO-1762, Progesterone caproate, Prulifloxacin; Ramelteon, Ranelic acid distrontium salt, Reparixin, Rosuvastatin calcium; Rotigotine; Satraplatin, Sertraline, Sipuleucel-T, SLIT-cisplatin, SNDX-275, Solifenacin succinate, Sunitinib malate; Tadalafil, Talnetant, Tanespimycin, Taxus, Tegaserod maleate, Telmisartan/hydrochlorothiazide, Tenofovir disoproxil fumarate/emtricitabine, Teriparatide, tgAAC-94, Tiotropium bromide, Tocilizumab, Tolvaptan, Trimethoprim; Vardenafil hydrochloride hydrate, Vatalanib succinate, Vinflunine, Voriconazole, VX-680; XL-880; Yttrium 90 (90Y) ibritumomab tiuxetan.
Gateways to clinical trials.[Pubmed:16636723]
Methods Find Exp Clin Pharmacol. 2006 Mar;28(2):121-42.
Gateways to Clinical Trials are a guide to the most recent clinical trials in current literature and congresses. The data in the following tables have been retrieved from the Clinical Trials Knowledge Area of Prous Science Integrity, the drug discovery and development portal, http://integrity.prous.com. This issue focuses on the following selection of drugs: 131I-labetuzumab; Abacavir sulfate, abatacept, adalimumab, ademetionine, adjuvanted influenza vaccine, alefacept, alemtuzumab, amlodipine, amphotericin B, anakinra, aripiprazole, aspirin, axitinib; Betamethasone dipropionate, bevacizumab, biphasic insulin aspart, bortezomib, bosentan, botulinum toxin type B, BQ-123; Calcium folinate, Canertinib dihydrochloride, carboplatin, carmustine, cetirizine hydrochloride, cetuximab, cholecalciferol, ciclesonide, ciclosporin, cinacalcet hydrochloride, cisplatin, clarithromycin, clofazimine, cold-adapted influenza vaccine trivalent, CpG-7909; Darbepoetin alfa, darifenacin hydrobromide, DB-289, desloratadine, Dexamet, dicycloverine hydrochloride, dimethyl fumarate, docetaxel, dolastatin 10, drospirenone, drospirenone/estradiol, duloxetine hydrochloride; Ecogramostim, edotecarin, efaproxiral sodium, enalapril maleate, epoetin beta, epoprostenol sodium, epratuzumab, erlotinib hydrochloride, escitalopram oxalate, estradiol, etanercept; Fluconazole, fludarabine phosphate, fluorouracil; Gefitinib, gemcitabine, Ghrelin (human), glibenclamide, glimepiride, GTI-2040; Haloperidol, human insulin, hydrocortisone probutate; Imatinib mesylate, indisulam, influenza vaccine, inhaled insulin, insulin aspart, insulin glulisine, insulin lispro, irinotecan, ispronicline; Lamivudine, lamivudine/zidovudine/abacavir sulfate, lapatinib, letrozole, levocetirizine, lomustine, lonafarnib, lumiracoxib;Magnesium sulfate, MD-1100, melphalan, metformin hydrochloride, methotrexate, metoclopramide hydrochloride, mitiglinide calcium hydrate, monophosphoryl lipid A, montelukast sodium, motexafin gadolinium, mycophenolate mofetil, mycophenolic acid sodium salt; Nitisinone; Omalizumab, omapatrilat, ONYX-015, oxaliplatin; Paclitaxel, paclitaxel nanoparticles, panitumumab, parathyroid hormone (human recombinant), peginterferon alfa-2a, peginterferon alfa-2b, peginterferon alfa-2b/ribavirin, pertuzumab, phosphatidylcholine-rich phospholipid mixture, pimecrolimus, pioglitazone hydrochloride, pramlintide acetate, prasterone; QR-333; Ranelic acid distrontium salt, ranolazine, rasagiline mesilate, RFB4(dsFv)-PE38, ribavirin, rifabutin, risperidone, rituximab, rofecoxib, rosiglitazone maleate, rosiglitazone maleate/metformin hydrochloride, rotavirus vaccine; S-236, salmeterol xinafoate, sarizotan hydrochloride, sildenafil, sildenafil citrate, sunitinib malate; Tadalafil, tegaserod maleate, temozolomide, tenofovir disoproxil fumarate, teriparatide, tiotropium bromide, tipifarnib, trabectedin, treprostinil sodium; Vandetanib, vardenafil hydrochloride hydrate, vatalanib succinate, vinflunine, virosome influenza vaccine, voriconazole; Zidovudine.
Gateways to clinical trials.[Pubmed:16273137]
Methods Find Exp Clin Pharmacol. 2005 Oct;27(8):569-612.
Gateways to Clinical Trials is a guide to the most recent clinical trials in current literature and congresses. The data in the following tables have been retrieved from the Clinical Trials Knowledge Area of Prous Science Integrity, the drug discovery and development portal, http://integrity.prous.com. This issue focuses on the following selection of drugs: (-)-Epigallocatechin gallate, (Z)-4-hydroxytamoxifen; Ad.muIFN-beta AD-237, adalimumab, adefovir dipivoxil, agalsidase alfa, alemtuzumab, almotriptan, ALVAC vCP1452, alvimopan hydrate, ambrisentan, anakinra, anti-IFN-gamma MAb; Bimatoprost, BMS-188797, BMS-214662, bortezomib, bosentan, bovine lactoferrin; Caffeine, Canertinib dihydrochloride, canfosfamide hydrochloride, cannabidiol, caspofungin acetate, cetuximab, cH36, ChimeriVax-JE, ciclesonide, cilansetron, cinacalcet hydrochloride, clopidogrel, CpG-7909, Cypher; Daptomycin, darbepoetin alfa, darifenacin hydrobromide, decitabine, denufosol tetrasodium, Dexamet, diindolemethane, drotrecogin alfa (activated), duloxetine hydrochloride, DX-9065a; E-7010, edaravone, efalizumab, eicosapentaenoic acid/docosahexaenoic acid, elacridar, eletriptan, emtricitabine, epratuzumab, erlotinib hydrochloride, ertapenem sodium, eszopiclone, everolimus, ezetimibe; Fludarabine, fondaparinux sodium; gamma-Hydroxybutyrate sodium, gavestinel sodium, gefitinib, granisetron-Biochronomer; Human Albumin, human insulin; Imatinib mesylate, indiplon, interleukin-2 XL, isatoribine, ISS-1018, i.v. gamma-globulin, ivabradine hydrochloride, ixabepilone; Lanthanum carbonate, L-arginine hydrochloride, liposomal doxorubicin, LY-450139; Magnesium sulfate, melatonin, motexafin gadolinium, mycophenolic acid sodium salt; Natalizumab, nesiritide, niacin/lovastatin; OGX-011, olmesartan medoxomil, omalizumab, ospemifene; PACAP38, panitumumab, parathyroid hormone (human recombinant), parecoxib sodium, patupilone, pegfilgrastim, peginterferon alfa-2a, peginterferon alfa-2b, peginterferon alfa-2b/ribavirin, pemetrexed disodium, pimecrolimus, pirfenidone, posaconazole, prasterone, pregabalin; R-112, ramelteon, ranolazine, rasagiline mesilate, rebimastat, roflumilast, rosuvastatin calcium, rotigotine hydrochloride, rupatadine fumarate; S-3013, S-3304, semustine, sitaxsentan sodium, St. John's Wort extract; Tadalafil, tamoxifen, Taxus, Tc-99m-EDDA-HYNIC-TOC, TH-9507, tiotropium bromide, tipifarnib, tocilizumab, tolvaptan, torcetrapib, TR-14035, tramadol hydrochloride/acetaminophen, treprostinil diethanolamine, troglitazone, troxacitabine; Valdecoxib, valganciclovir hydrochloride, vandetanib, vardenafil hydrochloride hydrate, VAS-991, veglin, vinflunine, voriconazole; White sweet potato extract; Ximelagatran.
Early phase I data on an irreversible pan-erb inhibitor: CI-1033. What did we learn?[Pubmed:15688609]
J Chemother. 2004 Nov;16 Suppl 4:44-8.
CI-1033 (Canertinib dihydrochloride) is an orally available pan-erbB receptor tyrosine kinase inhibitor that, unlike the majority of receptor inhibitors, effectively blocks signal transduction through all four members of the erbB family. In addition, it blocks the highly tumorigenic, constitutively activated variant of erbB-1, EGFRvIII, and inhibits downstream signaling through both the Ras/ MAP kinase, and PI-3 kinase/AKT pathways (Figure 1). CI-1033 is also unique in that it is an irreversible inhibitor; thereby providing prolonged suppression of erbB receptor-mediated signaling. Preclinical data show CI-1033 to be efficacious against a variety of human tumors in mouse xenograft models, including breast carcinomas.
Gateways to clinical trials.[Pubmed:14571286]
Methods Find Exp Clin Pharmacol. 2003 Sep;25(7):565-97.
Gateways to Clinical Trials is a guide to the most recent clinical trials in current literature and congresses. The data in the following tables has been retrieved from the Clinical Studies Knowledge Area of Prous Science Integrity, the drug discovery and development portal, http://integrity.prous.com. This issue focuses on the following selection of drugs: Abetimus sodium, adefovir dipivoxil, AGI-1067, alefacept, alemtuzumab, ALVAC-p53, aminolevulinic acid hydrochloride, aminolevulinic acid methyl ester, Anti-CTLA-4 Mab, AOD-9604, apafant, aprinocarsen sodium, arsenic trioxide; Balaglitazone, BIM-23190, bimatoprost, bortezomib, bosentan, BR-1; Canertinib dihydrochloride, CDP-850, cevimeline hydrochloride, cinacalcet hydrochloride, clenoliximab, clevudine, CN-787; D-003, darusentan, deferasirox, desloratadine dexanabinol, duloxetine hydrochloride; E-5564, edaravone, efaproxiral sodium, elvucitabine emfilermin, EN-101, enfuvirtide, entecavir, epithalon, eplerenone, erlotinib hydrochloride, escitalopram oxalate, esomeprazole magnesium, eszopiclone, etilefrine pivalate hydrochloride etoricoxib, everolimus, exenatide; Fidarestat, fondaparinux sodium; Ganstigmine hydrochloride; Homoharringtonine, HuMax-IL-15, hyperimmune IVIG; Imatinib mesylate, IMC-1C11, Inhaled insulin, irofulven, iseganan hydrochloride, ISIS-14803, ISIS-5132, ivabradine hydrochloride; Keratinocyte growth factor; Lafutidine, lanthanum carbonate, LAS-34475, levocetirizine, liraglutide, LY-307161 SR; Magnesium sulfate, maribavir, melatonin, mycobacterium cell wall complex; NN-414, NO-aspirin, nociceptin, nolomirole hydrochloride; Olmesartan medoxomil oral insulin, ospemifene; PDX, perillyl alcohol, pimecrolimus, pitavastatin calcium, pramlintide acetate, prasterone, pregabalin, PRO-542, PV-701, pyrazoloacridine; R-744, ranelic acid distrontium salt, rasburicase, rDNA insulin, resiniferatoxin, reslizumab, ridogrel, riplizumab ropivacaine, rosuvastatin calcium, roxifiban acetate, ruboxistaurin mesilate hydrate; Satraplatin, Sch-58500, semaxanib, sitaxsentan sodium, SMP-114, SU-6668; Teriparatide, tetrathiomolybdate, tipifarnib, tolvaptan, travoprost, treprostinil sodium; Valdecoxib, valganciclovir hydrochloride, vardenafil hydrochloride hydrate, vatalanib succinate; Ximelagatran; Z-335, ziprasidone hydrochloride, zoledronic acid monohydrate, ZYC-00101.
Tyrosine kinase inhibitors. 17. Irreversible inhibitors of the epidermal growth factor receptor: 4-(phenylamino)quinazoline- and 4-(phenylamino)pyrido[3,2-d]pyrimidine-6-acrylamides bearing additional solubilizing functions.[Pubmed:10753475]
J Med Chem. 2000 Apr 6;43(7):1380-97.
4-Anilinoquinazoline- and 4-anilinopyrido[3,2-d]pyrimidine-6-acrylamides substituted with solubilizing 7-alkylamine or 7-alkoxyamine side chains were prepared by reaction of the corresponding 6-amines with acrylic acid or acrylic acid anhydrides. In the pyrido[3,2-d]pyrimidine series, the intermediate 6-amino-7-alkylamines were prepared from 7-bromo-6-fluoropyrido[3,2-d]pyrimidine via Stille coupling with the appropriate stannane under palladium(0) catalysis. This proved a versatile method for the introduction of cationic solubilizing side chains. The compounds were evaluated for their inhibition of phosphorylation of the isolated EGFR enzyme and for inhibition of EGF-stimulated autophosphorylation of EGFR in A431 cells and of heregulin-stimulated autophosphorylation of erbB2 in MDA-MB 453 cells. Quinazoline analogues with 7-alkoxyamine solubilizing groups were potent irreversible inhibitors of the isolated EGFR enzyme, with IC(50[app]) values from 2 to 4 nM, and potently inhibited both EGFR and erbB2 autophosphorylation in cells. 7-Alkylamino- and 7-alkoxyaminopyrido[3,2-d]pyrimidines were also irreversible inhibitors with equal or superior potency against the isolated enzyme but were less effective in the cellular autophosphorylation assays. Both quinazoline- and pyrido[3,2-d]pyrimidine-6-acrylamides bound at the ATP site alkylating cysteine 773, as shown by electrospray ionization mass spectrometry, and had similar rates of absorptive and secretory transport in Caco-2 cells. A comparison of two 7-propoxymorpholide analogues showed that the pyrido[3,2-d]pyrimidine-6-acrylamide had greater amide instability and higher acrylamide reactivity, being converted to glutathione adducts in cells more rapidly than the corresponding quinazoline. This difference may contribute to the observed lower cellular potency of the pyrido[3,2-d]pyrimidine-6-acrylamides. Selected compounds showed high in vivo activity against A431 xenografts on oral dosing, with the quinazolines being superior to the pyrido[3,2-d]pyrimidines. Overall, the quinazolines proved superior to previous analogues in terms of aqueous solubility, potency, and in vivo antitumor activity, and one example (CI 1033) has been selected for clinical evaluation.


