Shizukaol ACAS# 131984-98-0 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 131984-98-0 | SDF | Download SDF |
| PubChem ID | 127045559 | Appearance | Powder |
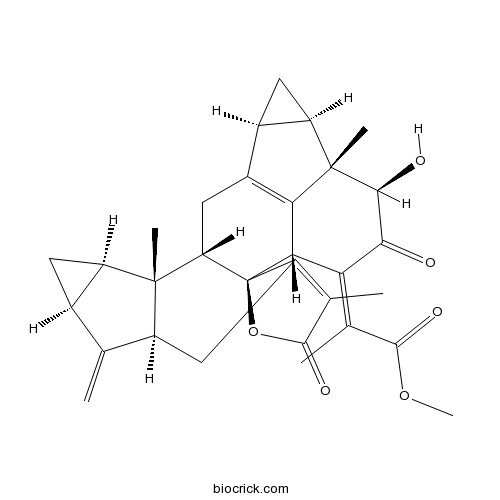
| Formula | C31H34O6 | M.Wt | 502.60 |
| Type of Compound | Sesquiterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| SMILES | CC1=C2CC3C(=C)C4CC4C3(C5C2(C6C7=C(C5)C8CC8C7(C(C(=O)C6=C(C)C(=O)OC)O)C)OC1=O)C | ||
| Standard InChIKey | GQSUZVYXPAKHQW-SFVFYCNCSA-N | ||
| Standard InChI | InChI=1S/C31H34O6/c1-11-14-7-19(14)29(4)17(11)10-18-12(2)28(35)37-31(18)21(29)9-16-15-8-20(15)30(5)23(16)24(31)22(25(32)26(30)33)13(3)27(34)36-6/h14-15,17,19-21,24,26,33H,1,7-10H2,2-6H3/b22-13-/t14-,15-,17+,19-,20-,21+,24+,26+,29-,30+,31+/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Shizukaol A is a natural product from Chloranthus japonicus. |
| Structure Identification | Phytochemistry, 1990, 29(7):2332-2334.Shizukaol a, a sesquiterpene dimer from Chloranthus japonicus.[Reference: WebLink]Shizukaol A, a novel dimeric sesquiterpene consisted of two lindenane (modified eudesmane) units was isolated from roots of Chloranthus japonicus. The structure was elucidated by 1D and 2D NMR analyses and by chemical methods. |

Shizukaol A Dilution Calculator

Shizukaol A Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.9897 mL | 9.9483 mL | 19.8965 mL | 39.7931 mL | 49.7413 mL |
| 5 mM | 0.3979 mL | 1.9897 mL | 3.9793 mL | 7.9586 mL | 9.9483 mL |
| 10 mM | 0.199 mL | 0.9948 mL | 1.9897 mL | 3.9793 mL | 4.9741 mL |
| 50 mM | 0.0398 mL | 0.199 mL | 0.3979 mL | 0.7959 mL | 0.9948 mL |
| 100 mM | 0.0199 mL | 0.0995 mL | 0.199 mL | 0.3979 mL | 0.4974 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 3,6,19-Trihydroxy-23-oxo-12-ursen-28-oic acid
Catalog No.:BCN1584
CAS No.:131984-82-2
- CC0651
Catalog No.:BCC4200
CAS No.:1319207-44-7
- 1,3,6,8-tetrahydroxy-4-(3-methyl-2-buten-1-yl)-9H-Xanthen-9-one
Catalog No.:BCN1585
CAS No.:1319198-98-5
- Paricalcitol
Catalog No.:BCC1839
CAS No.:131918-61-1
- Solanesol
Catalog No.:BCN2596
CAS No.:13190-97-1
- Goitrin
Catalog No.:BCN2764
CAS No.:13190-34-6
- Fudosteine
Catalog No.:BCC4661
CAS No.:13189-98-5
- 2-Hydroxyethyl Salicylate
Catalog No.:BCN3579
CAS No.:87-28-5
- (3R)-(+)-1-Benzyl-3-(tert-butoxycarbonylamino)pyrrolidine
Catalog No.:BCC8389
CAS No.:131878-23-4
- Lexacalcitol
Catalog No.:BCC1704
CAS No.:131875-08-6
- (R,R)-2,6-Bis(4-isopropyl-2-oxazolin-2-yl)pyridine
Catalog No.:BCC8396
CAS No.:131864-67-0
- Aphagranin A
Catalog No.:BCN6889
CAS No.:1318173-53-3
- Benztropine mesylate
Catalog No.:BCC4524
CAS No.:132-17-2
- Diphenylpyraline HCl
Catalog No.:BCC3768
CAS No.:132-18-3
- Pheniramine Maleate
Catalog No.:BCC4700
CAS No.:132-20-7
- Benzydamine HCl
Catalog No.:BCC4637
CAS No.:132-69-4
- Dihydrocucurbitacin B
Catalog No.:BCN3118
CAS No.:13201-14-4
- UNC 0646
Catalog No.:BCC2431
CAS No.:1320288-17-2
- UNC 0631
Catalog No.:BCC4143
CAS No.:1320288-19-4
- Cryptoacetalide
Catalog No.:BCN3139
CAS No.:132059-23-5
- Marmesin angelate
Catalog No.:BCN8139
CAS No.:13209-79-5
- Ropivacaine hydrochloride monohydrate
Catalog No.:BCC5169
CAS No.:132112-35-7
- O,O-diacetyldaurisoline
Catalog No.:BCC8221
CAS No.:132139-17-4
- Epi-Cryptoacetalide
Catalog No.:BCN3140
CAS No.:132152-57-9
[Sesquiterpenes with anti-metastasis breast cancer activity from Chloranthus henryi].[Pubmed:29243431]
Zhongguo Zhong Yao Za Zhi. 2017 Oct;42(20):3938-3944.
To study sesquiterpenes with anti-metastasis breast cancer activity from Chloranthus henryi, ten sesquiterpenes ,zedoarofuran (1), chlorajapolide D (2), 4beta, 8beta-dihydroxy-5alpha(H)-eudesm-7(11)-en-8, 12-olide (3), curcolonol (4), lasianthuslactone A (5), chlomultin C (6), (1E,4Z)-8-hydroxy-6-oxogermacra-1(10), 4, 7(11) -trieno-12, 8-lactone (7), shizukanolide E (8) , shizukanolide F (9) , 9alpha-hydroxycurcolonol (10), and five bis-sesquiterpenes, shizukaol B (11), shizukaol C (12) , cycloShizukaol A (13) , sarcandrolide B (14) , henriol A(15), were isolated by using different kinds of column chromatography methods from the ethyl acetate part of Ch.henryi and their structures were identified based on spectroscopic methods. Compounds 2, 8, 9, and 10 were obtained from the genus Chloranthus for the first time. Compounds 2, 5, 8-10, 12,and 14 were obtained from this plant for the first time. Some isolated compounds were subjected to evaluate the anti-metastasis breast cancer activity by using pharmacological methods, and only compounds 4, 11, and 12 were potent active.
Spicachlorantins G-J, new lindenane sesquiterpenoid dimers from the roots of Chloranthus spicatus.[Pubmed:21963639]
Chem Pharm Bull (Tokyo). 2011;59(10):1281-4.
Four new lindenane sesquiterpenoid dimers, spicachlorantins G-J (1-4), were isolated from the roots of Chloranthus spicatus together with seven known compounds, including chloramultilide A, shizukaol B, shizukaol D, shizukaol F, shizukaol P, chlorahololide D, and cycloShizukaol A. The planar structures of the new compounds were established by 1D-, 2D-NMR, and MS analyses. The absolute configurations of these compounds were determined by analyzing rotating Overhauser enhancement and exchange spectroscopy (ROESY) and circular dichroism (CD) spectra.
Terpenoids from Chloranthus multistachys.[Pubmed:18855217]
Nat Prod Res. 2008;22(13):1163-8.
Two new diterpenoids, ent-17-hydroxyl-16beta-methoxyl-kauran-3-one (1) and ent-17-acetoxyl-16beta-methoxyl-kauran-3-one (2), along with nine known compounds (3-12), ent-17-hydroxyl-kaur-15-en-3-one (3), ent-3beta-acetoxyl-kaur-15-en-16beta, 17-diol (4), ent-kauran-3beta, 16beta, 17-triol (5), ent-3beta-acetoxyl-kauran-16beta, 17-diol (6), ent-kauran-16beta, 17-diol (7), abbeokutone (8), ent-17alpha-acetyl-16beta-hydroxyl- kauran-3-one (9), shizukaol F (10), cycloShizukaol A (11) and curcolonol (12), were isolated from Chloranthus multistachys (Chloranthaceae). Their structures were elucidated by spectroscopic spectra.
Dimeric sesquiterpenoids isolated from Chloranthus japonicus inhibited the expression of cell adhesion molecules.[Pubmed:16229979]
J Ethnopharmacol. 2006 Mar 8;104(1-2):270-7.
In the search for cell adhesion inhibitors from natural sources, three active compounds were isolated from Chloranthus japonicus Sieb. (Chloranthaceae) roots. The compounds were identified as dimeric sesquiterpenoids of shizukaol B (1), cycloShizukaol A (2) and shizukaol F (3). These compounds inhibited PMA-induced homotypic aggregation of HL-60 cells without cytotoxicity with MIC values of 34.1 nM (1), 0.9 microM (2) and 27.3 nM (3), respectively. Although 1-3 did not affect the direct binding of LFA-1 to ICAM-1, these compounds markedly inhibited ICAM-1 expression in HL-60 cells in a dose-dependent fashion. On the other hand, when HUVEC were pretreated with 1-3 and stimulated with TNF-alpha, adhesion of THP-1 cells to HUVEC decreased in dose-dependent manner with IC(50) values of 54.6 nM, 1.2 microM and 34.1 nM, respectively. In fact, 1 inhibited TNF-alpha-induced surface expression of the ICAM-1, VCAM-1 and E-selectin in HUVEC with IC(50) values of 5.4 nM, 13.6 microM and 95.6 nM, respectively. The present findings suggest that 1-3 prevent monocyte adhesion to HUVEC through the inhibition of cell adhesion molecules expression stimulated by TNF-alpha.


