Schisanwilsonin HCAS# 1181216-83-0 |
- Schisantherin A
Catalog No.:BCN1024
CAS No.:58546-56-8
- Benzoylgomisin P
Catalog No.:BCN8803
CAS No.:129445-43-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1181216-83-0 | SDF | Download SDF |
| PubChem ID | 21672542 | Appearance | Cryst. |
| Formula | C30H32O9 | M.Wt | 536.6 |
| Type of Compound | Lignans | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
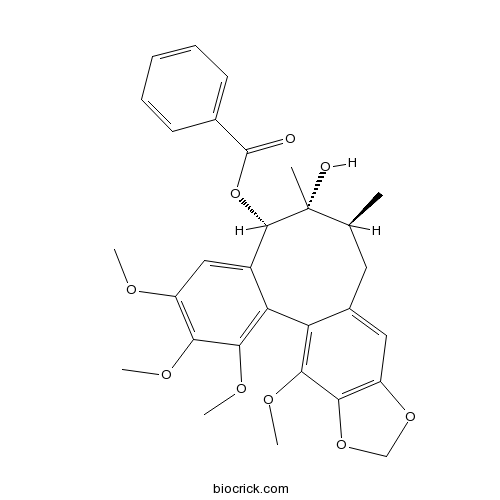
| Chemical Name | [(8S,9R,10S)-9-hydroxy-3,4,5,19-tetramethoxy-9,10-dimethyl-15,17-dioxatetracyclo[10.7.0.02,7.014,18]nonadeca-1(19),2,4,6,12,14(18)-hexaen-8-yl] benzoate | ||
| SMILES | CC1CC2=CC3=C(C(=C2C4=C(C(=C(C=C4C(C1(C)O)OC(=O)C5=CC=CC=C5)OC)OC)OC)OC)OCO3 | ||
| Standard InChIKey | UFCGDBKFOKKVAC-VLPWJMHXSA-N | ||
| Standard InChI | InChI=1S/C30H32O9/c1-16-12-18-13-21-25(38-15-37-21)26(35-5)22(18)23-19(14-20(33-3)24(34-4)27(23)36-6)28(30(16,2)32)39-29(31)17-10-8-7-9-11-17/h7-11,13-14,16,28,32H,12,15H2,1-6H3/t16-,28-,30+/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| In vitro | Schisanwilsonins A–G and related anti-HBV lignans from the fruits of Schisandra wilsoniana.[Reference: WebLink]Bioorganic & medicinal chemistry letters, 2009, 19(17):4958-4962.
|

Schisanwilsonin H Dilution Calculator

Schisanwilsonin H Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.8636 mL | 9.3179 mL | 18.6359 mL | 37.2717 mL | 46.5896 mL |
| 5 mM | 0.3727 mL | 1.8636 mL | 3.7272 mL | 7.4543 mL | 9.3179 mL |
| 10 mM | 0.1864 mL | 0.9318 mL | 1.8636 mL | 3.7272 mL | 4.659 mL |
| 50 mM | 0.0373 mL | 0.1864 mL | 0.3727 mL | 0.7454 mL | 0.9318 mL |
| 100 mM | 0.0186 mL | 0.0932 mL | 0.1864 mL | 0.3727 mL | 0.4659 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Karounidiol
Catalog No.:BCN2704
CAS No.:118117-31-0
- Cyprodime hydrochloride
Catalog No.:BCC7425
CAS No.:118111-54-9
- Prehelminthosporolactone
Catalog No.:BCN7289
CAS No.:118101-72-7
- L-365,260
Catalog No.:BCC7477
CAS No.:118101-09-0
- Zoledronic Acid
Catalog No.:BCC1067
CAS No.:118072-93-8
- Dihydroprehelminthosporol
Catalog No.:BCN7288
CAS No.:118069-95-7
- PS 48
Catalog No.:BCC7859
CAS No.:1180676-32-7
- 7-Hydroxy-3-(4-hydroxybenzyl)chroman
Catalog No.:BCN3578
CAS No.:1180504-64-6
- 4-O-Methylgrifolic acid
Catalog No.:BCN7287
CAS No.:118040-60-1
- Eriodictyol-6-glucoside
Catalog No.:BCN8026
CAS No.:118040-45-2
- PHM 27 (human)
Catalog No.:BCC5869
CAS No.:118025-43-7
- Blumeatin
Catalog No.:BCN6055
CAS No.:118024-26-3
- Schisanwilsonin I
Catalog No.:BCN5548
CAS No.:1181216-84-1
- [Phe8Ψ(CH-NH)-Arg9]-Bradykinin
Catalog No.:BCC5995
CAS No.:118122-39-7
- Volvaltrate B
Catalog No.:BCN6736
CAS No.:1181224-13-4
- 6''-O-Acetylastragalin
Catalog No.:BCN6058
CAS No.:118169-27-0
- 6-Oxo-23-norpristimerol
Catalog No.:BCN8054
CAS No.:118172-79-5
- 2-Picenecarboxylic acid
Catalog No.:BCN3063
CAS No.:118172-80-8
- EMD638683
Catalog No.:BCC1551
CAS No.:1181770-72-8
- Soyasaponin Ab
Catalog No.:BCN2896
CAS No.:118194-13-1
- 1,4-Dicaffeoylquinic acid
Catalog No.:BCN5912
CAS No.:1182-34-9
- Isodorsmanin A
Catalog No.:BCN6460
CAS No.:118266-99-2
- Lafutidine
Catalog No.:BCC4544
CAS No.:118288-08-7
- AF-DX 384
Catalog No.:BCC7024
CAS No.:118290-26-9
Spectroscopic characteristics of Rubricoccus marinus xenorhodopsin (RmXeR) and a putative model for its inward H(+) transport mechanism.[Pubmed:29034950]
Phys Chem Chem Phys. 2018 Jan 31;20(5):3172-3183.
A new group of microbial rhodopsins named xenorhodopsins (XeR), which are closely related to the cyanobacterial Anabaena sensory rhodopsin, show a light-driven "inward" proton transport activity, as reported for one representative of this group from Parvularcula oceani (PoXeR). In this study, we functionally and spectroscopically characterized a new member of the XeR clade from a marine bacterium Rubricoccus marinus SG-29(T) (RmXeR). Escherichia coli cells expressing recombinant RmXeR showed a light-induced alkalization of the cell suspension, which was strongly impaired by a protonophore, suggesting that RmXeR is a light-driven "inward" proton pump as is PoXeR. The spectroscopic properties of purified RmXeR were investigated and compared with those of PoXeR and a light-driven "outward" proton pump, bacteriorhodopsin (BR) from the archaeon Halobacterium salinarum. Action spectroscopy revealed that RmXeR with all-trans retinal is responsible for the light-driven inward proton transport activity, but not with 13-cis retinal. From pH titration experiments and mutational analysis, we estimated the pKa values for the protonated Schiff base of the retinal chromophore and its counterion as 11.1 +/- 0.07 and 2.1 +/- 0.07, respectively. Of note, the direction of both the retinal composition change upon light-dark adaptation and the acid-induced spectral shift was opposite that of BR, which is presumably related to the opposite directions of ion transport (from outside to inside for RmXeR and from inside to outside for BR). Flash photolysis experiments revealed the appearances of three intermediates (L, M and O) during the photocycle. The proton uptake and release were coincident with the formation and decay of the M intermediate, respectively. Together with associated findings from other microbial rhodopsins, we propose a putative model for the inward proton transport mechanism of RmXeR.
In vivo characterization of the downfield part of (1) H MR spectra of human brain at 9.4 T: Magnetization exchange with water and relation to conventionally determined metabolite content.[Pubmed:29034505]
Magn Reson Med. 2018 Jun;79(6):2863-2873.
PURPOSE: To perform exchange-rate measurements on the in vivo human brain downfield spectrum (5-10 ppm) at 9.4 T and to compare the variation in concentrations of the downfield resonances and of known upfield metabolites to determine potential peak labels. METHODS: Non-water-suppressed metabolite cycling was used in combination with an inversion transfer technique in two brain locations in healthy volunteers to measure the exchange rates and T1 values of exchanging peaks. Spectra were fitted with a heuristic model of a series of 13 or 14 Voigt lines, and a Bloch-McConnell model was used to fit the exchange rate curves. Concentrations from non-water-inverted spectra upfield and downfield were compared. RESULTS: Mean T1 values ranged from 0.40 to 0.77 s, and exchange rates from 0.74 to 13.8 s(-1) . There were no significant correlations between downfield and upfield concentrations, except for N-acetylaspartate, with a correlation coefficient of 0.63 and P < 0.01. CONCLUSIONS: Using ultrahigh field allowed improved separation of peaks in the 8.2 to 8.5 ppm amide proton region, and the exchange rates of multiple downfield resonances including the 5.8-ppm peak, previously tentatively assigned to urea, were measured in vivo in human brain. Downfield peaks consisted of overlapping components, and largely missing correlations between upfield and downfield resonances-although not conclusive-indicate limited contributions from metabolites present upfield to the downfield spectrum. Magn Reson Med 79:2863-2873, 2018. (c) 2017 International Society for Magnetic Resonance in Medicine.
Cu(I) Coordination Polymers as the Green Heterogeneous Catalysts for Direct C-H Bonds Activation of Arylalkanes to Ketones in Water with Spatial Confinement Effect.[Pubmed:29035050]
Inorg Chem. 2017 Nov 6;56(21):13329-13336.
To develop coordination polymers (CPs) as catalysts to selectively catalyze the reaction of C-H bond activation of arylalkanes to their homologous ketones, three new Cu(I)-based coordination polymers (Cu(I)-CPs) [CuI(aas-TPB)]n (1), [CuBr(ass-TPB)CH3CN]n (2), and {[Cu(ass-TPB)]Cl}n (3) (TPB = N,N,N-tris(3-pyridinyl)-1,3,5-benzenetricarboxamide) were synthesized. Structural variations from a herringbone fashion one-dimensional framework of 1 to a two-dimensional framework of 2 containing a 48-membered macrocycle and a cationic three-dimensional framework of 3 filled with Cl(-) anions were observed arising from the different halogen ions (I(-), Br(-), and Cl(-)). 1-3 were used as the green heterogeneous catalysts to catalyze direct C-H bond activation reactions of arylalkanes to ketones under mild reaction conditions with water as solvent. Handy product separation, convenient reaction procedures, and recyclability of these catalysts make the catalytic system fascinating. Moreover, the Cu(I)-CPs performed the reaction with high regioselectivity due to the unique spatial confinement effect of CPs.


