Odoratisol ACAS# 891182-93-7 |
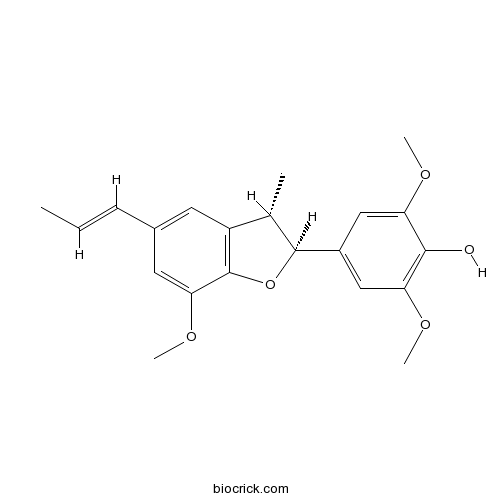
- (+)-5-methoxydeydrodiisoeugenol
Catalog No.:BCX0862
CAS No.:1967042-42-7
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 891182-93-7 | SDF | Download SDF |
| PubChem ID | 11703153 | Appearance | Powder |
| Formula | C21H24O5 | M.Wt | 356.41 |
| Type of Compound | Lignans | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 2,6-dimethoxy-4-[(2S,3S)-7-methoxy-3-methyl-5-[(E)-prop-1-enyl]-2,3-dihydro-1-benzofuran-2-yl]phenol | ||
| SMILES | CC=CC1=CC2=C(C(=C1)OC)OC(C2C)C3=CC(=C(C(=C3)OC)O)OC | ||
| Standard InChIKey | LYZVPGMCGPXCQO-QWTYKDCCSA-N | ||
| Standard InChI | InChI=1S/C21H24O5/c1-6-7-13-8-15-12(2)20(26-21(15)18(9-13)25-5)14-10-16(23-3)19(22)17(11-14)24-4/h6-12,20,22H,1-5H3/b7-6+/t12-,20-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Odoratisol A is a natural product from Myristica fragrans Houtt. |
| Kinase Assay | New acyclic bis phenylpropanoid and neolignans, from Myristica fragrans Houtt., exhibiting PARP-1 and NF-κB inhibitory effects.[Pubmed: 26920294 ]Food Chem. 2016 Jul 1;202:269-75.The bioassay-guided fractionation of the aril of Myristica fragrans (mace spice) yielded five phenolic compounds, one new acyclic bis phenylpropanoid (1) and four previously known phenolic compounds: compounds (1) (S) 1-(3,4,5-trimethoxyphenyl)-2-(3-methoxy-5-(prop-1-yl) phenyl)-propan-1-ol, (2) benzenemethanol; α-[1-[2,6-dimethoxy-4-(2-propen-1-yl)phenoxy]ethyl]-3,4-dimethoxy-1-acetate, (3) Odoratisol A, phenol, 4-[(2S,3S)-2,3-dihydro-7-methoxy-3-methyl-5-(1E)-1-propenyl-2-benzofuranyl]-2,6-dimethoxy, (4) 1,3-benzodioxate-5-methanol,α-[1-[2,6-dimethoxy-4-(2-propenyl)phenoxy]ethyl]-acetate, (5) licarin C; benzofuran,2,3-dihydro-7-methoxy-3-methyl-5-(1E)-1-yl-2-(3,4,5-trimethoxyphenyl).
|

Odoratisol A Dilution Calculator

Odoratisol A Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.8058 mL | 14.0288 mL | 28.0576 mL | 56.1151 mL | 70.1439 mL |
| 5 mM | 0.5612 mL | 2.8058 mL | 5.6115 mL | 11.223 mL | 14.0288 mL |
| 10 mM | 0.2806 mL | 1.4029 mL | 2.8058 mL | 5.6115 mL | 7.0144 mL |
| 50 mM | 0.0561 mL | 0.2806 mL | 0.5612 mL | 1.1223 mL | 1.4029 mL |
| 100 mM | 0.0281 mL | 0.1403 mL | 0.2806 mL | 0.5612 mL | 0.7014 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 4-O-Demethylisokadsurenin D
Catalog No.:BCN6650
CAS No.:89104-59-6
- BAMB-4
Catalog No.:BCC5428
CAS No.:891025-25-5
- 24,25-Epoxytirucall-7-en-3,23-dione
Catalog No.:BCN4437
CAS No.:890928-81-1
- GSK 650394
Catalog No.:BCC4070
CAS No.:890842-28-1
- ML 349
Catalog No.:BCC5612
CAS No.:890819-86-0
- VU 1545
Catalog No.:BCC7649
CAS No.:890764-63-3
- VU 29
Catalog No.:BCC7936
CAS No.:890764-36-0
- erythro-Guaiacylglycerol beta-coniferyl ether
Catalog No.:BCN1315
CAS No.:890317-92-7
- Dregeoside A11
Catalog No.:BCN3993
CAS No.:89020-11-1
- WDR5 0103
Catalog No.:BCC5626
CAS No.:890190-22-4
- Nutlin-3
Catalog No.:BCC2254
CAS No.:890090-75-2
- LUF6000
Catalog No.:BCC1710
CAS No.:890087-21-5
- Betulin caffeate
Catalog No.:BCN4438
CAS No.:89130-86-9
- MK-8776 (SCH-900776)
Catalog No.:BCC3817
CAS No.:891494-63-6
- SCH900776 S-isomer
Catalog No.:BCC1936
CAS No.:891494-64-7
- 15,16-Dihydrotanshindiol B
Catalog No.:BCN3213
CAS No.:891854-86-7
- 1,2-Didehydrocryptotanshinone
Catalog No.:BCN3122
CAS No.:891854-92-5
- 15,16-Dihydrotanshindiol C
Catalog No.:BCN3214
CAS No.:891854-96-9
- Efaroxan hydrochloride
Catalog No.:BCC6797
CAS No.:89197-00-2
- Fraxiresinol 1-O-glucoside
Catalog No.:BCN4439
CAS No.:89199-94-0
- Mulberrofuran H
Catalog No.:BCN3371
CAS No.:89199-99-5
- LY2334737
Catalog No.:BCC4060
CAS No.:892128-60-8
- Manidipine
Catalog No.:BCC4404
CAS No.:89226-50-6
- Manidipine 2HCl
Catalog No.:BCC4405
CAS No.:89226-75-5
New acyclic bis phenylpropanoid and neolignans, from Myristica fragrans Houtt., exhibiting PARP-1 and NF-kappaB inhibitory effects.[Pubmed:26920294]
Food Chem. 2016 Jul 1;202:269-75.
The bioassay-guided fractionation of the aril of Myristica fragrans (mace spice) yielded five phenolic compounds, one new acyclic bis phenylpropanoid (1) and four previously known phenolic compounds: compounds (1) (S) 1-(3,4,5-trimethoxyphenyl)-2-(3-methoxy-5-(prop-1-yl) phenyl)-propan-1-ol, (2) benzenemethanol; alpha-[1-[2,6-dimethoxy-4-(2-propen-1-yl)phenoxy]ethyl]-3,4-dimethoxy-1-acetate, (3) Odoratisol A, phenol, 4-[(2S,3S)-2,3-dihydro-7-methoxy-3-methyl-5-(1E)-1-propenyl-2-benzofuranyl]-2,6-d imethoxy, (4) 1,3-benzodioxate-5-methanol,alpha-[1-[2,6-dimethoxy-4-(2-propenyl)phenoxy]ethyl]- acetate, (5) licarin C; benzofuran,2,3-dihydro-7-methoxy-3-methyl-5-(1E)-1-yl-2-(3,4,5-trimethoxyphenyl). An NMR tube Mosher ester reaction was used in an approach to characterize and determine the assignment of the absolute configuration of the new isolated chiral alcohol (1). The PARP-1 inhibitory activity was evaluated for compound (1) (IC50=3.04muM), compound (2) (IC50=0.001muM), compound (4) (IC50=22.07muM) and compound (5) (IC50=3.11muM). Furthermore, the isolated secondary metabolites were tested for NF-kappaB and K-Ras inhibitory activities. When tested in the p65 assay, compounds (2) and (4) displayed potent NF-kappaB inhibition (IC50=1.5 nM and 3.4nM, respectively).


