GatifloxacinFluoroquinolone antibiotic,inhibits bacterial TOPO II CAS# 112811-59-3 |
- Dexrazoxane HCl (ICRF-187, ADR-529)
Catalog No.:BCC1087
CAS No.:149003-01-0
- Doxorubicin (Adriamycin) HCl
Catalog No.:BCC1117
CAS No.:25316-40-9
- Etoposide
Catalog No.:BCC1151
CAS No.:33419-42-0
- Genistein
Catalog No.:BCN5499
CAS No.:446-72-0
- Ellagic acid
Catalog No.:BCN5533
CAS No.:476-66-4
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 112811-59-3 | SDF | Download SDF |
| PubChem ID | 5379 | Appearance | Powder |
| Formula | C19H22FN3O4 | M.Wt | 375.39 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | AM 1155 | ||
| Solubility | Soluble to 10 mM in DMSO with gentle warming | ||
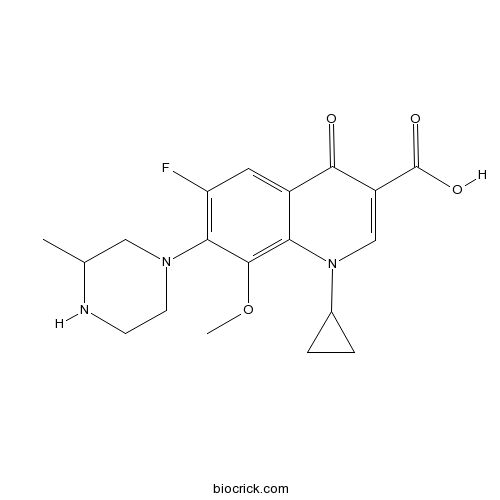
| Chemical Name | 1-Cyclopropyl-6-fluoro-1,4-dihydro- | ||
| SMILES | COc1c(N2CCNC(C)C2)c(F)cc3C(=O)C(=CN(C4CC4)c13)C(O)=O | ||
| Standard InChIKey | XUBOMFCQGDBHNK-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C19H22FN3O4/c1-10-8-22(6-5-21-10)16-14(20)7-12-15(18(16)27-2)23(11-3-4-11)9-13(17(12)24)19(25)26/h7,9-11,21H,3-6,8H2,1-2H3,(H,25,26) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Fluoroquinolone antibiotic. Inhibits bacterial type II topoisomerases (IC50 values are 0.109 and 13.8 μg/ml for E.coli DNA gyrase and S.aureus topoisomerase IV respectively). Displays potent activity against gram-positive and gram-negative bacteria. Stimulates short-term self-renewal in both human and mouse embryonic stem cells in vitro. |

Gatifloxacin Dilution Calculator

Gatifloxacin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.6639 mL | 13.3195 mL | 26.639 mL | 53.2779 mL | 66.5974 mL |
| 5 mM | 0.5328 mL | 2.6639 mL | 5.3278 mL | 10.6556 mL | 13.3195 mL |
| 10 mM | 0.2664 mL | 1.3319 mL | 2.6639 mL | 5.3278 mL | 6.6597 mL |
| 50 mM | 0.0533 mL | 0.2664 mL | 0.5328 mL | 1.0656 mL | 1.3319 mL |
| 100 mM | 0.0266 mL | 0.1332 mL | 0.2664 mL | 0.5328 mL | 0.666 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Fluoroquinolone antibiotic. Inhibits bacterial type II topoisomerases (IC50 values are 0.109 and 13.8 μg/ml for E.coli DNA gyrase and S.aureus topoisomerase IV respectively). Displays potent activity against gram-positive and
- Letrozole
Catalog No.:BCC1063
CAS No.:112809-51-5
- Osthenone
Catalog No.:BCN4731
CAS No.:112789-90-9
- Clemastanin B
Catalog No.:BCC8152
CAS No.:112747-98-5
- Galanin (1-15) (porcine, rat)
Catalog No.:BCC5762
CAS No.:112747-70-3
- exo-IWR 1
Catalog No.:BCC7823
CAS No.:1127442-87-8
- IWR-1-endo
Catalog No.:BCC5102
CAS No.:1127442-82-3
- Oleonuezhenide
Catalog No.:BCN6011
CAS No.:112693-21-7
- Erigeside C
Catalog No.:BCN6010
CAS No.:112667-09-1
- Fragransin A2
Catalog No.:BCN6008
CAS No.:112652-46-7
- 1,7-Dihydroxyacridone
Catalog No.:BCN7275
CAS No.:112649-95-3
- BR-Xanthone A
Catalog No.:BCN6007
CAS No.:112649-48-6
- Garcinone E
Catalog No.:BCN3604
CAS No.:112649-21-5
- 1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid ethyl ester
Catalog No.:BCC8465
CAS No.:112811-71-9
- 1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid
Catalog No.:BCC8464
CAS No.:112811-72-0
- Dynole 34-2
Catalog No.:BCC7891
CAS No.:1128165-88-7
- Methyllycaconitine citrate
Catalog No.:BCC6897
CAS No.:112825-05-5
- 24R-Calcipotriol
Catalog No.:BCC1304
CAS No.:112827-99-3
- (24R)-MC 976
Catalog No.:BCC1289
CAS No.:112828-09-8
- (24S)-MC 976
Catalog No.:BCC1291
CAS No.:112849-14-6
- Fmoc-D-Met-OH
Catalog No.:BCC3532
CAS No.:112883-40-6
- Fmoc-D-Nle-OH
Catalog No.:BCC3300
CAS No.:112883-41-7
- Fmoc-N-Me-Nle-OH
Catalog No.:BCC3299
CAS No.:112883-42-8
- Fmoc-2-Nal-OH
Catalog No.:BCC3292
CAS No.:112883-43-9
- Mosapride
Catalog No.:BCC4078
CAS No.:112885-41-3
Besifloxacin Ophthalmic Suspension 0.6% Compared with Gatifloxacin Ophthalmic Solution 0.3% for the Treatment of Bacterial Conjunctivitis in Neonates.[Pubmed:28078599]
Drugs R D. 2017 Mar;17(1):167-175.
PURPOSE: The aim of this study was to evaluate the safety and efficacy of topical besifloxacin ophthalmic suspension 0.6% compared with Gatifloxacin ophthalmic solution 0.3% in the treatment of bacterial conjunctivitis in neonates. METHODS: This was a multicenter, randomized, double-masked, parallel group study. Subjects /=1 (scale 0-3) for both conjunctival discharge and conjunctival hyperemia were randomized to besifloxacin or Gatifloxacin instilled three times daily for 7 days, and completed five study visits (three clinic visits and two phone calls). Primary endpoints included clinical resolution (absence of both conjunctival discharge and conjunctival hyperemia) at visit 5 (day 8 or 9) and ocular and non-ocular treatment-emergent adverse events (AEs). Bacterial eradication was a secondary endpoint. RESULTS: Thirty-three subjects were included in the intent-to-treat (ITT) population. All were aged <28 days, with a mean (standard deviation) age of 15.5 days (6.0), and 57.6% were female. Twenty-two subjects had culture-confirmed conjunctivitis in at least one eye (modified ITT [mITT] population), most often with Gram-positive bacteria. Visit 5 clinical resolution and bacterial eradication rates were comparable among besifloxacin- and Gatifloxacin-treated study eyes (clinical resolution: 12/16 [75.0%] vs. 12/17 [70.6%] for the ITT population, and 11/13 [84.6%] vs. 7/9 [77.8%] for the mITT population; bacterial eradication: 12/13 [92.3%] vs. 8/9 [88.9%] for the mITT population, respectively). No AEs were reported in the besifloxacin treatment group, and AEs reported in the Gatifloxacin group were considered not treatment-related. CONCLUSIONS: In this small study in neonates, both besifloxacin and Gatifloxacin appeared effective and safe in the treatment of bacterial conjunctivitis. Larger studies are warranted.
Surface-modified gatifloxacin nanoparticles with potential for treating central nervous system tuberculosis.[Pubmed:28331318]
Int J Nanomedicine. 2017 Mar 13;12:1959-1968.
A new nanocarrier is developed for the passage of Gatifloxacin through the blood-brain barrier to treat central nervous system tuberculosis. Gatifloxacin nanoparticles were prepared by nanoprecipitation using poly(lactic-co-glycolic acid) (PLGA) 502 and polysorbate 80 or Labrafil as surface modifiers. The evaluation of in vivo blood-brain barrier transport was carried out in male Wistar rats using rhodamine-loaded PLGA nanoparticles prepared with and without the surface modifiers. At 30 and 60 minutes after administration, nanoparticle biodistribution into the brain (hippocampus and cortex), lungs, and liver was studied. The results obtained from the cerebral cortex and hippocampus showed that functionalization of rhodamine nanoparticles significantly increased their passage into the central nervous system. At 60 minutes, rhodamine concentrations decreased in both the lungs and the liver but were still high in the cerebral cortex. To distinguish the effect between the surfactants, Gatifloxacin-loaded PLGA nanoparticles were prepared. The best results corresponded to the formulation prepared with polysorbate 80 with regard to encapsulation efficiency (28.2%), particle size (176.5 nm), and zeta-potential (-20.1 mV), thereby resulting in a promising drug delivery system to treat cerebral tuberculosis.
Simultaneous quantification of gatifloxacin, moxifloxacin, and besifloxacin concentrations in cornea and aqueous humor by LC-QTOF/MS after topical ocular dosing.[Pubmed:27670369]
J Pharmacol Toxicol Methods. 2017 Jan - Feb;83:87-93.
The fourth-generation fluoroquinolones are widely used as ophthalmic antimicrobials. This study aimed to validate a new analytical technique for simultaneous quantification of Gatifloxacin, moxifloxacin, and besifloxacin concentrations in the cornea and aqueous humor by liquid chromatography (LC) coupled to quadrupole time-of-flight mass spectrometry (QTOF/MS) at 10min and 1h after instillation of topical ophthalmic antimicrobial suspensions. It was used twenty-two male dogs without ocular lesions verified by ophthalmic and histologic examinations. Methanol:water (4:1) was used for the extraction procedure for cornea and acetonitrile:water (4:1) was used for aqueous humor. The chromatographic separations were carried out on a C18 column with a linear gradient of water and methanol, both containing 0.1% formic acid. The total chromatographic run time was 4min. Mass spectrometry analyses were performed on a Xevo G2-S QTof tandem mass spectrometer, operated in a positive ion electrospray ionization (ESI+) mode. The retention times were approximately 1.42min for Gatifloxacin, 1.87min for moxifloxacin, and 3.01min for besifloxacin. No interference peak was detected for the three tested antimicrobials in samples obtained from both cornea and aqueous humor, ensuring that the peak response was exclusive to the analyte of interest. The limit of detection for the three antimicrobials was 0.11mug/mL and the limit of quantification was 0.42mug/mL for both cornea and aqueous humor samples. At both time points post instillation of the three antimicrobials, moxifloxacin had the highest corneal concentration and besifloxacin demonstrated the highest concentration in the aqueous humor.
High-throughput screening assay for the identification of compounds regulating self-renewal and differentiation in human embryonic stem cells.[Pubmed:18522853]
Cell Stem Cell. 2008 Jun 5;2(6):602-12.
High-throughput screening (HTS) of chemical libraries has become a critical tool in basic biology and drug discovery. However, its implementation and the adaptation of high-content assays to human embryonic stem cells (hESCs) have been hampered by multiple technical challenges. Here we present a strategy to adapt hESCs to HTS conditions, resulting in an assay suitable for the discovery of small molecules that drive hESC self-renewal or differentiation. Use of this new assay has led to the identification of several marketed drugs and natural compounds promoting short-term hESC maintenance and compounds directing early lineage choice during differentiation. Global gene expression analysis upon drug treatment defines known and novel pathways correlated to hESC self-renewal and differentiation. Our results demonstrate feasibility of hESC-based HTS and enhance the repertoire of chemical compounds for manipulating hESC fate. The availability of high-content assays should accelerate progress in basic and translational hESC biology.
Antibacterial activity of gatifloxacin (AM-1155, CG5501, BMS-206584), a newly developed fluoroquinolone, against sequentially acquired quinolone-resistant mutants and the norA transformant of Staphylococcus aureus.[Pubmed:9687384]
Antimicrob Agents Chemother. 1998 Aug;42(8):1917-22.
Alternate mutations in the grlA and gyrA genes were observed through the first- to fourth-step mutants which were obtained from four Staphylococcus aureus strains by sequential selection with several fluoroquinolones. The increases in the MICs of Gatifloxacin accompanying those mutational steps suggest that primary targets of Gatifloxacin in the wild type and the first-, second-, and third-step mutants are wild-type topoisomerase IV (topo IV), wild-type DNA gyrase, singly mutated topo IV, and singly mutated DNA gyrase, respectively. Gatifloxacin had activity equal to that of tosufloxacin and activity more potent than those of norfloxacin, ofloxacin, ciprofloxacin, and sparfloxacin against the second-step mutants (grlA gyrA; Gatifloxacin MIC range, 1.56 to 3.13 microg/ml) and had the most potent activity against the third-step mutants (grlA gyrA grlA; Gatifloxacin MIC range, 1.56 to 6.25 microg/ml), suggesting that Gatifloxacin possesses the most potent inhibitory activity against singly mutated topo IV and singly mutated DNA gyrase among the quinolones tested. Moreover, Gatifloxacin selected resistant mutants from wild-type and the second-step mutants at a low frequency. Gatifloxacin possessed potent activity (MIC, 0.39 microg/ml) against the NorA-overproducing strain S. aureus NY12, the norA transformant, which was slightly lower than that against the parent strain SA113. The increases in the MICs of the quinolones tested against NY12 were negatively correlated with the hydrophobicity of the quinolones (correlation coefficient, -0.93; P < 0.01). Therefore, this slight decrease in the activity of Gatifloxacin is attributable to its high hydrophobicity. Those properties of Gatifloxacin likely explain its good activity against quinolone-resistant clinical isolates of S. aureus harboring the grlA, gyrA, and/or norA mutations.
Inhibitory activities of gatifloxacin (AM-1155), a newly developed fluoroquinolone, against bacterial and mammalian type II topoisomerases.[Pubmed:9756776]
Antimicrob Agents Chemother. 1998 Oct;42(10):2678-81.
We determined the inhibitory activities of Gatifloxacin against Staphylococcus aureus topoisomerase IV, Escherichia coli DNA gyrase, and HeLa cell topoisomerase II and compared them with those of several quinolones. The inhibitory activities of quinolones against these type II topoisomerases significantly correlated with their antibacterial activities or cytotoxicities (correlation coefficient [r] = 0.926 for S. aureus, r = 0.972 for E. coli, and r = 0.648 for HeLa cells). Gatifloxacin possessed potent inhibitory activities against bacterial type II topoisomerases (50% inhibitory concentration [IC50] = 13.8 microg/ml for S. aureus topoisomerase IV; IC50 = 0.109 microg/ml for E. coli DNA gyrase) but the lowest activity against HeLa cell topoisomerase II (IC50 = 265 microg/ml) among the quinolones tested. There was also a significant correlation between the inhibitory activities of quinolones against S. aureus topoisomerase IV and those against E. coli DNA gyrase (r = 0.969). However, the inhibitory activity against HeLa cell topoisomerase II did not correlate with that against either bacterial enzyme. The IC50 of Gatifloxacin for HeLa cell topoisomerase II was 19 and was more than 2,400 times higher than that for S. aureus topoisomerase IV and that for E. coli DNA gyrase. These ratios were higher than those for other quinolones, indicating that Gatifloxacin possesses a higher selectivity for bacterial type II topoisomerases.


