ZosuquidarMDR modulator CAS# 167354-41-8 |
- Dofequidar
Catalog No.:BCC4176
CAS No.:129716-58-1
- Dofequidar fumarate
Catalog No.:BCC4177
CAS No.:153653-30-6
- LY335979 (Zosuquidar 3HCL)
Catalog No.:BCC3878
CAS No.:167465-36-3
- Tariquidar
Catalog No.:BCC3625
CAS No.:206873-63-4
- Tariquidar methanesulfonate, hydrate
Catalog No.:BCC1986
CAS No.:625375-83-9
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 167354-41-8 | SDF | Download SDF |
| PubChem ID | 3036703 | Appearance | Powder |
| Formula | C32H31F2N3O2 | M.Wt | 527.6 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | LY335979 | ||
| Solubility | DMSO : 8.33 mg/mL (15.79 mM; Need ultrasonic) H2O : < 0.1 mg/mL (insoluble) | ||
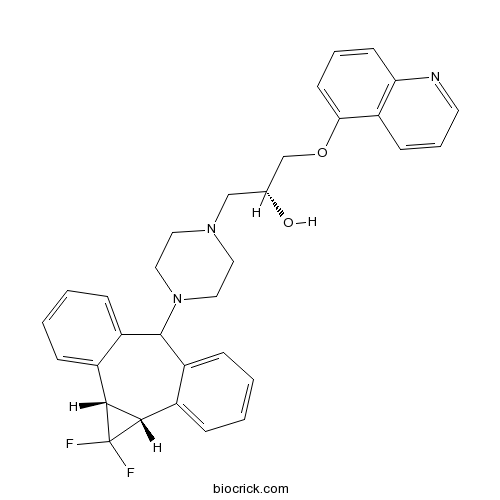
| Chemical Name | (2R)-1-[4-[(2R,4S)-3,3-difluoro-11-tetracyclo[10.4.0.02,4.05,10]hexadeca-1(16),5,7,9,12,14-hexaenyl]piperazin-1-yl]-3-quinolin-5-yloxypropan-2-ol | ||
| SMILES | C1CN(CCN1CC(COC2=CC=CC3=C2C=CC=N3)O)C4C5=CC=CC=C5C6C(C6(F)F)C7=CC=CC=C47 | ||
| Standard InChIKey | IHOVFYSQUDPMCN-XAKVHENESA-N | ||
| Standard InChI | InChI=1S/C32H31F2N3O2/c33-32(34)29-22-7-1-3-9-24(22)31(25-10-4-2-8-23(25)30(29)32)37-17-15-36(16-18-37)19-21(38)20-39-28-13-5-12-27-26(28)11-6-14-35-27/h1-14,21,29-31,38H,15-20H2/t21-,29-,30+,31?/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Zosuquidar (LY335979) is a potent negative modulator of P-glycoprotein-mediated multi-drug resistance with Ki of 60 nM.
IC50 value: 60 nM (Ki)
Target: P-glycoprotein
Zosuquidar (LY335979) is a potent modulator of P-glycoprotein-mediated multidrug resistance with Ki of 60 nM. Zosuquidar binds with high affinity to P-glycoprotein and inhibits P-glycoprotein-mediated multidrug resistance (MDR). P-glycoprotein, encoded by the MDR-1 gene, is a member of the ATP-binding cassette superfamily of transmembrane transporters and prevents the intracellular accumulation of many natural product-derived cytotoxic agents. Zosuquidar is a compound of antineoplastic drug candidates currently under development. References: | |||||

Zosuquidar Dilution Calculator

Zosuquidar Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.8954 mL | 9.4769 mL | 18.9538 mL | 37.9075 mL | 47.3844 mL |
| 5 mM | 0.3791 mL | 1.8954 mL | 3.7908 mL | 7.5815 mL | 9.4769 mL |
| 10 mM | 0.1895 mL | 0.9477 mL | 1.8954 mL | 3.7908 mL | 4.7384 mL |
| 50 mM | 0.0379 mL | 0.1895 mL | 0.3791 mL | 0.7582 mL | 0.9477 mL |
| 100 mM | 0.019 mL | 0.0948 mL | 0.1895 mL | 0.3791 mL | 0.4738 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Zosuquidar (LY335979) is a potent modulator of P-glycoprotein-mediated multidrug resistance with Ki of 60 nM. Zosuquidar binds with high affinity to P-glycoprotein and inhibits P-glycoprotein-mediated multidrug resistance (MDR).
- 9-Hydroxyeriobofuran
Catalog No.:BCN7444
CAS No.:167278-41-3
- Malvidin 3,5-Diglucoside
Catalog No.:BCC8206
CAS No.:16727-30-3
- Cyanidin 3-Sophoroside-5-Glucoside
Catalog No.:BCC8158
CAS No.:16727-02-9
- Clevidipine Butyrate
Catalog No.:BCC4401
CAS No.:167221-71-8
- Furagin
Catalog No.:BCC1582
CAS No.:1672-88-4
- R-96544 hydrochloride
Catalog No.:BCC7164
CAS No.:167144-80-1
- Autocamtide-2-related inhibitory peptide
Catalog No.:BCC7153
CAS No.:167114-91-2
- 10-Oxo Docetaxel
Catalog No.:BCC5409
CAS No.:167074-97-7
- Pitavastatin ethyl ester
Catalog No.:BCC9122
CAS No.:167073-19-0
- Fmoc-D-Cys(Trt)-OH
Catalog No.:BCC3481
CAS No.:167015-11-4
- BMS 453
Catalog No.:BCC7679
CAS No.:166977-43-1
- N-Acetoacetylmorpholine
Catalog No.:BCC9078
CAS No.:16695-54-8
- Fmoc-Lys(Mtt)-OH
Catalog No.:BCC3523
CAS No.:167393-62-6
- H-Cys(Bzl)-OMe.HCl
Catalog No.:BCC2907
CAS No.:16741-80-3
- Sophoracarpan A
Catalog No.:BCN6980
CAS No.:1674359-82-0
- Sophoracarpan B
Catalog No.:BCN6979
CAS No.:1674359-84-2
- LY335979 (Zosuquidar 3HCL)
Catalog No.:BCC3878
CAS No.:167465-36-3
- BADGE
Catalog No.:BCC7022
CAS No.:1675-54-3
- PD-1/PD-L1 inhibitor 2
Catalog No.:BCC6520
CAS No.:1675203-84-5
- 11-Hydroxy-12-methoxyabietatriene
Catalog No.:BCN3253
CAS No.:16755-54-7
- Benoxafos
Catalog No.:BCC5470
CAS No.:16759-59-4
- H-Glu(OBzl)-OH
Catalog No.:BCC2926
CAS No.:1676-73-9
- Z-Ser(tBu)-OH
Catalog No.:BCC2740
CAS No.:1676-75-1
- Boc-Asp(OtBu)-OH
Catalog No.:BCC3368
CAS No.:1676-90-0
Zosuquidar and an albumin-binding prodrug of zosuquidar reverse multidrug resistance in breast cancer cells of doxorubicin and an albumin-binding prodrug of doxorubicin.[Pubmed:22228402]
Breast Cancer Res Treat. 2012 Jul;134(1):117-29.
The P-glycoprotein (P-gp) is a 170-kDa protein that acts as an energy dependent, transmembrane efflux pump and is encoded by the MDR1 gene. It has been shown to be responsible for multidrug resistance (MDR) in a defined subpopulation of breast cancer patients and thus represents a molecular target for circumventing MDR in this tumor indication. MDR modulators have been developed and demonstrated high selectivity for P-gp with inhibitory activities in the low nanomolar range. Although some objective responses were achieved in clinical trials, combination therapy with these MDR modulators, such as Ca2+ antagonists caused unacceptable toxicity. Targeting P-gp inhibitors to the tumor site is a mean to increase their therapeutic index, and in this context binding of tailor-made prodrugs to circulating albumin is an established technology to reduce the toxicity and enhance the efficacy of anticancer drugs. In this study, we consequently developed an acid-sensitive albumin-binding prodrug of the P-gp inhibitor Zosuquidar (LY335979) in a two-step synthesis using a maleimide hydrazone linker system established in our laboratory that first introduces acetylbenzoic acid at the HO-group of Zosuquidar followed by derivatization with 6-maleimidocaproyl hydrazide to form the acid-sensitive hydrazone bond. The maleimide group enables the prodrug to bind rapidly and selectively to the cysteine-34 position of endogenous albumin after intravenous administration. HPLC analysis demonstrated rapid albumin binding of the Zosuquidar prodrug as well as the quantitative release of the acetylbenzoic ester derivative of Zosuquidar at pH 5.0. Subsequently, its ability to circumvent MDR was tested in two doxorubicin-resistant breast carcinoma cell lines (MCF-7/ADR and MT-3/ADR). The MDR status of these cell lines can be reversed by Zosuquidar which was confirmed in a rhodamine 123 assay using fluorescence microscopy and FACS analysis. Furthermore, Zosuquidar as well its acid-sensitive albumin conjugate re-sensitized cells to doxorubicin as well as to an albumin-binding prodrug of doxorubicin, i.e., the 6-maleimidocaproyl hydrazone derivative of doxorubicin, achieving IC50 values in the same order of magnitude as the parental cell lines. Thus, a novel formulation of Zosuquidar has been developed that could have the potential to improve the toxicity issues and tumor targeting properties of the original compound.
Triggering of Suicidal Erythrocyte Death by Zosuquidar.[Pubmed:26646161]
Cell Physiol Biochem. 2015;37(6):2355-65.
BACKGROUND: The P-glycoprotein inhibitor Zosuquidar (LY335979) is clinically used to augment the effect of cytostatic drugs on suicidal tumor cell death or apoptosis. The present study explored whether the substance is cytotoxic to erythrocytes. Upon injury, erythrocytes may undergo suicidal cell death or eryptosis, which is characterized by cell shrinkage and translocation of cell membrane phosphatidylserine to the erythrocyte surface. Signaling of eryptosis include increase of cytosolic Ca2+-activity ([Ca2+]i), oxidative stress and activation of several kinases, such as p38 kinase and protein kinase C. METHODS: Phosphatidylserine abundance at the erythrocyte surface was quantified from binding of FITC-labelled annexin-V, cell volume from forward scatter, [Ca2+]i from Fluo3-fluorescence, and reactive oxygen species (ROS) from 2',7'-dichlorodihydrofluorescein diacetate (DCFDA) fluorescence. RESULTS: A 48 h treatment of human erythrocytes with Zosuquidar significantly increased the percentage of annexin-V-binding cells (2 and 4 microg/ml), significantly decreased forward scatter (4 microg/ml), significantly increased [Ca2+]i (4 microg/ml), but did not significantly modify ROS. The up-regulation of annexin-V-binding following Zosuquidar (4 microg/ml) treatment was significantly blunted by removal of extracellular Ca2+, by presence of p38 kinase inhibitor SB203580 (2 microM) and by presence of protein kinase C inhibitor calphostin (100 nM). CONCLUSIONS: Exposure of erythrocytes to Zosuquidar triggers suicidal erythrocyte death with erythrocyte shrinkage and erythrocyte membrane scrambling, an effect involving Ca2+ entry and requiring activity of SB203580 and calphostin sensitive kinases.
Zosuquidar, a novel modulator of P-glycoprotein, does not improve the outcome of older patients with newly diagnosed acute myeloid leukemia: a randomized, placebo-controlled trial of the Eastern Cooperative Oncology Group 3999.[Pubmed:20716770]
Blood. 2010 Nov 18;116(20):4077-85.
Zosuquidar, which modulates P-glycoprotein (P-gp) with minimal delay of anthracycline clearance, may reverse P-gp-mediated resistance in acute myeloid leukemia without increased toxicity. A total of 449 adults older than 60 years with acute myeloid leukemia or high-risk myelodysplastic syndrome enrolled in a randomized placebo-controlled double-blind trial (Eastern Cooperative Oncology Group 3999). Overall survival was compared between patients receiving conventional-dose cytarabine and daunorubicin and either Zosuquidar (550 mg; 212 patients) or placebo (221 patients). Median and 2-year overall survival values were 7.2 months and 20% on Zosuquidar and 9.4 months and 23% on placebo, respectively (P = .281). Remission rate was 51.9% on Zosuquidar and 48.9% on placebo. All cause mortality to day 42 was not different (Zosuquidar 22.2% vs placebo 16.3%; P = .158). In vitro modulation of P-gp activity by Zosuquidar and expression of P-gp, multidrug resistance-related protein 1, lung resistance protein, and breast cancer resistance protein, were comparable in the 2 arms. Poor-risk cytogenetics were more common in P-gp(+) patients. P-gp expression and cytogenetics were correlated, though independent prognostic factors. We conclude that Zosuquidar did not improve outcome in older acute myeloid leukemia, in part, because of the presence P-gp independent mechanisms of resistance. This trial is registered at www.clinicaltrials.gov as #NCT00046930.
Analysis of the inhibition potential of zosuquidar derivatives on selected bacterial and fungal ABC transporters.[Pubmed:23356389]
Mol Membr Biol. 2013 Mar;30(2):217-27.
The increasing number of multidrug-resistant pathogenic microorganisms is a serious public health issue. Among the multitude of mechanisms that lead to multidrug resistance, the active extrusion of toxic compounds, mediated by MDR efflux pumps, plays an important role. In our study we analyzed the inhibitory capability of 26 synthesized Zosuquidar derivatives on three ABC-type MDR efflux pumps, namely Saccharomyces cerevisiae Pdr5 as well as Lactococcus lactis LmrA and LmrCD. For Pdr5, five compounds could be identified that inhibited rhodamine 6G transport more efficiently than Zosuquidar. One of these is a compound with a new catechol acetal structure that might represent a new lead compound. Furthermore, the determination of IC(50) values for rhodamine 6G transport of Pdr5 with representative compounds reveals values between 0.3 and 0.9 muM. Thus the identified compounds are among the most potent inhibitors known for Pdr5. For the ABC-type efflux pumps LmrA and LmrCD from L. lactis, seven and three compounds, which inhibit the transport activity more than the lead compound Zosuquidar, were found. Interestingly, transport inhibition for LmrCD was very specific, with a drastic reduction by one compound while its diastereomers showed hardly an effect. Thus, the present study reveals new potent inhibitors for the ABC-type MDR efflux pumps studied with the inhibitors of Pdr5 and LmrCD being of particular interest as these proteins are well known model systems for their homologs in pathogenic fungi and Gram-positive bacteria.


